Physicochemical Properties
| Molecular Formula | C14H17N5O |
| Molecular Weight | 271.32 |
| Exact Mass | 271.143 |
| CAS # | 1997319-92-2 |
| PubChem CID | 137045944 |
| Appearance | Light yellow to light brown solid powder |
| LogP | 0.3 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 20 |
| Complexity | 310 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | SZKHGLTYXIDOFH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H17N5O/c15-14-12(19-7-5-16-6-8-19)9-11(17-18-14)10-3-1-2-4-13(10)20/h1-4,9,16,20H,5-8H2,(H2,15,18) |
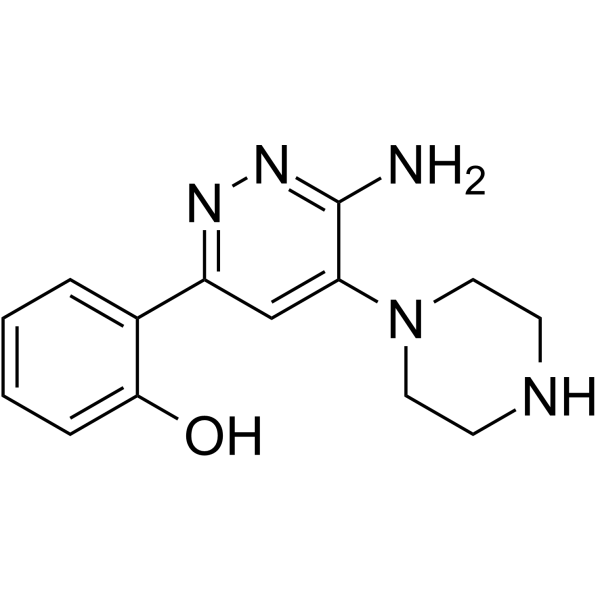
| Chemical Name | 2-(6-amino-5-piperazin-1-ylpyridazin-3-yl)phenol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
SMARCA2 Bromodomain 1 (SMARCA2 BD1) (Ki = 0.12 μM, ITC; Ki = 0.11 μM, AlphaScreen) [1] SMARCA4 Bromodomain 1 (SMARCA4 BD1) (Ki = 0.15 μM, ITC; Ki = 0.13 μM, AlphaScreen) [1] Other bromodomains (BRD4 BD1, BRD2 BD1, BRD3 BD1, BRD9 BD) (Ki > 10 μM, AlphaScreen) [1] |
| ln Vitro |
SMARCA-BD ligand 1 for Protac is a structure-based, high-affinity and selective ligand targeting the bromodomain 1 (BD1) of SMARCA2 and SMARCA4 (key subunits of the BAF chromatin remodeling complex). [1] It exhibits potent binding to SMARCA2 BD1 and SMARCA4 BD1: ITC assays confirm direct binding with Ki values of 0.12 μM (SMARCA2 BD1) and 0.15 μM (SMARCA4 BD1), while AlphaScreen assays yield consistent Ki values of 0.11 μM and 0.13 μM, respectively. [1] The ligand shows exceptional subtype selectivity: It has negligible binding affinity for other bromodomains (e.g., BRD4 BD1, BRD2 BD1, BRD3 BD1, BRD9 BD) with Ki > 10 μM, representing a selectivity ratio >80-fold for SMARCA BD1 over non-target bromodomains. [1] It acts as a competitive inhibitor of acetylated lysine (Kac) binding to SMARCA BD1: AlphaScreen competition assays demonstrate that SMARCA-BD ligand 1 for Protac dose-dependently displaces a Kac-containing peptide from SMARCA2/4 BD1, confirming binding to the canonical acetyllysine recognition pocket of the bromodomain. [1] |
| Enzyme Assay |
Isothermal Titration Calorimetry (ITC) for SMARCA BD binding affinity: Recombinant SMARCA2 BD1 or SMARCA4 BD1 protein was dialyzed against a buffer system and placed in the ITC sample cell. SMARCA-BD ligand 1 for Protac dissolved in the same buffer was titrated into the sample cell in sequential injections. The heat change associated with each binding event was recorded, and data were fitted to a one-site binding model to calculate the dissociation constant (Ki), enthalpy change (ΔH), and entropy change (ΔS). [1] AlphaScreen bromodomain-peptide competition assay: Streptavidin-coated donor beads and anti-GST acceptor beads were used. GST-tagged SMARCA BD1 protein was incubated with biotinylated acetyllysine (Kac) peptide and serial dilutions of SMARCA-BD ligand 1 for Protac at room temperature for 1 hour. Donor and acceptor beads were added to the mixture, and after further incubation, the AlphaScreen signal (luminescence) was measured. The inhibition of the BD1-Kac peptide interaction by the ligand was quantified, and Ki values were derived from dose-response curves. [1] Surface Plasmon Resonance (SPR) for binding kinetics: SMARCA2 BD1 or SMARCA4 BD1 protein was covalently immobilized on a sensor chip. SMARCA-BD ligand 1 for Protac was injected over the chip at serial concentrations (0.01 μM-10 μM) at a constant flow rate. The association rate (ka), dissociation rate (kd), and equilibrium dissociation constant (KD = Ki) were calculated by fitting the real-time resonance signal curves to a 1:1 binding model. [1] |
| Cell Assay |
Intracellular SMARCA BD binding validation assay: HEK293T cells were transiently transfected with GST-tagged SMARCA2 BD1 or SMARCA4 BD1 expression plasmids. After 24 hours, cells were treated with SMARCA-BD ligand 1 for Protac (1 μM) for 4 hours. Cell lysates were prepared and incubated with glutathione beads to pull down GST-SMARCA BD1. The beads were washed, and bound ligand was detected by LC-MS/MS analysis to confirm intracellular binding. [1] Bromodomain selectivity cellular assay: Cells expressing different GST-tagged bromodomains (SMARCA2 BD1, SMARCA4 BD1, BRD4 BD1, BRD2 BD1) were treated with SMARCA-BD ligand 1 for Protac (1 μM) as described above. GST pull-down followed by LC-MS/MS was used to quantify ligand binding to each bromodomain, confirming selective intracellular binding to SMARCA BD1. [1] |
| References |
[1]. BAF complex vulnerabilities in cancer demonstrated via structure-based PROTAC design. Nat Chem Biol. 2019 Jul;15(7):672-680. |
| Additional Infomation |
SMARCA-BD ligand 1 for Protac is a structure-based designed small molecule, developed through virtual screening and medicinal chemistry optimization targeting the bromodomain 1 (BD1) of SMARCA2 and SMARCA4—core subunits of the mammalian SWI/SNF (BAF) chromatin remodeling complex. [1] The BAF complex is frequently mutated or dysregulated in various cancers (e.g., ovarian cancer, lung cancer, synovial sarcoma), making SMARCA2/4 attractive therapeutic targets. SMARCA-BD ligand 1 for Protac serves as a high-selectivity targeting warhead for the construction of PROTAC molecules, which induce ubiquitination and proteasomal degradation of SMARCA2/4. [1] The ligand’s binding mode is characterized by X-ray crystallography: It occupies the acetyllysine recognition pocket of SMARCA BD1, forming key hydrogen bonds with conserved residues (e.g., Asn156 in SMARCA2) and hydrophobic interactions with the pocket’s aromatic residues, which contributes to its high affinity and selectivity. [1] Unlike full PROTAC molecules, SMARCA-BD ligand 1 for Protac itself does not induce SMARCA2/4 degradation or exhibit antiproliferative activity; its primary function is to provide specific targeting to SMARCA BD1, enabling the PROTAC to recruit an E3 ubiquitin ligase (e.g., CRBN) to the BAF complex for selective degradation of SMARCA subunits. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6857 mL | 18.4284 mL | 36.8568 mL | |
| 5 mM | 0.7371 mL | 3.6857 mL | 7.3714 mL | |
| 10 mM | 0.3686 mL | 1.8428 mL | 3.6857 mL |