Physicochemical Properties
| Molecular Formula | C21H19N4O2CL.2[HCL] |
| Molecular Weight | 467.77604 |
| Exact Mass | 466.073 |
| Elemental Analysis | C, 53.92; H, 4.53; Cl, 22.74; N, 11.98; O, 6.84 |
| CAS # | 181632-25-7 |
| Related CAS # | SB 242084 dihydrochloride;1049747-87-6 |
| PubChem CID | 3644637 |
| Appearance | Off-white to pink solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 620.1±55.0 °C at 760 mmHg |
| Flash Point | 328.8±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.671 |
| LogP | 3.76 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 28 |
| Complexity | 551 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | GCMNSEILNIPNSX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H19ClN4O2.2ClH/c1-13-10-15-7-9-26(18(15)11-17(13)22)21(27)25-16-5-6-20(24-12-16)28-19-4-3-8-23-14(19)2;;/h3-6,8,10-12H,7,9H2,1-2H3,(H,25,27);2*1H |
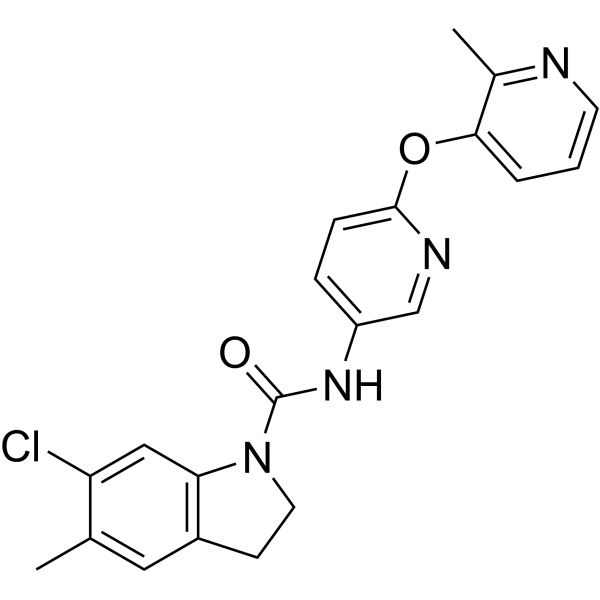
| Chemical Name | 6-chloro-5-methyl-N-[6-(2-methylpyridin-3-yl)oxypyridin-3-yl]-2,3-dihydroindole-1-carboxamide;dihydrochloride |
| Synonyms | 6-Chloro-5-methyl-N-(6-((2-methylpyridin-3-yl)oxy)pyridin-3-yl)indoline-1-carboxamide dihydrochloride; SB 242084 hydrochloride; SB 242084 (hydrochloride); SB-242084 Dihydrochloride; SB 242084 dihydrochloride; SB 242084 dihydrochloride hydrate; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT2C Receptor (pKi = 9.0) |
| ln Vitro | Multiple antagonistic effects of SB 242084 (100 nM; 45 min) on the increase in phosphatidylinositol workstation of 5-HT in human 5-HT2C workstation in SH-SY5Y cells have been reported [1]. SB 242084 (1–100 Nm; twenty-four hours) |
| ln Vivo | In social interaction tests, SB 242084 (0.1–1 mg/kg; i.p.; single dosage; 20 min pretest) improves behavior [1]. SB 242084 (160-640 μg/kg; IV; single dose) dose-dependently and significantly increases basal current rates in dopaminergic neurons in the VTA (ventral tegmental area), as well as intrabody (DOPAC) levels. SB 242084 (5 mg/kg; i.p.; single dose; 20 minutes) SB 242084 (5, 10 mg/kg; i.p.; single dose) increases basal dialysate dopamine (DA) and hexaphenyl forms in the nucleus accumbens [ 3]. The same region also experiences increased burst activity [3]. |
| Enzyme Assay | SB 242084 has a high affinity (pKi 9.0) for the cloned human 5-HT2C receptor and 100- and 158-fold selectivity over the closely related cloned human 5-HT2B and 5-HT2A subtypes respectively. SB 242084 had over 100-fold selectivity over a range of other 5-HT, dopamine and adrenergic receptors. In studies of 5-HT-stimulated phosphatidylinositol hydrolysis using SH-SY5Y cells stably expressing the cloned human 5-HT2C receptor, SB 242084 acted as an antagonist with a pKb of 9.3, which closely resembled its corresponding receptor binding affinity[1]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: SH-SY5Y cells Tested Concentrations: 100 nM Incubation Duration: 45 min Experimental Results: Antagonizes 5-HT-induced concentration-related increase) Increases RPTC in RPTC Respiration and PGC-1α mRNA expression [2]. In PI hydrolysis. RT-PCR[2] Cell Types: RPTC Cell Tested Concentrations: 1-100 nM Incubation Duration: 24 hrs (hours) Experimental Results: FCCP uncouples respiration and increases PGC-1α mRNA expression. |
| Animal Protocol |
Animal/Disease Models: Male SD (SD (Sprague-Dawley)) (CD) rat [1]. Doses: 0.1-1 mg/kg Route of Administration: intraperitoneal (ip) injection; single; 20 minutes Pre-test Experimental Results: Rats spent Dramatically more time in social interactions over 15 minutes under bright light conditions and in unfamiliar test boxes. Animal/Disease Models: Male SD (SD (Sprague-Dawley)) (CD) rats (mCPP-induced Hypophagia model) [1]. Doses: 5 mg/kg Route of Administration: intraperitoneal (ip) injection; single; 20-minute pre-test Experimental Results: 23 hrs (hrs (hours)) of food deprivation during a 1-hour test period from the date of food presentation The amount of food consumed by rats was Dramatically diminished. Animal/Disease Models: Rat [2]. Doses: 5, 10 mg/kg Route of Administration: intraperitoneal (ip) injection; Single Experimental Results:Basal dialysate dopamine (DA) and diamine in the nucleus accumbens Hydroxyphenylacetic acid (DOPAC) was Dramatically increased. |
| References |
[1]. SB 242084, a selective and brain penetrant 5-HT2C receptor antagonist. Neuropharmacology. 1997 Apr-May;36(4-5):609-20. [2]. 5-HT2 Receptor Regulation of Mitochondrial Genes: Unexpected Pharmacological Effects of Agonists and Antagonists. J Pharmacol Exp Ther. 2016 Apr;357(1):1-9. |
| Additional Infomation |
6-chloro-5-methyl-N-[6-(2-methylpyridin-3-yl)oxypyridin-3-yl]-2,3-dihydroindole-1-carboxamide;dihydrochloride is an organic molecular entity, an indole alkaloid and a pyridine. It has a role as a receptor modulator. SB 242084 has a high affinity (pKi 9.0) for the cloned human 5-HT2C receptor and 100- and 158-fold selectivity over the closely related cloned human 5-HT2B and 5-HT2A subtypes respectively. SB 242084 had over 100-fold selectivity over a range of other 5-HT, dopamine and adrenergic receptors. In studies of 5-HT-stimulated phosphatidylinositol hydrolysis using SH-SY5Y cells stably expressing the cloned human 5-HT2C receptor, SB 242084 acted as an antagonist with a pKb of 9.3, which closely resembled its corresponding receptor binding affinity. SB 242084 potently inhibited m-chlorophenylpiperazine (mCPP, 7 mgkg i.p. 20 min pre-test)-induced hypolocomotion in rats, a model of in vivo central 5-HT2C receptor function, with an ID50 of 0.11 mg/kg i.p., and 2.0 mg/kg p.o. SB 242084 (0.1-1 mg/kg i.p.) exhibited an anxiolytic-like profile in the rat social interaction test, increasing time spent in social interaction, but having no effect on locomotion. SB 242084 (0.1-1 mg/kg i.p.) also markedly increased punished responding in a rat Geller-Seifter conflict test of anxiety, but had no consistent effect on unpunished responding. A large acute dose of SB 242084 (30 mg/kg p.o.) had no effect on seizure susceptibility in the rat maximal electroshock seizure threshold test. Also, while SB 242084 (2 and 6 mg/kg p.o. 1 hr pre-test) antagonized the hypophagic response to mCPP, neither acute nor subchronic administration of the drug, for 5 days at 2 or 6 mg/kg p.o. twice daily, affected food intake or weight gain. The results suggest that SB 242084 is the first reported selective potent and brain penetrant 5-HT2C receptor antagonist and has anxiolytic-like activity, but does not possess either proconvulsant or hyperphagic properties which are characteristic of mutant mice lacking the 5-HT2C receptor.[1] In acute organ injuries, mitochondria are often dysfunctional, and recent research has revealed that recovery of mitochondrial and renal functions is accelerated by induction of mitochondrial biogenesis (MB). We previously reported that the nonselective 5-HT2 receptor agonist DOI [1-(4-iodo-2,5-dimethoxyphenyl)propan-2-amine] induced MB in renal proximal tubular cells (RPTCs). The goal of this study was to determine the role of 5-HT2 receptors in the regulation of mitochondrial genes and oxidative metabolism in the kidney. The 5-HT2C receptor agonist CP-809,101 [2-[(3-chlorophenyl)methoxy]-6-(1-piperazinyl)pyrazine] and antagonist SB-242,084 [6-chloro-2,3-dihydro-5-methyl-N-[6-[(2-methyl-3-pyridinyl)oxy]-3-pyridinyl]-1H-indole-1-carboxyamide dihydrochloride] were used to examine the induction of renal mitochondrial genes and oxidative metabolism in RPTCs and in mouse kidneys in the presence and absence of the 5-HT2C receptor. Unexpectedly, both CP-809,101 and SB-242,084 increased RPTC respiration and peroxisome proliferator-activated receptor γ coactivator-1α (PGC-1α) mRNA expression in RPTCs at 1-10 nM. In addition, CP-809,101 and SB-242,084 increased mRNA expression of PGC-1α and the mitochondrial proteins NADH dehydrogenase subunit 1 and NADH dehydrogenase (ubiquinone) β subcomplex 8 in mice. These compounds increased mitochondrial genes in RPTCs in which the 5-HT2C receptor was downregulated with small interfering RNA and in the renal cortex of mice lacking the 5-HT2C receptor. By contrast, the ability of these compounds to increase PGC-1α mRNA and respiration was blocked in RPTCs treated with 5-HT2A receptor small interfering RNA or the 5-HT2A receptor antagonist eplivanserin. In addition, the 5-HT2A receptor agonist NBOH-2C-CN [4-[2-[[(2-hydroxyphenyl)methyl]amino]ethyl]-2,5-dimethoxybenzonitrile] increased RPTC respiration at 1-100 nM. These results suggest that agonism of the 5-HT2A receptor induces MB and that the classic 5-HT2C receptor agonist CP-809,101 and antagonist SB-242,084 increase mitochondrial genes and oxidative metabolism through the 5-HT2A receptor. To our knowledge, this is the first report that links 5-HT2A receptor agonism to mitochondrial function.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 44 mg/mL (~111.43 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1378 mL | 10.6888 mL | 21.3776 mL | |
| 5 mM | 0.4276 mL | 2.1378 mL | 4.2755 mL | |
| 10 mM | 0.2138 mL | 1.0689 mL | 2.1378 mL |