Physicochemical Properties
| Molecular Formula | C14H18N2O3 |
| Molecular Weight | 262.3043 |
| Exact Mass | 262.131 |
| CAS # | 1292285-54-1 |
| PubChem CID | 52911386 |
| Appearance | Off-white to gray solid powder |
| LogP | 1.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 19 |
| Complexity | 311 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | YBXBWBBVLXZQBJ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H18N2O3/c1-9-11(5-6-15-14(18)8-19-2)12-7-10(17)3-4-13(12)16-9/h3-4,7,16-17H,5-6,8H2,1-2H3,(H,15,18) |
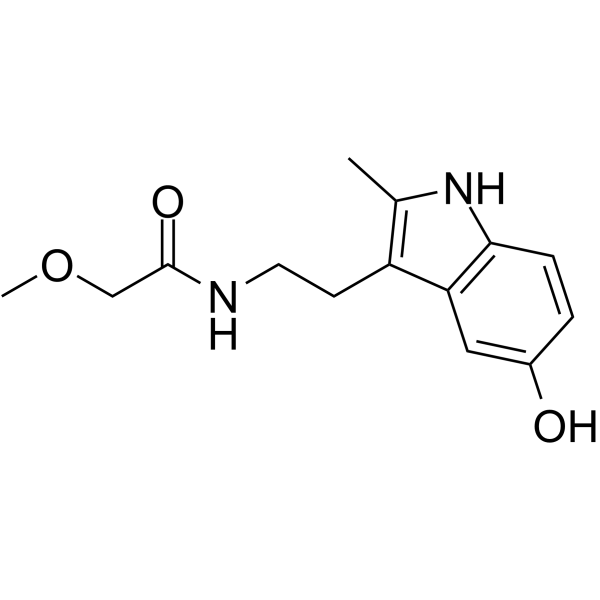
| Chemical Name | N-[2-(5-hydroxy-2-methyl-1H-indol-3-yl)ethyl]-2-methoxyacetamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | SPR inhibitor 3 (SPRi3) also considerably lowers SPR activity in primary cultures of mouse sensory neurons (IC50=0.45 μM) without influencing the activity of GTP ring hydroxylase 1 enzyme (GCH1) [1] . |
| References |
[1]. Latremoliere A, et al. Reduction of Neuropathic and Inflammatory Pain through Inhibition of the Tetrahydrobiopterin Pathway. Neuron. 2015 Jun 17;86(6):1393-406. [2]. Cronin SJF, et al. The metabolite BH4 controls T cell proliferation in autoimmunity and cancer. Nature. 2018 Nov;563(7732):564-568. [3]. Haruki H, et al. Tetrahydrobiopterin Biosynthesis as a Potential Target of the Kynurenine Pathway Metabolite Xanthurenic Acid. J Biol Chem. 2016 Jan 8;291(2):652-7. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~381.24 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.53 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.53 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (9.53 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.8124 mL | 19.0621 mL | 38.1243 mL | |
| 5 mM | 0.7625 mL | 3.8124 mL | 7.6249 mL | |
| 10 mM | 0.3812 mL | 1.9062 mL | 3.8124 mL |