Physicochemical Properties
| Molecular Formula | C16H21NO6S |
| Molecular Weight | 355.406043767929 |
| Exact Mass | 355.108 |
| Elemental Analysis | C, 54.07; H, 5.96; N, 3.94; O, 27.01; S, 9.02 |
| CAS # | 911714-45-9 |
| Related CAS # | 911714459; 911714 45 9; 911714-45-9; |
| PubChem CID | 135511704 |
| Appearance | Typically exists as Light yellow to khaki solids at room temperature |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 562.2±60.0 °C at 760 mmHg |
| Flash Point | 293.8±32.9 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.580 |
| LogP | -0.07 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 24 |
| Complexity | 460 |
| Defined Atom Stereocenter Count | 1 |
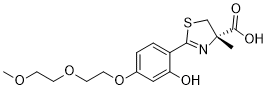
| SMILES | OC1C=C(OCCOCCOC)C=CC=1C1SC[C@](C)(C(=O)O)N=1 |
| InChi Key | YASYAEVZKXPYIZ-MRXNPFEDSA-N |
| InChi Code | InChI=1S/C16H21NO6S/c1-16(15(19)20)10-24-14(17-16)12-4-3-11(9-13(12)18)23-8-7-22-6-5-21-2/h3-4,9,18H,5-8,10H2,1-2H3,(H,19,20)/t16-/m1/s1 |
| Chemical Name | (4S)-4,5-Dihydro-2-[2-hydroxy-4-[2-(2-methoxyethoxy)ethoxy]phenyl]-4-methyl-4-thiazolecarboxylic acid |
| Synonyms | SP-420; SP420; UNII-WBX54NZ436; WBX54NZ436; (4S)-2-[2-hydroxy-4-[2-(2-methoxyethoxy)ethoxy]phenyl]-4-methyl-5H-1,3-thiazole-4-carboxylic acid; (4S)-2-{2-Hydroxy-4-[2-(2-methoxyethoxy)ethoxy]phenyl}-4-methyl-4,5-dihydro-1,3-thiazole-4-carboxylic acid; (4S)-4,5-Dihydro-2-(2-hydroxy-4-(2-(2-methoxyethoxy)ethoxy)phenyl)-4-methyl-4-thiazolecarboxylic acid; (S)-2-(2-hydroxy-4-(2-(2-methoxyethoxy)ethoxy)phenyl)-4-methyl-4,5-dihydrothiazole-4-carboxylic acid;SP 420 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Iron chelator |
| ln Vitro | Desferrithiocin (DFT, 1) is a very efficient iron chelator when given orally. However, it is severely nephrotoxic. Structure-activity studies with 1 demonstrated that removal of the aromatic nitrogen to provide desazadesferrithiocin (DADFT, 2) and introduction of either a hydroxyl group or a polyether fragment onto the aromatic ring resulted in orally active iron chelators that were much less toxic than 1. The purpose of the current study was to determine if a comparable reduction in renal toxicity could be achieved by performing the same structural manipulations on 1 itself[1]. |
| ln Vivo | The iron-clearing efficiency and ferrokinetics were evaluated in rats and primates; toxicity assessments were carried out in rodents. The resulting DFT ligands demonstrated a reduction in toxicity that was equivalent to that of the DADFT analogues and presented with excellent iron-clearing properties[1]. |
| Enzyme Assay | The stoichiometries of the ligand-Fe(III) complexes of 6, 8 and 10 were determined spectrophotometrically using Job’s plots. Solutions were monitored at the visible λmax of the Fe(III) complexes (498 nm for 6, 484 nm for 8, and 485 nm for 10). A 100 mM Tris HCl buffer was used to maintain the pH at 7.4. Solutions containing different ligand/Fe(III) ratios were prepared by mixing appropriate volumes of 1.0 mM ligand solution and 1.0 mM Fe(III)-nitriloacetate (NTA) in Tris-HCl buffer. The 1.0 mM Fe(III)-NTA solution was prepared immediately prior to use by dilution of a 41.6 mM Fe(III)-NTA stock solution with the Tris HCl buffer, whereas the ligand’s stock solution was prepared by dissolving the ligand as its monosodium salt in Tris HCl buffer at pH 7.4. The Fe(III)-NTA stock solution was prepared by mixing equal volumes of 90 mM of FeCl3 and 180 mM trisodium NTA. The iron content of the Fe(III)-NTA solution was verified by AAS[1]. |
| Animal Protocol |
Cannulation of Bile Duct in Non-iron-overloaded Rats[1] The cannulation has been described previously. Bile samples were collected from male Sprague-Dawley rats (400–450 g) at 3 h intervals for up to 48 h. The urine sample(s) was taken at 24 h intervals. Sample collection and handling are as previously described.[1] Iron Loading of C. apella Monkeys[1] The monkeys were iron overloaded with intravenous iron dextran as specified in earlier publications to provide about 500 mg of iron per kg of body weight; the serum transferrin iron saturation rose to between 70 and 80%. At least 20 half-lives, 60 days, elapsed before any of the animals were used in experiments evaluating iron-chelating agents. |
| References |
[1]. Substituent effects on desferrithiocin and desferrithiocin analogue iron-clearing and toxicity profiles. J Med Chem. 2012 Aug 23;55(16):7090-103. [2]. Understanding the intricacies of iron overload associated with β-thalassemia: a comprehensive review. Thalassemia Reports, 2023, 13(3): 179-194. [3]. Safety and pharmacokinetics of the oral iron chelator SP‐420 in β‐thalassemia. American Journal of Hematology, 2017, 92(12): 1356-1361. |
| Additional Infomation |
SP-420 is under investigation in clinical trial NCT03801889 (SP-420 in Subjects With Transfusion-dependent Beta-thalassemia or Other Rare Anemias). Iron Chelating Agent SP-420 is an orally bioavailable iron-chelating agent and derivative of desferrithiocin, with iron chelating and protective activities in diseases of iron overload. Upon oral administration, the iron chelating agent SP-420 targets, binds to and chelates free iron. This induces the excretion of iron, prevents iron accumulation and prevents cellular and/or tissue damage associated with iron overload. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 38 mg/mL (~106.92 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8137 mL | 14.0683 mL | 28.1365 mL | |
| 5 mM | 0.5627 mL | 2.8137 mL | 5.6273 mL | |
| 10 mM | 0.2814 mL | 1.4068 mL | 2.8137 mL |