Physicochemical Properties
| Molecular Formula | C45H57N7O6 |
| Molecular Weight | 791.977391004562 |
| Exact Mass | 791.437 |
| CAS # | 2546091-70-5 |
| PubChem CID | 155318440 |
| Appearance | Brown to reddish brown solid powder |
| LogP | 1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 58 |
| Complexity | 1800 |
| Defined Atom Stereocenter Count | 0 |
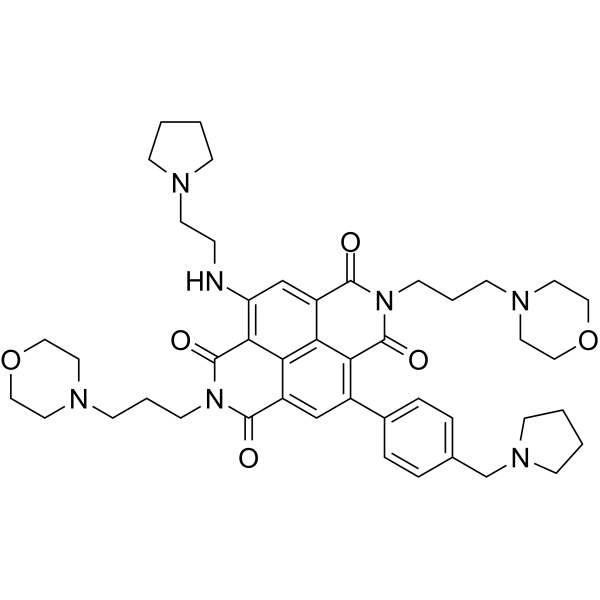
| SMILES | OC1=C2C=C(C3C=CC(=CC=3)CN3CCCC3)C3C(N(CCCN4CCOCC4)C(C4=C/C(/C(C(N1CCCN1CCOCC1)=O)=C2C=34)=N\CCN1CCCC1)=O)=O |
| InChi Key | TZQVPFRBHPQZNM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C45H57N7O6/c53-42-35-29-34(33-9-7-32(8-10-33)31-50-14-3-4-15-50)40-38-36(43(54)51(44(40)55)18-5-16-48-21-25-57-26-22-48)30-37(46-11-20-47-12-1-2-13-47)41(39(35)38)45(56)52(42)19-6-17-49-23-27-58-28-24-49/h7-10,29-30,53H,1-6,11-28,31H2 |
| Chemical Name | 14-hydroxy-6,13-bis(3-morpholin-4-ylpropyl)-10-(2-pyrrolidin-1-ylethylimino)-3-[4-(pyrrolidin-1-ylmethyl)phenyl]-6,13-diazatetracyclo[6.6.2.04,16.011,15]hexadeca-1(14),2,4(16),8,11(15)-pentaene-5,7,12-trione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | HuTel21 G4 and hTERT G4 have a quarterly affinity for SOP1812 (0 -800 nM; 6-24 h) [1]. 40 nM; 6–24 hours) impacts the pigments Rap1, Hippo, MAPK, and Wnt/β-catenin [1]. |
| ln Vivo | MIA PaCa-2 xenograft mice and KPC transplanted mice demonstrated anti-tumor activity in response to intravenous administration of SOP1812 (1 mg/kg) once or twice weekly for 28 days [1]. |
| Cell Assay |
Cell Proliferation Assay[1] Cell Types: MIA PaCa-2, PANC-1, Capan- 1 and BxPC-3 Cell Line Tested Concentrations: 0-50 nM Incubation Duration: 96 hrs (hours) Experimental Results: For MIA PaCa-2, PANC-1, Capan-1 and BxPC-3 cells have anti-proliferative abilities, with GI50 values of 1.3, 1.4, 5.9 and 2.6 nM, respectively. Cell viability assay [1] Cell Types: PANC-1 Cell Tested Concentrations: 0, 100, 400 and 800 nM Incubation Duration: 6 and 24 hrs (hours) Experimental Results: Binding to hTERT G4 and HuTel21 G4, KD values were 4.9 and 28.4 nM respectively. Cell viability assay[1] Cell Types: MIA PaCa-2 Cell Tested Concentrations: 40 nM Incubation Duration: 6 hrs (hours) and 24 hrs (hours) Experimental Results: WNT5B, DVL1, AXIN and APC2 expression were affected, including the Wnt/β-catenin pathway, and on Axon guidance, Hippo, MAPK and Rap1 pathways. |
| Animal Protocol |
Animal/Disease Models: Female athymic nude mice using MIA PaCa-2 xenografts [1] Doses: 1 mg/kg Route of Administration: intravenous (iv) (iv)injection; 1 mg/kg once or twice weekly; 28-day Experimental Results: Day 28 After several days, several animals had complete tumor regression with no obvious tumor regrowth. Animal/Disease Models: KPC mice with PDAC symptoms [1] Doses: 1 mg/kg Route of Administration: intravenous (iv) (iv)injection; 1 mg/kg, once a week; 3 consecutive weeks Experimental Results: Dramatically prolonged the survival of KPC mice, And the effect is better than gemcitabine. |
| References |
[1]. Asymmetrically Substituted Quadruplex-Binding Naphthalene Diimide Showing Potent Activity in Pancreatic Cancer Models. ACS Med Chem Lett. 2020 Jul 16;11(8):1634-1644. |
| Additional Infomation | G-Quadruplex-selective Transcription Inhibitor QN-302 is a naphthalene diimide (ND) derivative and selective G-quadruplex (G4) transcription inhibitor, with potential antineoplastic activity. Upon administration, QN-302 targets and binds to highly stable G-quadruplex DNA sequences that are prevalent within the promoter regions of cancer-related genes, thereby further stabilizing these complexes. This stabilization prevents unwinding and inhibits transcription factor binding, which leads to an inhibition of target gene transcription and expression and decreases G4-expressing cancer cell proliferation. Specifically, QN-302 downregulates the expression of the S100P gene in tumor cells. S100P, forming many quadruplex-forming sequences, plays a key role in the proliferation and motility pathways in several cancers and is upregulated in various tumor cell types. G4 sequences are over-represented in the promoter regions of many oncogenes. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~200 mg/mL (~252.53 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2627 mL | 6.3133 mL | 12.6266 mL | |
| 5 mM | 0.2525 mL | 1.2627 mL | 2.5253 mL | |
| 10 mM | 0.1263 mL | 0.6313 mL | 1.2627 mL |