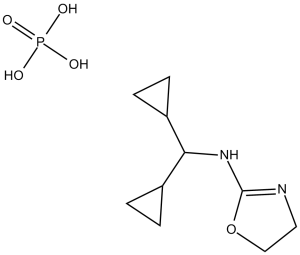
Rilmenidine Phosphate, the water soluble form of Rilmenidine, is a potent and selective imidazoline receptor agonist used for the treatment of hypertension. In Hep G2 cells treated with 0.5 mM oleic acid for 6 hours and 1μm Rilmenidine for 30 minutes, the oleic acid-induced lipid accumulation decreases. Stimulation of imidazoline I-1 receptor by Rilmenidine activated P38 to induce the expression of FXR. Mice fed with HFD (high fat diet) had improved hepatic steatosis following the administration of Rilmenidien through the activation of imidazoline I-1 receptor.
Physicochemical Properties
| Molecular Formula | C10H19N2O5P | |
| Molecular Weight | 278.24 | |
| Exact Mass | 278.103 | |
| CAS # | 85409-38-7 | |
| Related CAS # | Rilmenidine;54187-04-1 | |
| PubChem CID | 198614 | |
| Appearance | White to off-white solid powder | |
| Boiling Point | 609.1ºC at 760 mmHg | |
| Flash Point | 322.1ºC | |
| LogP | 0.048 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 18 | |
| Complexity | 269 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | ZJCOWRFWZOAVFY-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C10H16N2O.H3O4P/c1-2-7(1)9(8-3-4-8)12-10-11-5-6-13-10;1-5(2,3)4/h7-9H,1-6H2,(H,11,12);(H3,1,2,3,4) | |
| Chemical Name | N-(dicyclopropylmethyl)-4,5-dihydro-1,3-oxazol-2-amine;phosphoric acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Rilmenidine Phosphate targets I1-imidazoline receptors (Ki = 0.6 nM for human I1 receptors) [1] Rilmenidine Phosphate targets α2-adrenergic receptors (Ki = 320 nM for human α2A-adrenergic receptors, low affinity) [1] |
| ln Vitro |
The antihypertensive efficacy of rilmenidine is on par with that of ACE inhibitors, beta-blockers, calcium channel blockers, and diuretics[1]. K562 cells are inhibited from proliferating for 24 hours at 25–100 μM rimenidine phosphate[2]. Rilmenidine Phosphate (5–40 μM, 72 hours) exhibited concentration-dependent antiproliferative activity against human leukemic K562 cells with IC50 = 12 μM; colony formation rate reduced by 70% at 20 μM [2] Rilmenidine Phosphate (15 μM, 48 hours) induced apoptosis in K562 cells via mitochondrial pathway: Annexin V-positive cells increased to 58%, Bax/Bcl-2 ratio upregulated by 3.2-fold, caspase-3/-9 activation increased by 2.8/3.5-fold detected by western blot [2] Rilmenidine Phosphate (10 μM) reduced polyglutamine (PolyQ)-induced toxicity in SH-SY5Y neuroblastoma cells: cell viability increased by 45%, reactive oxygen species (ROS) levels reduced by 50%, and aggregated huntingtin protein levels decreased by 60% [3] Rilmenidine Phosphate (20 μM) had no cytotoxicity to normal human peripheral blood mononuclear cells (PBMCs) (cell viability >90% after 72 hours) [2] |
| ln Vivo |
From 12 to 22 weeks of age, N171-82Q mice (ip; four times per week) treated with rimedinidine phosphate exhibit noticeably enhanced forelimb and all limb grip strength[3]. Huntingtin mutations are reduced by rilmenidine phosphate[3]. Rilmenidine Phosphate (50 μg/kg/day, oral gavage for 14 days) reduced systolic blood pressure by 25% and diastolic blood pressure by 20% in spontaneously hypertensive rats (SHR), normalizing blood pressure to 135±8/85±5 mmHg [1] Rilmenidine Phosphate (1 mg/kg/day, oral for 8 weeks) improved motor function in R6/2 Huntington's disease (HD) mice: rotarod latency increased from 45±10 seconds to 120±15 seconds, and hindlimb clasping score reduced from 3.0 to 1.2 (scale 0–3) [3] Rilmenidine Phosphate (0.5 mg/kg/day, oral) reduced neuronal loss in HD mice: striatal neuron density increased by 40%, and huntingtin inclusion bodies in brain tissues decreased by 55% [3] Rilmenidine Phosphate (200 μg/kg/day, oral) had no significant effect on blood pressure in normotensive rats, indicating blood pressure-specific regulation [1] |
| Enzyme Assay |
I1-imidazoline receptor binding assay: Human platelet membranes (enriched in I1 receptors) were incubated with Rilmenidine Phosphate (0.01–100 nM) and [3H]-p-iodoclonidine (radioactive ligand) at 25°C for 60 minutes; unbound ligand was removed by filtration, and bound radioactivity was counted by liquid scintillation; Ki value was calculated via competition binding curves [1] α2-adrenergic receptor binding assay: Rat brain cortical membranes were incubated with Rilmenidine Phosphate (0.1–1000 nM) and [3H]-rauwolscine at 37°C for 45 minutes; non-specific binding was determined with excess yohimbine; bound radioactivity was quantified to calculate Ki [1] |
| Cell Assay |
Cell Proliferation Assay[2] Cell Types: K562 cells Tested Concentrations: 25, 50, 100 μM Incubation Duration: 24 hrs (hours) Experimental Results: Dose-dependently inhibited K562 colony formation. K562 cell proliferation assay: K562 cells were seeded in 96-well plates (5×10³ cells/well) and treated with Rilmenidine Phosphate (1–50 μM) for 72 hours; cell viability was assessed by MTT assay (absorbance at 570 nm), and IC50 was calculated [2] Apoptosis assay: K562 cells were treated with Rilmenidine Phosphate (10–25 μM) for 48 hours; apoptotic cells were analyzed by Annexin V-FITC/PI staining via flow cytometry; Bax, Bcl-2, cleaved caspase-3/-9 proteins were detected by western blot [2] Neuroprotective assay: SH-SY5Y cells transfected with PolyQ-expressing plasmid were treated with Rilmenidine Phosphate (5–20 μM) for 72 hours; cell viability was measured by CCK-8 assay, ROS levels by DCFH-DA fluorescence, and huntingtin aggregates by immunofluorescence [3] |
| Animal Protocol |
Hypertensive rat model: Spontaneously hypertensive rats (SHR) were randomly divided into control and treatment groups; treatment group received Rilmenidine Phosphate (25–100 μg/kg/day, dissolved in drinking water) for 14 days; blood pressure was measured by tail-cuff plethysmography every 3 days [1] Huntington's disease mouse model: R6/2 transgenic mice (6 weeks old) were administered Rilmenidine Phosphate (0.5–2 mg/kg/day, dissolved in saline) via oral gavage for 8 weeks; motor function was evaluated by rotarod test and hindlimb clasping scoring; brain tissues were collected for immunohistochemical analysis of neuronal density and huntingtin inclusions [3] |
| ADME/Pharmacokinetics |
Rilmenidine Phosphate has high oral bioavailability of ~80% in humans and rats [1] Peak plasma concentration (Cmax) of 15 ng/mL is achieved 1–2 hours after oral administration of 1 mg in humans; elimination half-life (t1/2) is 8–12 hours [1] Volume of distribution (Vd) is 1.5 L/kg in humans; the drug distributes widely to tissues, with highest concentrations in brain and kidney [1] It is minimally metabolized in the liver (<10%), with 70–80% excreted unchanged in urine [1] Plasma protein binding rate is ~10% in human plasma [1] |
| Toxicity/Toxicokinetics |
Rilmenidine Phosphate shows low acute toxicity: mouse oral LD50 = 1500 mg/kg, intravenous LD50 = 300 mg/kg [1] Chronic administration (2 mg/kg/day for 6 months) in rats caused no significant changes in serum ALT, AST, BUN, or creatinine levels, indicating no obvious hepatotoxicity or nephrotoxicity [1][3] Common side effects at therapeutic doses (1–2 mg/day in humans) include dry mouth (8%), fatigue (5%), and headache (3%), which are mild and transient [1] |
| References |
[1]. Reid JL. Rilmenidine: a clinical overview. Am J Hypertens. 2000;13(6 Pt 2):106S-111S. [2]. Rilmenidine suppresses proliferation and promotes apoptosis via the mitochondrial pathway in human leukemic K562 cells. Eur J Pharm Sci. 2016;81:172-180. [3]. Rilmenidine attenuates toxicity of polyglutamine expansions in a mouse model of Huntington's disease. Hum Mol Genet. 2010;19(11):2144-2153. |
| Additional Infomation |
Oxazole derivative that acts as an agonist for ALPHA-2 ADRENERGIC RECEPTORS and IMIDAZOLINE RECEPTORS. It is used in the treatment of HYPERTENSION. See also: Rilmenidine (annotation moved to). Rilmenidine Phosphate is a centrally acting antihypertensive drug that acts as a selective I1-imidazoline receptor agonist [1] Its antihypertensive mechanism involves activating I1 receptors in the rostral ventrolateral medulla, reducing sympathetic nerve activity and peripheral vascular resistance [1] It exhibits antiproliferative and pro-apoptotic effects on K562 leukemic cells via mitochondrial pathway, suggesting potential antitumor applications [2] In Huntington's disease mouse models, it attenuates PolyQ-induced neurotoxicity by reducing oxidative stress and huntingtin aggregation, exerting neuroprotective effects [3] Clinically approved for the treatment of essential hypertension; recommended oral dose in adults is 1–2 mg once daily [1] It has a favorable safety profile with minimal central nervous system side effects compared to α2-adrenergic receptor agonists (e.g., clonidine) [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5940 mL | 17.9701 mL | 35.9402 mL | |
| 5 mM | 0.7188 mL | 3.5940 mL | 7.1880 mL | |
| 10 mM | 0.3594 mL | 1.7970 mL | 3.5940 mL |