Physicochemical Properties
| Molecular Weight | 555.660769939423 |
| Exact Mass | 555.22 |
| CAS # | 1327258-57-0 |
| Related CAS # | Rucaparib monocamsylate;1859053-21-6 |
| PubChem CID | 131648247 |
| Appearance | Solid powder |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 39 |
| Complexity | 869 |
| Defined Atom Stereocenter Count | 2 |
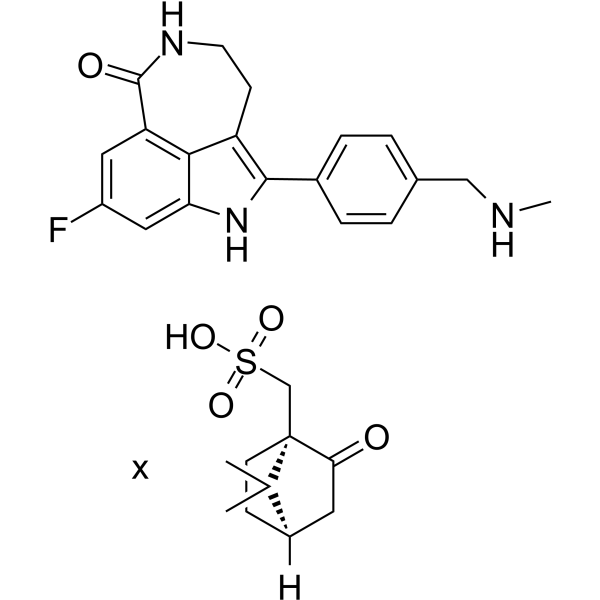
| SMILES | CC1([C@@H]2CC[C@@]1(C(=O)C2)CS(=O)(=O)O)C.CNCC1=CC=C(C=C1)C2=C3CCNC(=O)C4=C3C(=CC(=C4)F)N2 |
| InChi Key | INBJJAFXHQQSRW-MGYINCTJSA-N |
| InChi Code | InChI=1S/C19H18FN3O.C10H16O4S/c1-21-10-11-2-4-12(5-3-11)18-14-6-7-22-19(24)15-8-13(20)9-16(23-18)17(14)15;1-9(2)7-3-4-10(9,8(11)5-7)6-15(12,13)14/h2-5,8-9,21,23H,6-7,10H2,1H3,(H,22,24);7H,3-6H2,1-2H3,(H,12,13,14)/t;7-,10+/m.1/s1 |
| Chemical Name | [(1R,4R)-7,7-dimethyl-2-oxo-1-bicyclo[2.2.1]heptanyl]methanesulfonic acid;6-fluoro-2-[4-(methylaminomethyl)phenyl]-3,10-diazatricyclo[6.4.1.04,13]trideca-1,4,6,8(13)-tetraen-9-one |
| Synonyms | AG14699 (as phosphate salt); AG 14699; AG-14699; AG014447 (as free base); AG-014447; AG 014447; PF01367338; PF-01367338; PF 01367338; Rucaparib camsylate; Rubraca; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | AG14644 may have an N-desmethylated metabolite called rucaparib (AG014699) camsylate [1]. An LC50 of 5 μM was observed in Capan-1 (BRCA2 mutant) cells and only 100 μM in MX-1 (BRCA1 mutant) cells, indicating the cytotoxicity of rutaparib (0.1, 1, 10, 100 μM; 24 hours) camsylate[2]. Irrespective of SSB repair inhibition, rucaparib camsylate's radiosensitizing impact results from blocking downstream NF-κB activation. By focusing on NF-κB that is triggered by DNA damage, rucaparib camsylate can bypass the toxicity of traditional NF-κB inhibitors while maintaining other crucial inflammatory functions [5]. In permeabilized D283Med cells, rutapibacarib camsylate reduced PARP-1 activity by 97.1 % at a 1 μM dose [6]. |
| ln Vivo | Rucaparib (AG014699) camsylate and AG14584 substantially increase the toxicity of temozolomide. Rucaparib (1 mg/kg) camsylate greatly accelerates temozolomide-induced weight loss. Rucaparib (0.1 mg/kg) camsylate led in a 50% increase in temozolomide-induced tumor growth delay [1]. Rucaparib (10 mg/kg intraperitoneally or 50 or 150 mg/kg orally; 5 days a day, 5 days a week for 6 weeks) camsylate substantially reduced tumor growth, with 1 total tumor regression and 2 prolonged partial regressions occurring. 2]. Rucaparib (150 mg/kg; oral; once weekly for 6 weeks or 3 times weekly for 6 weeks) camsylate had the best anticancer effect, with three full regressions [2]. Rucaparib camsylate increased the anticancer efficacy of temozolomide and displayed complete and sustained tumor shrinkage in NB1691 and SHSY5Y xenografts [6]. |
| Animal Protocol |
Animal/Disease Models: 10-12 weeks old female CD-1 nude mice, Capan-1 cells [2] Doses: 10 mg/kg or 50 or 150 mg/kg Route of Administration: intraperitoneal (ip) injection of 10 mg/kg or 50 or 150 mg /kg Experimental Results:Dramatically inhibited tumor growth. |
| References |
[1]. Preclinical selection of a novel poly(ADP-ribose) polymerase inhibitor for clinical trial. Mol Cancer Ther, 2007, 6(3), 945-956. [2]. Tumour cell retention of rucaparib, sustained PARP inhibition and efficacy of weekly as well as daily schedules. Br J Cancer. 2014 Apr 15;110(8):1977-84. [3]. Rucaparib: A Review in Ovarian Cancer. Target Oncol. 2019 Apr;14(2):237-246. [4]. Hexose-6-phosphate dehydrogenase blockade reverses prostate cancer drug resistance in xenograft models by glucocorticoid inactivation. Sci Transl Med. 2021 May 26;13(595):eabe8226. [5]. NF-κB mediates radio-sensitization by the PARP-1 inhibitor, AG-014699. Oncogene, 2012, 31(2), 251-264. [6]. Inhibition of poly(ADP-ribose) polymerase-1 enhances temozolomide and topotecan activity against childhood neuroblastoma. Clin Cancer Res, 2009, 15(4), 1241-1249. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7997 mL | 8.9983 mL | 17.9966 mL | |
| 5 mM | 0.3599 mL | 1.7997 mL | 3.5993 mL | |
| 10 mM | 0.1800 mL | 0.8998 mL | 1.7997 mL |