SJ000291942 is a new small-molecule activator of the canonical BMP (bone morphogenetic proteins) signaling pathway. BMPs are members of the transforming growth factor beta (TGFβ) family of secreted signaling molecules. SJ000291942 was found by high throughput screening of a library of ∼600 000 small molecules. Using a cell-based luciferase assay in the BMP4-responsive human cervical carcinoma clonal cell line, C33A-2D2, SJ000291942 can ventralize zebrafish embryos and stimulate increased expression of the BMP target genes, bmp2b and szl. Because SJ000291942 ventralizes zebrafish embryos, it was termed as 'ventromorphins.' As expected for a BMP pathway activator, SJ000291942 induces the differentiation of C2C12 myoblasts to osteoblasts. Affymetrix RNA analysis confirmed the differentiation results and showed that SJ000291942 treatment elicits a genetic response similar to BMP4 treatment. Unlike isoliquiritigenin (SJ000286237), a flavone that maximally activates the pathway after 24 h of treatment, SJ000291942 induced SMAD1/5/8 phosphorylation within 30 min of treatment and achieved peak activity within 1 h, indicating that their responses are consistent with directly activating BMP signaling.
Physicochemical Properties
| Molecular Formula | C16H15FN2O4 | |
| Molecular Weight | 318.30 | |
| Exact Mass | 318.1 | |
| Elemental Analysis | C, 60.37; H, 4.75; F, 5.97; N, 8.80; O, 20.11 | |
| CAS # | 425613-09-8 | |
| Related CAS # |
|
|
| PubChem CID | 875819 | |
| Appearance | White to off-white solid powder | |
| LogP | 3.5 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 23 | |
| Complexity | 407 | |
| Defined Atom Stereocenter Count | 0 | |
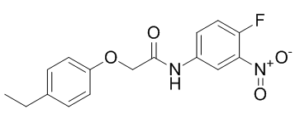
| SMILES | O(CC(NC1C=CC(F)=C(C=1)[N+]([O-])=O)=O)C1C=CC(=CC=1)CC |
|
| InChi Key | YSZJLXPXVFGTNK-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H15FN2O4/c1-2-11-3-6-13(7-4-11)23-10-16(20)18-12-5-8-14(17)15(9-12)19(21)22/h3-9H,2,10H2,1H3,(H,18,20) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Canonical BMP Signaling Pathway
- SJ000291942 activates the canonical BMP signaling pathway by stimulating phosphorylation of SMAD1/5/8, with maximal activity observed within 1 hour of treatment [1] |
| ln Vitro |
SJ000291942 was the most powerful and the most severe ventralization was observed in embryos treated with it. At lesser dosages, SJ000291942 likewise produced greater mortality than the other two drugs and the control group. This is consistent with an increase in BMP signaling activity and suggests that our drugs induce embryonic ventralization. SJ000291942 led to upregulated bmp2b and szl expression. The conventional BMP signaling pathway is activated by SJ000291842, as demonstrated by zebrafish test. Immunoblotting was done on protein lysates from C33A-2D2 cells treated with SJ000291942 at various intervals in order to expand on these findings. In a serum-free media, SJ000291942 triggers the phosphorylation of SMAD1/5/8. The most active was SJ000291842, just like in zebrafish embryos. After one hour of treatment, SJ000291942 maximally promotes p-SMAD1/5/8. Phosphorylated extracellular signal-regulated protein kinase ERK1/2 (P-ERK1/2) was considerably increased by SJ000291942, according to immunoblot examination of C33A-2D2 lysates treated with the drug. The gene expression fingerprints obtained by treatment with the highest dosages (100 and 300ng) of BMP4 were most similar to osteoblastic expression. Treatment with low dose (10ng) BMP4 was closely associated with treatment with 25μM Compound 3 and 25μM SJ000291942[1].
- BMP Signaling Activation: 1. Luciferase Reporter Assay: SJ000291942 (1–10 μM) induced dose-dependent activation of a BMP-responsive luciferase reporter gene in C33A-2D2 cells, with maximal activity at 10 μM [1] 2. Western Blot Analysis: Treatment of C33A-2D2 cells with SJ000291942 (10 μM) for 1 hour resulted in robust phosphorylation of SMAD1/5/8 and ERK1/2, consistent with activation of both canonical and non-canonical BMP signaling pathways [1] - Osteogenic Differentiation: 1. C2C12 Myoblasts: SJ000291942 (25 μM) induced osteoblast differentiation, as indicated by increased alkaline phosphatase (ALP) activity and mineralization nodule formation after 7 days of treatment [1] 2. RNA Sequencing: Affymetrix RNA analysis revealed that SJ000291942 treatment induced a gene expression signature similar to BMP4, including upregulation of osteogenic markers (e.g., RUNX2, OSTERIX) [1] |
| ln Vivo |
- Zebrafish Embryo Ventralization: 1. Dose-Dependent Effect: SJ000291942 (0.1–10 μM) caused dose-dependent ventralization of zebrafish embryos, with severe ventralization observed at ≥1 μM. This phenotype was accompanied by increased expression of BMP target genes (bmp2b, szl) and reduced expression of dorsal markers (chordin) [1] 2. Morphological Analysis: Treated embryos displayed shortened axes, reduced head structures, and expanded blood island regions, consistent with enhanced BMP signaling activity [1] |
| Enzyme Assay |
- BMP Receptor Kinase Activity Assay [1]: 1. Reaction Setup: Recombinant BMPR1A kinase domain (10 nM) was incubated with SJ000291942 (0.1–10 μM) and ATP (100 μM) in kinase buffer (50 mM Tris-HCl, pH 7.5, 10 mM MgCl₂, 1 mM DTT). 2. Substrate Phosphorylation: Phosphorylation of a synthetic peptide substrate (KKKRLQGSVYVATR) was detected by ELISA using a phospho-specific antibody. SJ000291942 increased phosphorylation by 2- to 3-fold at 10 μM, indicating direct activation of BMPR1A kinase activity [1] |
| Cell Assay |
- Luciferase Reporter Assay [1]: 1. Cell Culture: C33A-2D2 cells were transfected with a BMP-responsive luciferase reporter plasmid (BRE-luc) and treated with SJ000291942 (0.1–10 μM) in serum-free medium for 24 hours. 2. Detection: Luciferase activity was measured using a luminometer, showing a concentration-dependent increase with an EC₅₀ of 2.3 μM [1] - Osteogenic Differentiation Assay [1]: 1. Cell Treatment: C2C12 myoblasts were cultured in osteogenic medium supplemented with SJ000291942 (25 μM) for 7 days. 2. ALP Staining: ALP activity was visualized using BCIP/NBT staining, and mineralization was assessed by Alizarin Red S staining. Both assays showed osteogenic differentiation compared to vehicle controls [1] |
| Animal Protocol |
- Zebrafish Embryo Treatment [1]: 1. Drug Preparation: SJ000291942 was dissolved in DMSO to prepare a 10 mM stock solution, then diluted in embryo medium to final concentrations of 0.1–10 μM (DMSO ≤0.1%). 2. Exposure: Fertilized zebrafish embryos were incubated in 6-well plates containing SJ000291942 solutions from 4 hours post-fertilization (hpf) to 24 hpf. Embryos were scored for ventralization phenotypes under a stereomicroscope [1] |
| ADME/Pharmacokinetics |
- Distribution: 1. Zebrafish Embryo Uptake: SJ000291942 (1 μM) accumulated in zebrafish embryos within 2 hours of exposure, with highest concentrations detected in the developing somites and neural tube [1] |
| Toxicity/Toxicokinetics |
- Embryotoxicity: 1. Zebrafish Studies: SJ000291942 caused dose-dependent mortality in zebrafish embryos, with 50% lethality (LC₅₀) at 8.7 μM after 24 hours of exposure [1] |
| References |
[1]. Ventromorphins: A New Class of Small Molecule Activators of the Canonical BMP Signaling Pathway. ACS Chem Biol. 2017 Sep 15;12(9):2436-2447. |
| Additional Infomation |
- Mechanism of Action: 1. SJ000291942 directly binds to BMP type I receptors (BMPR1A/B) and enhances their kinase activity, leading to SMAD1/5/8 phosphorylation and transcriptional activation of BMP target genes [1] - Structure-Activity Relationship (SAR): 1. The pyrimidine core and 4-chlorophenyl substituent of SJ000291942 are critical for BMP signaling activation, as demonstrated by reduced activity in analogs lacking these moieties [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.85 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.85 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1417 mL | 15.7085 mL | 31.4169 mL | |
| 5 mM | 0.6283 mL | 3.1417 mL | 6.2834 mL | |
| 10 mM | 0.3142 mL | 1.5708 mL | 3.1417 mL |