Physicochemical Properties
| Molecular Formula | C22H32O2 |
| Molecular Weight | 328.4883 |
| Exact Mass | 328.24 |
| CAS # | 127-47-9 |
| Related CAS # | Retinyl acetate-d4;118139-40-5 |
| PubChem CID | 638034 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 440.5±14.0 °C at 760 mmHg |
| Melting Point | 57-58 °C |
| Flash Point | 124.8±18.5 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.532 |
| LogP | 7.39 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 24 |
| Complexity | 596 |
| Defined Atom Stereocenter Count | 0 |
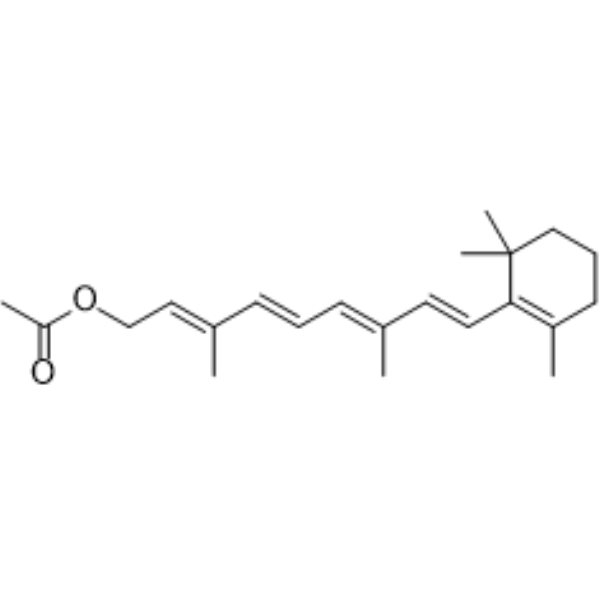
| SMILES | CC1=C(C(CCC1)(C)C)/C=C/C(=C/C=C/C(=C/COC(=O)C)/C)/C |
| InChi Key | QGNJRVVDBSJHIZ-QHLGVNSISA-N |
| InChi Code | InChI=1S/C22H32O2/c1-17(9-7-10-18(2)14-16-24-20(4)23)12-13-21-19(3)11-8-15-22(21,5)6/h7,9-10,12-14H,8,11,15-16H2,1-6H3/b10-7+,13-12+,17-9+,18-14+ |
| Chemical Name | [(2E,4E,6E,8E)-3,7-dimethyl-9-(2,6,6-trimethylcyclohexen-1-yl)nona-2,4,6,8-tetraenyl] acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Retinyl acetate (10 μM) suppresses cell growth (such as melanoma cell lines B 16 and S91, the inhibition rates are 48% and 79% respectively) [2]. The study evaluated the growth inhibitory effects of retinyl acetate (at a concentration of 10⁻⁵ M) on 31 untransformed, transformed, and tumor cell lines in vitro. Cells were categorized based on their sensitivity: (a) Cells not affected (<10% inhibition) or only slightly inhibited (<25%) by retinyl acetate, including untransformed BHK and CHO-K1-pro cells, virally transformed PyBHK, human fibrosarcoma HT1080, and others like untransformed Balb/3T3, SV3T3, MSV3T3, and chemically transformed BP3T3. (b) Cells exhibiting growth inhibition (25-50%), such as neuroblastoma C1300 (40% inhibition), mouse mammary adenocarcinomas M12 and DD3, human breast carcinosarcoma H,0578, and murine lymphomas S49 and EL4. (c) Cells that were moderately growth inhibited (50-75%), including mouse sarcoma S180, mastocytoma P815 (21% inhibition), rat mammary adenocarcinoma R3230AC, and others. (d) Cells extremely sensitive to retinyl acetate (growth inhibited >75%), such as murine melanoma S91 (79% inhibition), rat mammary adenocarcinoma 13762NF, murine lymphosarcoma RAW117, and others. In most cases, retinoic acid was more potent than retinyl acetate, but for some cell lines (e.g., C1300 neuroblastoma, S91 melanoma, Mm5mT, and 13762NF mammary adenocarcinomas), retinyl acetate was nearly as active. [2] |
| ln Vivo |
The progression of Sprague-Dawley tumors produced by DMBA is inhibited by retinyl acetate (250 mg/kg in diet) [1]. Administration of retinyl acetate in the diet (250 mg per kg of diet) significantly inhibited mammary carcinogenesis induced by N-methyl-N-nitrosourea (NMU) in female Sprague-Dawley rats. It reduced both the incidence and the total number of mammary cancers (including benign tumors) across different NMU dose groups. Additionally, retinyl acetate markedly prolonged the latency period for the appearance of palpable mammary tumors. For instance, in rats treated with a high dose of NMU, the time to first palpable cancer was approximately doubled compared to the placebo control group. No mammary cancers developed in rats receiving the low dose of NMU along with retinyl acetate by the end of the experiment (175 days). [1] |
| Cell Assay |
Cell growth inhibition was assessed using a standardized assay. Briefly, cells (0.5-1.0 x 10⁵) were seeded in growth medium in culture dishes. The test groups received fresh growth medium containing retinyl acetate at a final concentration of 10⁻⁵ M, dissolved in 0.1% ethanol. Control groups received medium with 0.1% ethanol only. All procedures involving retinoids were performed under subdued light, and culture dishes were wrapped in aluminum foil. Cells were incubated at 37°C in a humidified atmosphere of 13% CO₂. Monolayer or multilayer cultures were refed with fresh medium every third day. Suspension cultures were refed by adding fresh medium to the existing culture. Cells were grown until control cultures were nearly confluent (typically 5-6 days) or, for suspension cells, until control cells underwent five to six replication cycles. At the endpoint, adherent cells were detached using EDTA in calcium- and magnesium-free PBS and counted using an electronic particle counter or hemacytometer. Cell viability was assessed by trypan blue exclusion. The percent inhibition of proliferation was calculated relative to control cultures. Experiments were performed at least twice in duplicate. [2] |
| Animal Protocol |
Female Sprague-Dawley rats, 42 days old, were used. At 50 days of age (average weight 150g), rats received an intravenous injection of NMU dissolved in physiological saline (adjusted to pH 5.0) at doses of 5.0, 2.5, or 1.25 mg per 100g body weight. A second injection of NMU or saline (for controls) was administered 7 days later. Three days after the second injection, all rats were placed on experimental diets for the remainder of the study. Retinyl acetate was blended into the standard stock diet in the form of stable gelatinized beadlets at a concentration of 250 mg per kg diet. Control groups received placebo beadlets without the retinoid. Rats were palpated twice weekly for mammary tumor detection, weighed twice weekly, and observed daily for signs of toxicity. All animals were sacrificed 175 days after the initial NMU injection, and mammary tumors were excised for histological examination. [1] |
| ADME/Pharmacokinetics |
The article mentions that the half-life of the carcinogen NMU at body pH is only a few hours. However, no ADME or pharmacokinetic parameters (e.g., absorption, distribution, metabolism, excretion, half-life, oral bioavailability) for retinyl acetate itself are described in the provided text. [1] |
| Toxicity/Toxicokinetics |
In this study, no signs of 'hypervitaminosis A' (retinoid toxicity) were observed in rats fed retinyl acetate. Body weights and liver histology of rats receiving retinyl acetate were essentially identical to those of the placebo-fed control rats. Vaginal smears from rats fed retinyl acetate revealed normal estrous cycles, suggesting no significant alteration in ovarian steroid metabolism. The inhibition of carcinogenesis was not attributed to a general toxic effect of the compound. [1] In the cell culture experiments, the observed growth inhibition by retinyl acetate was not attributed to direct cytotoxicity. Daily observation of cultures exposed to 10⁻⁵ M retinyl acetate did not reveal increased cell debris or detached cells. The viability of cells treated with retinoids for the duration of the experiments (5-8 days) was consistently over 90% and similar to that of untreated control cells. [2] |
| References |
[1]. Retinyl acetate inhibits mammary carcinogenesis induced by N-methyl-N-nitrosourea. Nature. 1977 Jun 16;267(5612):620-1. [2]. Lotan R, Nicolson GL. Inhibitory effects of retinoic acid or retinyl acetate on the growth of untransformed, transformed, and tumor cells in vitro. J Natl Cancer Inst. 1977 Dec;59(6):1717-22. |
| Additional Infomation |
Retinyl acetate is an acetate ester. It is functionally related to an all-trans-retinol. Retinyl Acetate is a naturally-occurring fatty acid ester form of retinol (vitamin A) with potential antineoplastic and chemopreventive activities. Retinyl acetate binds to and activates retinoid receptors, inducing cell differentiation and decreasing cell proliferation. This agent also inhibits carcinogen-induced neoplastic transformation in some cancer cell types and exhibits immunomodulatory properties. (NCI04) Retinyl acetate is a retinoid (vitamin A analogue). The study demonstrates its efficacy in preventing experimental mammary cancer in an NMU-induced rat model, which is considered to more closely resemble human breast cancer (in terms of invasiveness, hormone dependence, and metastasis) compared to other models like DMBA-induced cancer. The mechanism by which retinoids inhibit mammary carcinogenesis is not fully understood but is suggested to possibly involve the maintenance of normal epithelial cell differentiation. The authors note that while toxicity was not observed in this study, the potential toxicity of retinyl esters makes the development of new synthetic retinoids with greater activity and lesser toxicity important. [1] Retinyl acetate is a vitamin A derivative (retinoid). The study demonstrates its ability to directly inhibit the proliferation of a broad spectrum of transformed and tumor cell lines in vitro, suggesting that at least part of the antitumor activity observed for retinoids in vivo may be due to direct effects on tumor cell growth rather than solely indirect mechanisms like immune modulation. The mechanism of action is not fully elucidated but may involve interaction with specific intracellular binding proteins, potentially affecting DNA synthesis. The variability in sensitivity among different cell lines indicates that screening tumor cells for susceptibility to retinoids in vitro might help predict potential responsiveness to retinoid-based therapies. [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~50 mg/mL (~152.21 mM) H2O : ~0.67 mg/mL (~2.04 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.61 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: 2 mg/mL (6.09 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0442 mL | 15.2212 mL | 30.4423 mL | |
| 5 mM | 0.6088 mL | 3.0442 mL | 6.0885 mL | |
| 10 mM | 0.3044 mL | 1.5221 mL | 3.0442 mL |