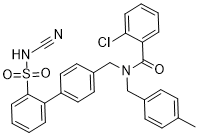
S0859 (S-0859) is an N-cyanosulphonamide compound acting as a novel, selective, high-affinity generic inhibitor of NBC [(Na(+)-coupled HCO(3)(-) transporters]. It is potentially important for probing the transporter's functional role in heart and other tissues. S0859 reversibly inhibited NBC-mediated pH(i) recovery (K (i)=1.7 microM, full inhibition at approximately 30 microM). In HEPES-buffered superfusates, NHE-mediated pH(i) recovery was unaffected by 30 microM S0859. With CO(2)/HCO(3) (-) buffer, pH(i) recovery from intracellular alkalosis (mediated by Cl(-)/HCO(3) (-) and Cl(-)/OH(-) exchange) was also unaffected. The time course of recovery of pH(i) and contraction was slowed by S0859, confirming that NBC is a significant controller of contractility during acidosis.
Physicochemical Properties
| Molecular Formula | C29H24CLN3O3S | |
| Molecular Weight | 530.04 | |
| Exact Mass | 529.122 | |
| CAS # | 1019331-10-2 | |
| Related CAS # |
|
|
| PubChem CID | 70675849 | |
| Appearance | White to yellow solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 748.5±70.0 °C at 760 mmHg | |
| Flash Point | 406.5±35.7 °C | |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C | |
| Index of Refraction | 1.639 | |
| LogP | 5.68 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 8 | |
| Heavy Atom Count | 37 | |
| Complexity | 897 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | ITDBPOSLOROLMT-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C29H24ClN3O3S/c1-21-10-12-22(13-11-21)18-33(29(34)26-7-2-4-8-27(26)30)19-23-14-16-24(17-15-23)25-6-3-5-9-28(25)37(35,36)32-20-31/h2-17,32H,18-19H2,1H3 | |
| Chemical Name | 2-chloro-N-[[4-[2-(cyanosulfamoyl)phenyl]phenyl]methyl]-N-[(4-methylphenyl)methyl]benzamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
S0859 targets sodium bicarbonate cotransporters (NBCs), specifically NBCn1 (SLC4A7) (IC50 = 0.4 μM in HEK293 cells overexpressing human NBCn1; IC50 = 0.6 μM in rat coronary endothelial cells) [3][4] S0859 shows no significant affinity for Na+/H+ exchanger 1 (NHE1) (IC50 > 30 μM) [2][4] |
||
| ln Vitro |
When NBC inhibitor S0859 was administered, caspase-3 activity and the quantity of apoptotic EC were both markedly increased. S0859 may be crucial for examining the functional role of the transporter in the heart and other tissues[1]. In HEK293 cells overexpressing human NBCn1, S0859 (0.1-10 μM) dose-dependently inhibited HCO3⁻-dependent pH recovery, with an IC50 of 0.4 μM; maximal inhibition (≥90%) was achieved at 5 μM [4] - In MCF7 breast cancer cells (wild-type and ErbB2-overexpressing), S0859 (1-30 μM) did not affect cell proliferation but significantly inhibited cell migration: at 10 μM, migration was reduced by ~40% (wild-type) and ~35% (ErbB2-overexpressing) compared to vehicle, as measured by transwell assay [2] - In human breast cancer cells (MDA-MB-231, BT-474), S0859 (5-20 μM) dose-dependently inhibited sodium bicarbonate cotransport activity, with IC50 values of 3.2 μM (MDA-MB-231) and 2.8 μM (BT-474); this was associated with reduced intracellular pH (pHi) alkalinization (from 7.45 ± 0.05 to 7.21 ± 0.04 at 10 μM) [1] - In rat coronary endothelial cells (CECs) subjected to hypoxia-reoxygenation (H/R) injury, S0859 (0.5-5 μM) dose-dependently reduced mitochondrial apoptosis: at 2 μM, caspase-3 activity was decreased by ~55%, and Bcl-2/Bax ratio was increased by 2.3-fold compared to H/R alone; intracellular ROS production was also reduced by ~40% [3] - S0859 (up to 30 μM) showed no significant inhibition of NHE1 activity in A549 cells, confirming selectivity for NBCs over NHE1 [4] |
||
| ln Vivo |
|
||
| Enzyme Assay |
NBCn1-mediated HCO3⁻ transport assay (pH-sensitive dye): HEK293 cells overexpressing human NBCn1 were seeded in 96-well plates and loaded with the pH-sensitive fluorescent dye BCECF-AM for 30 minutes at 37°C. Cells were acidified by NH4Cl prepulse, then exposed to HCO3⁻-containing buffer with serial dilutions of S0859. Fluorescence intensity (excitation 440/490 nm, emission 535 nm) was monitored to measure pH recovery rate, reflecting NBCn1 activity. IC50 values were calculated by nonlinear regression of dose-response curves [4] - Sodium bicarbonate cotransport assay (radioactive tracer): MDA-MB-231 cells were seeded in 24-well plates and preincubated with S0859 (0.1-30 μM) for 30 minutes. Cells were then incubated with 14C-labeled HCO3⁻ for 15 minutes at 37°C. Unincorporated tracer was removed by washing, and cell-associated radioactivity was measured using a scintillation counter. Inhibition rates were calculated relative to vehicle control [1] |
||
| Cell Assay |
Breast cancer cell migration assay: MCF7 cells (wild-type or ErbB2-overexpressing) were seeded in the upper chamber of transwell inserts (8 μm pores) at 5×10⁴ cells/well. S0859 (1-30 μM) was added to both upper and lower chambers, and cells were incubated for 24 hours. Migrated cells were fixed, stained with crystal violet, and counted under a microscope; migration rate was normalized to vehicle control [2] - Intracellular pH (pHi) measurement: BT-474 cells were loaded with BCECF-AM for 30 minutes, then treated with S0859 (5-20 μM) for 1 hour. pHi was calculated from fluorescence ratios (490/440 nm excitation) using a calibration curve generated with high-K+ buffers of known pH [1] - Hypoxia-reoxygenation (H/R) apoptosis assay: Rat coronary endothelial cells were seeded in 6-well plates and treated with S0859 (0.5-5 μM) for 1 hour before exposure to hypoxia (1% O2) for 4 hours, followed by reoxygenation (21% O2) for 24 hours. Apoptosis was assessed by Annexin V-FITC/PI staining and flow cytometry; caspase-3 activity was measured using a colorimetric assay kit [3] - Western blot assay: H/R-treated coronary endothelial cells were lysed in RIPA buffer, and proteins were separated by SDS-PAGE. Membranes were probed with antibodies against Bcl-2, Bax, cleaved caspase-3, and GAPDH (loading control). Chemiluminescent detection and densitometric analysis were used to quantify protein expression [3] - NHE1 selectivity assay: A549 cells were loaded with BCECF-AM and acidified by NH4Cl prepulse. Na+-dependent pH recovery (mediated by NHE1) was measured in the presence of S0859 (1-30 μM) or NHE1 inhibitor EIPA (positive control). Fluorescence intensity was monitored to assess NHE1 activity [4] |
||
| Animal Protocol |
|
||
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: S0859 (up to 30 μM) did not affect the viability of MCF7, MDA-MB-231, BT-474, or coronary endothelial cells after 72-hour treatment, as measured by MTT assay [1][2][3] |
||
| References |
[1]. Gram-scale solution-phase synthesis of selective sodium bicarbonate co-transport inhibitor S0859: in vitro efficacy studies in breast cancer cells. ChemMedChem. 2012 Oct;7(10):1808-14. [2]. The Na+/H+ exchanger NHE1, but not the Na+, HCO3(-) cotransporter NBCn1, regulates motility of MCF7 breast cancer cells expressing constitutively active ErbB2. Cancer Lett. 2012 Apr 28;317(2):172-83. [3]. SLC4A7 sodium bicarbonate co-transporter controls mitochondrial apoptosis in ischaemic coronary endothelial cells. Cardiovasc Res. 2011 Feb 1;89(2):392-400. [4]. S0859, an N-cyanosulphonamide inhibitor of sodium-bicarbonate cotransport in the heart. Br J Pharmacol. 2008 Mar;153(5):972-82. |
||
| Additional Infomation |
S0859 is a selective small-molecule inhibitor of sodium bicarbonate cotransporters (NBCs), belonging to the N-cyanosulphonamide chemical class [4] - The primary mechanism of S0859 involves inhibition of NBC-mediated HCO3⁻ influx, leading to reduction of intracellular pH (pHi) alkalinization and modulation of cellular processes dependent on pH homeostasis (e.g., cell migration, mitochondrial apoptosis) [1][3][4] - S0859 exhibits high selectivity for NBCn1 (SLC4A7) over other acid-base transporters (e.g., NHE1), making it a valuable tool compound for studying NBC function in cancer and cardiovascular biology [2][4] - Preclinical in vitro data suggest S0859 has potential therapeutic applications in breast cancer (via inhibiting cell migration) and ischemic cardiovascular diseases (via protecting endothelial cells from apoptosis) [1][2][3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.72 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.72 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.72 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8867 mL | 9.4333 mL | 18.8665 mL | |
| 5 mM | 0.3773 mL | 1.8867 mL | 3.7733 mL | |
| 10 mM | 0.1887 mL | 0.9433 mL | 1.8867 mL |