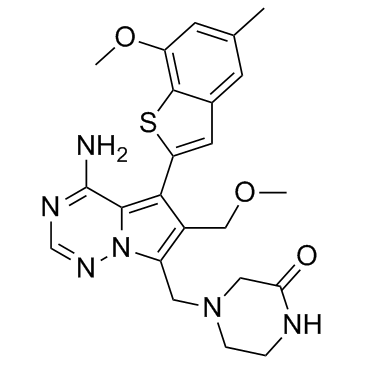
Rogaratinib (formerly also known as BAY1163877; BAY-1163877) is a novel, orally available, potent and selective inhibitor of aberrant fibroblast growth factor receptor (FGFR) with anticancer activity. In lung cancer (LC), aberrant activation or expression of the fibroblast growth factor receptor (FGFR) is frequently observed. The receptor tyrosine kinases FGFR-1, -2, and -3 are inhibited by rogaratinib. These kinases are upregulated in different types of tumor cells and may play a role in the differentiation, proliferation, angiogenesis, and survival of tumor cells.
Physicochemical Properties
| Molecular Formula | C23H26N6O3S |
| Molecular Weight | 466.555943012238 |
| Exact Mass | 466.178 |
| Elemental Analysis | C, 59.21; H, 5.62; N, 18.01; O, 10.29; S, 6.87 |
| CAS # | 1443530-05-9 |
| Related CAS # | 1443530-05-9 |
| PubChem CID | 71611869 |
| Appearance | White to light yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.731 |
| LogP | 0.98 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 33 |
| Complexity | 703 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S1C2C(=CC(C)=CC=2C=C1C1=C2C(N)=NC=NN2C(=C1COC)CN1CC(NCC1)=O)OC |
| InChi Key | HNLRRJSKGXOYNO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H26N6O3S/c1-13-6-14-8-18(33-22(14)17(7-13)32-3)20-15(11-31-2)16(9-28-5-4-25-19(30)10-28)29-21(20)23(24)26-12-27-29/h6-8,12H,4-5,9-11H2,1-3H3,(H,25,30)(H2,24,26,27) |
| Chemical Name | 4-[[4-amino-6-(methoxymethyl)-5-(7-methoxy-5-methyl-1-benzothiophen-2-yl)pyrrolo[2,1-f][1,2,4]triazin-7-yl]methyl]piperazin-2-one |
| Synonyms | Rogaratinib; BAY1163877; BAY-1163877; BAY 1163877 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | FGFR1; FGFR2; FGFR3; FGFR4 |
| ln Vitro | The FGFR1-amplified lung cancer (LC) cell lines H1581 and DMS114, which have GI50 values ranging from 36 to 244 nM, exhibit extreme sensitivity to rogaratinib (BAY1163877) out of the 24 cell lines. When H1581P cells are treated with rogaratinib, their ability to form colonies is significantly reduced, but H1581AR and BR cell colonies are not affected. Rogaratinib resistance is markedly increased in MTT assays by ectopic expression of Met. Metabolic overexpression triggers the activation of AKT and downstream extracellular signal-regulated kinase 1/2 (ERK1/2), which is irreversible upon treatment with rogaratinib[1]. |
| Cell Assay | On 96-well plates, cells are seeded at 37°C (3000 cells/well). Following a night of incubation, the cells receive a 72-hour Rogaratinib treatment. After that, each well is filled with MTT reagent (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide), which is then incubated for four hours at 37°C. Each well is filled with MTT solubilization solution/stop mix, combined, and the plates are then incubated at 37°C for the entire night. The data are shown graphically once the absorbance at 570 nm has been measured[1]. |
| References |
[1]. Preclinical profile of BAY 1163877-a selective pan-FGFR inhibitor in phase 1 clinical trial[J]. Cancer Res, 2014, 74(suppl 19): 1739a. [2]. Activation of the Met kinase confers acquired drug resistance in FGFR-targeted lung cancer therapy. Oncogenesis. 2016 Jul 18;5(7):e241. |
| Additional Infomation |
Rogaratinib is under investigation in clinical trial NCT03762122 (Rogaratinib in Patients With Advanced Pretreated Squamous-cell Non-small Cell Lung Cancer (SQCLC)). Rogaratinib is a pan inhibitor of human fibroblast growth factor receptors (FGFRs) with potential antiangiogenic and antineoplastic activities. Rogaratinib inhibits the activities of FGFRs, which may result in the inhibition of both tumor angiogenesis and tumor cell proliferation, and the induction of tumor cell death. FGFRs are a family of receptor tyrosine kinases, which may be upregulated in various tumor cell types and may be involved in tumor cell differentiation and proliferation, tumor angiogenesis, and tumor cell survival. Drug Indication Treatment of urothelial carcinoma |
Solubility Data
| Solubility (In Vitro) | DMSO: ~5 mg/mL (~10.7 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.56 mg/mL (1.20 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.6 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.56 mg/mL (1.20 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.6 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.56 mg/mL (1.20 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.6 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1433 mL | 10.7167 mL | 21.4335 mL | |
| 5 mM | 0.4287 mL | 2.1433 mL | 4.2867 mL | |
| 10 mM | 0.2143 mL | 1.0717 mL | 2.1433 mL |