Physicochemical Properties
| Molecular Formula | C59H67F2N7O11S |
| Molecular Weight | 1120.26540112495 |
| Exact Mass | 1119.458 |
| CAS # | 2254609-27-1 |
| PubChem CID | 137628677 |
| Appearance | White to off-white solid powder |
| LogP | 8.7 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 27 |
| Heavy Atom Count | 80 |
| Complexity | 2010 |
| Defined Atom Stereocenter Count | 3 |
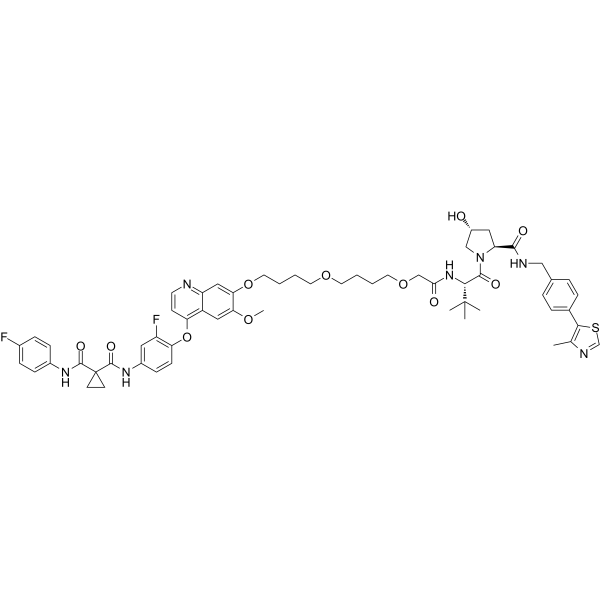
| SMILES | S1C=NC(C)=C1C1C=CC(=CC=1)CNC([C@@H]1C[C@H](CN1C([C@H](C(C)(C)C)NC(COCCCCOCCCCOC1C(=CC2C(C=1)=NC=CC=2OC1C=CC(=CC=1F)N(C1C=CC(=CC=1)F)C(C1(C(N)=O)CC1)=O)OC)=O)=O)O)=O |
| InChi Key | GXDYWQXTEYENEU-WFYKIECOSA-N |
| InChi Code | InChI=1S/C59H67F2N7O11S/c1-36-52(80-35-64-36)38-12-10-37(11-13-38)32-63-54(71)46-29-42(69)33-68(46)55(72)53(58(2,3)4)67-51(70)34-77-26-7-6-24-76-25-8-9-27-78-50-31-45-43(30-49(50)75-5)47(20-23-62-45)79-48-19-18-41(28-44(48)61)66-57(74)59(21-22-59)56(73)65-40-16-14-39(60)15-17-40/h10-20,23,28,30-31,35,42,46,53,69H,6-9,21-22,24-27,29,32-34H2,1-5H3,(H,63,71)(H,65,73)(H,66,74)(H,67,70)/t42-,46+,53-/m1/s1 |
| Chemical Name | 1-N'-[3-fluoro-4-[7-[4-[4-[2-[[(2S)-1-[(2S,4R)-4-hydroxy-2-[[4-(4-methyl-1,3-thiazol-5-yl)phenyl]methylcarbamoyl]pyrrolidin-1-yl]-3,3-dimethyl-1-oxobutan-2-yl]amino]-2-oxoethoxy]butoxy]butoxy]-6-methoxyquinolin-4-yl]oxyphenyl]-1-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
SJFα targets bromodomain-containing protein 4 (BRD4) (DC50 = 11 nM for BRD4 BD1/BD2 degradation in MV4;11 cells) [1] SJFα targets Cereblon (CRBN) ( functions as a CRBN-recruiting PROTAC warhead) [1] |
| ln Vitro |
- BRD4 degradation activity: SJFα is a proteolysis-targeting chimera (PROTAC) that dose-dependently induces BRD4 degradation in MV4;11 (acute myeloid leukemia), OCI-LY10 (diffuse large B-cell lymphoma), and HeLa cells. In MV4;11 cells, DC50 = 11 nM, and maximum degradation efficiency (Dmax) = 92% at 1 μM. Degradation was detectable as early as 2 hours post-treatment and sustained for 24 hours [1] - Selective degradation of BRD4: The compound specifically degrades BRD4 without affecting other bromodomain-containing proteins (BRD2, BRD3, BRD7, BRD9) or CRBN. In MV4;11 cells, BRD2 and BRD3 protein levels remained unchanged even at 10 μM SJFα [1] - Dependence on CRBN and proteasome: BRD4 degradation induced by SJFα (1 μM) was completely blocked in CRBN-knockout MV4;11 cells (degradation reduced by 95%) or in cells treated with proteasome inhibitor (MG132, 10 μM). This confirms CRBN-dependent, proteasome-mediated degradation [1] - Induction of BRD4 ubiquitination: SJFα (0.5 μM, 1 μM) dose-dependently increased ubiquitination levels of BRD4 in MV4;11 cells. Immunoprecipitation assay showed enhanced interaction between BRD4 and CRBN in the presence of SJFα [1] - Antiproliferative activity: SJFα inhibited proliferation of BRD4-dependent cancer cell lines. IC50 values were 32 nM (MV4;11), 45 nM (OCI-LY10), and 120 nM (HeLa). It had minimal effect on CRBN-knockout MV4;11 cells (IC50 > 1 μM) [1] |
| Enzyme Assay |
- BRD4 binding assay: Recombinant human BRD4 BD1/BD2 domains were immobilized on a sensor chip. SJFα at gradient concentrations (1 nM-1 μM) was injected, and binding affinity was measured by surface plasmon resonance (SPR). The equilibrium dissociation constant (KD) for BRD4 BD1 was 4.2 nM, and for BD2 was 6.8 nM [1] - CRBN binding validation assay: Recombinant human CRBN (complexed with DDB1) was incubated with SJFα (1 μM) and a fluorescently labeled CRBN ligand competitor. Fluorescence polarization was measured to assess competitive binding, confirming SJFα binds to the CRBN ligand-binding pocket [1] - In vitro ubiquitination assay: Purified BRD4, CRBN-DDB1 complex, E1, E2, ubiquitin, and SJFα (0.1-1 μM) were mixed in ubiquitination buffer (pH 7.5) and incubated at 37°C for 2 hours. Ubiquitinated BRD4 was detected by western blot with ubiquitin-specific antibody [1] |
| Cell Assay |
- BRD4 degradation western blot assay: MV4;11, OCI-LY10, or HeLa cells were seeded into 6-well plates (5×10⁵ cells/well) and incubated overnight. Cells were treated with SJFα (0.1 nM-10 μM) for 6 hours (concentration-dependent) or 0-24 hours (time-dependent). Cells were lysed, proteins separated by SDS-PAGE, and BRD4, BRD2, BRD3, CRBN, and GAPDH (loading control) detected by western blot. Band intensities were quantified to calculate DC50 and Dmax [1] - CRBN dependence assay: CRBN-knockout and wild-type MV4;11 cells were treated with SJFα (1 μM) for 6 hours. BRD4 protein levels were detected by western blot to compare degradation efficiency [1] - Proteasome dependence assay: MV4;11 cells were pre-treated with MG132 (10 μM) for 1 hour, then co-treated with SJFα (1 μM) for 6 hours. BRD4 degradation was analyzed by western blot [1] - BRD4-CRBN interaction assay: MV4;11 cells were treated with SJFα (1 μM) for 2 hours, lysed, and BRD4 was immunoprecipitated with BRD4-specific antibody. Co-immunoprecipitated CRBN was detected by western blot to verify ternary complex formation [1] - Cell proliferation assay: Cancer cells (MV4;11, OCI-LY10, HeLa) and CRBN-knockout MV4;11 cells were seeded into 96-well plates (5×10³ cells/well) and treated with SJFα (0.1 nM-10 μM) for 72 hours. Cell viability was measured by tetrazolium salt-based assay, and IC50 values were calculated [1] |
| References |
[1]. Differential PROTAC substrate specificity dictated by orientation of recruited E3 ligase. Nat Commun. 2019 Jan 10;10(1):131. |
| Additional Infomation |
- Chemical classification: SJFα is a heterobifunctional PROTAC composed of three moieties: a BRD4-binding ligand (JQ1 derivative), a CRBN-recruiting ligand (thalidomide derivative), and a flexible polyethylene glycol (PEG) linker [1] - Mechanism of action: SJFα bridges BRD4 (substrate protein) and CRBN (E3 ubiquitin ligase) to form a ternary complex. This recruits the ubiquitin-proteasome system (UPS), promoting ubiquitination of BRD4 and subsequent proteasomal degradation. The substrate specificity is dictated by the orientation of CRBN recruited to BRD4, enabling selective degradation of BRD4 over other BRD family members [1] - Target background: BRD4 is a transcriptional co-activator that binds acetylated histones, regulating expression of oncogenes (e.g., MYC). Overexpression of BRD4 is associated with hematological malignancies and solid tumors. CRBN is a substrate receptor for the CRL4 E3 ubiquitin ligase complex, commonly used in PROTAC design [1] - Research significance: SJFα serves as a tool compound to study PROTAC substrate specificity, demonstrating that E3 ligase orientation is a key determinant of selective protein degradation. It provides a basis for developing BRD4-targeting PROTACs for cancer therapy [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~89.26 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8926 mL | 4.4632 mL | 8.9264 mL | |
| 5 mM | 0.1785 mL | 0.8926 mL | 1.7853 mL | |
| 10 mM | 0.0893 mL | 0.4463 mL | 0.8926 mL |