Physicochemical Properties
| Molecular Formula | C26H31FN4O2 |
| Molecular Weight | 450.548349618912 |
| Exact Mass | 450.243 |
| CAS # | 1849603-72-0 |
| PubChem CID | 118610427 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 4.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 33 |
| Complexity | 642 |
| Defined Atom Stereocenter Count | 0 |
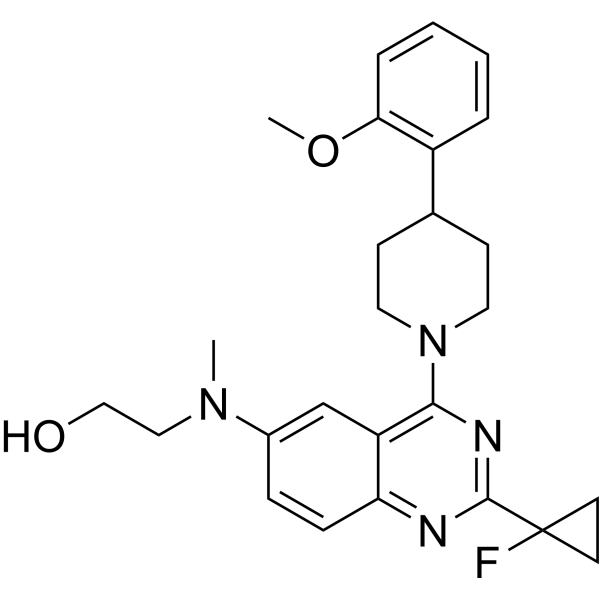
| SMILES | FC1(C2N=C3C=CC(=CC3=C(N=2)N2CCC(C3C=CC=CC=3OC)CC2)N(C)CCO)CC1 |
| InChi Key | BLWXTJQMEBQCIZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H31FN4O2/c1-30(15-16-32)19-7-8-22-21(17-19)24(29-25(28-22)26(27)11-12-26)31-13-9-18(10-14-31)20-5-3-4-6-23(20)33-2/h3-8,17-18,32H,9-16H2,1-2H3 |
| Chemical Name | 2-[[2-(1-fluorocyclopropyl)-4-[4-(2-methoxyphenyl)piperidin-1-yl]quinazolin-6-yl]-methylamino]ethanol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
SBI-553 is a β-arrestin-biased allosteric modulator of NTR1. In a β-arrestin2-GFP high-content assay, it exhibited an EC50 of 0.34 μM and did not affect the EC50 of neurotensin (NT) peptide in this assay (EC50 for NT ranged from 0.37 to 0.40 nM across tested concentrations of SBI-553). In contrast, in a Ca2+ flux assay, SBI-553 functionally acted like a competitive antagonist, shifting the EC50 of NT to the right by more than 30-fold (from 0.035 nM to 1.18 nM in the presence of 2.15 μM SBI-553). In a radioligand binding assay, SBI-553 enhanced the binding of radiolabeled neurotensin to human and mouse NTR1 (with an EC50 of 0.14 μM for enhancement at human NTR1 and a maximum efficacy of 140%), but not to human NTR2, confirming its selectivity and allosteric mechanism. SBI-553 showed no cross-reactivity against NTR2, GPR35, GPR55, and K-opioid receptors in the same high-content assay format. In a broad panel screening against 80 targets at 10 μM, SBI-553 showed moderate promiscuity, with significant binding (>30%) observed at 10 receptors. Subsequent dose titrations indicated binding at adrenergic α2A (55% at 5 μM), dopamine D1 (36% at 5 μM), histamine H1 (75% at 5 μM), and 5-HT2B (42% at 5 μM) receptors.[1] |
| ln Vivo |
In dopamine transporter knockout (DAT-/-) mice, a model of hyperdopaminergic activity, SBI-553 attenuated basal hyperlocomotion after both intraperitoneal (i.p., 12 mg/kg) and oral (p.o., 10, 30, 100 mg/kg) administration. Unlike the peptide NTR1 agonist PD149163, which caused significant hypotension and hypothermia, SBI-553 did not show any adverse effects in these animals.[1] |
| Enzyme Assay |
A radioligand binding assay was performed to examine the binding of radiolabeled neurotensin to membranes prepared from cells overexpressing human NTR1, human NTR2, or mouse NTR1. The assay measured the effect of increasing concentrations of SBI-553 on neurotensin binding. Enhancement of binding was observed for human and mouse NTR1, but not for human NTR2, indicating selectivity and an allosteric mechanism of action.[1] |
| Cell Assay |
A high-content assay measuring β-arrestin2-GFP redistribution was used as the primary assay to determine NTR1 modulator activity. Cells expressing NTR1 and β-arrestin2-GFP were treated with test compounds and neurotensin peptide. Redistribution of β-arrestin2-GFP to the membrane upon receptor activation was quantified. A Ca2+ flux assay was used as a counterscreen to assess the effect of compounds on Gq-mediated signaling. Cells expressing NTR1 were loaded with a calcium-sensitive dye, and changes in intracellular calcium upon stimulation with neurotensin peptide in the presence or absence of test compounds were measured.[1] |
| Animal Protocol |
Adult male and female dopamine transporter knockout (DAT-/-) mice on a C57BL/6J background were used. Animals were 12-24 weeks old. For locomotor activity studies, mice were acclimated to open field automated activity monitors for 30 minutes prior to treatment. For intraperitoneal (i.p.) dosing, animals received SBI-553 (12 mg/kg), PD149163 (1 mg/kg), or vehicle (4% DMSO in saline) in a volume of 10 ml/kg. For oral (p.o.) dosing, animals received SBI-553 HCl salt (10, 30, or 100 mg/kg) or vehicle (distilled water) in a volume of 10 ml/kg. Prior to oral dosing studies, animals were habituated to oral dosing via 5 consecutive daily sessions with water. After treatment, animals were immediately returned to activity monitors, and horizontal locomotion was recorded over the next 2 hours using infrared beam breaks.[1] |
| ADME/Pharmacokinetics |
In mouse pharmacokinetic studies following intravenous (i.v., 5 mg/kg) and oral (p.o., 30 mg/kg) administration, SBI-553 showed a plasma clearance (Clp) of 44.8 mL/min/kg, a volume of distribution (Vss) of 6.16 L/kg, a maximum concentration (Cmax) of 1460 ng/mL after oral dosing, an area under the curve (AUC) of 4824 ng·hr/mL after oral dosing, a half-life (t1/2) of 5.28 hours, and an oral bioavailability (%F) of 45%. The brain-to-plasma ratio 1 hour post-dose was 0.54. In rat pharmacokinetic studies (i.v. 5 mg/kg, p.o. 30 mg/kg), SBI-553 showed a plasma clearance of 81.0 mL/min/kg, a volume of distribution of 7.02 L/kg, a Cmax of 3482 ng/mL after oral dosing, an AUC of 2693 ng·hr/mL after oral dosing, a half-life of 2.23 hours, and an oral bioavailability of 48%. The brain-to-plasma ratio 1 hour post-dose was 0.98.[1] |
| Toxicity/Toxicokinetics |
In the dopamine transporter knockout mouse model, administration of SBI-553 did not cause the significant hypotension and hypothermia observed with the peptide NTR1 agonist PD149163, indicating a potentially improved safety profile regarding these specific adverse effects.[1] |
| References |
[1]. Discovery of β-Arrestin Biased, Orally Bioavailable, and CNS Penetrant Neurotensin Receptor 1 (NTR1) Allosteric Modulators. J Med Chem. 2019 Aug 20. [2]. β-Arrestin-Biased Allosteric Modulator of NTSR1 Selectively Attenuates Addictive Behaviors. Cell. 2020 Jun 11;181(6):1364-1379.e14. |
| Additional Infomation |
SBI-553 is an optimized quinazoline-based derivative discovered through structure-activity relationship (SAR) studies aimed at improving the potency and oral bioavailability of the earlier probe compound ML314. It acts as a β-arrestin-biased allosteric modulator of NTR1, selectively enhancing β-arrestin interaction while antagonizing the classical Gq-mediated Ca2+ signaling pathway, a profile described as a "PAM antagonist" for the Ca2+ pathway. This biased signaling profile is suggested to underlie its efficacy in attenuating hyperlocomotion in a hyperdopaminergic mouse model without causing certain adverse effects associated with standard neurotensin ligands. The compound is implicated in potential therapeutic applications for central nervous system disorders involving dopamine signaling, such as aspects of drug addiction.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~17 mg/mL (~37.73 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.62 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.62 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2195 mL | 11.0975 mL | 22.1951 mL | |
| 5 mM | 0.4439 mL | 2.2195 mL | 4.4390 mL | |
| 10 mM | 0.2220 mL | 1.1098 mL | 2.2195 mL |