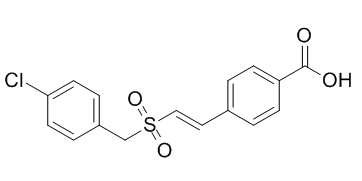
Recilisib (ON-01210; ON 01210; ON01210; EX-RAD) is a novel and potent radioprotectant that Onconova Therapeutics is testing in phase I clinical trials to treat acute radiation syndrome. Through the activation of the AKT pathway, it lessens radiation damage.
Physicochemical Properties
| Molecular Formula | C₁₆H₁₃CLO₄S |
| Molecular Weight | 336.79 |
| Exact Mass | 336.022 |
| Elemental Analysis | C, 57.06; H, 3.89; Cl, 10.53; O, 19.00; S, 9.52 |
| CAS # | 334969-03-8 |
| Related CAS # | Recilisib sodium;922139-31-9 |
| PubChem CID | 9884220 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 607.8±55.0 °C at 760 mmHg |
| Flash Point | 321.4±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.639 |
| LogP | 3.84 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 22 |
| Complexity | 493 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(O)C1=CC=C(/C=C/S(=O)(CC2=CC=C(Cl)C=C2)=O)C=C1 |
| InChi Key | KBEKQQJUNVQLDZ-MDZDMXLPSA-N |
| InChi Code | InChI=1S/C16H13ClO4S/c17-15-7-3-13(4-8-15)11-22(20,21)10-9-12-1-5-14(6-2-12)16(18)19/h1-10H,11H2,(H,18,19)/b10-9+ |
| Chemical Name | Benzoic acid, 4-((1E)-2-(((4-chlorophenyl)methyl)sulfonyl)ethenyl)- |
| Synonyms | ON-01210; ON01210; ON 01210; Recilisib Free Base |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PI3K |
| ln Vitro | Recilisib sodium (up to 50 μM) exhibits a consistent pattern of cells throughout the cell cycle, with a slight decrease in the proportion of cells in S-phase at this concentration. Recilisib Sodium (100 μM) does not cause cell death when administered continuously. The ability of human bone marrow cells to form colonies is not inhibited by recilisib sodium treatment. At each of the three IR doses, recilisib sodium offers human bone marrow cells dose-dependent protection. In HFL cells, recilisib sodium stimulates AKT and GSKα/β phosphorylation. In HFL-1 cells and murine bone marrow cells, recilisib sodium boosts PI3K activity in response to radiation exposure. The MAPK signaling pathway is altered by recilisib sodium treatment in conjunction with radiation[1]. |
| ln Vivo | In mice, recilisib sodium (500 mg/kg) significantly accelerates the recovery and differentiation of early myeloid progenitor cells from the bone marrow. When combined with radiation, Recilisib Sodium lowers the number of CFUs in mice, but mice treated with Recilisib Sodium consistently retain the ability to form differentiated colonies. The progenitor cell population in Recilisib Sodium-treated mice is never completely depleted by radiation exposure[1]. |
| Enzyme Assay | In PI3-kinase assays, freshly isolated murine bone marrow cells or exponentially growing HFL-1 cells are exposed to increasing concentrations of Recilisib sodium for two hours before being exposed to 10 Gy IR. After being lysed in HEPES pH 7.5 lysis buffer, these cells are then put back in the incubator for two to twenty-four hours. An anti-PI3 Kinas polyclonal antibody is used to immunoprecipitate PI-3K for two hours at room temperature. Immunoprecipitates are incubated with Protein A/G PLUS-Agarose for 8–16 hours at 4°C, and the resulting immunoprecipitates are then washed with the kinase buffer (20 mM Tris pH 7.5, 1 mM EGTA, 10 mM MgCl2, 2 mM DTT, 0.01% NP-40) once and twice, respectively. In the kinase buffer (60 µL per sample), L--Phosphatidylinositol (12.5 mM) and ATP (10 µM) are added. The mixture is then incubated at 30°C for 30 minutes. 100 µL of 1N HCl is added to stop the reaction, and 200 µL of CHCl3/CH3OH (1:1) is added to extract the product. The lower organic phases containing phospholipids are dried at 27°C for two hours after being vortexed and centrifuged with the extracted samples. After being dried, the samples are reconstituted in 10 µL of PI-4-P standard (0.5 mL CHCl3, 0.5 mL CH3OH, 2.5 µL HCl), and then spots are applied to TLC plates (VWR). Thin layer chromatography is performed on the spotted plate using CHCl3, CH3OH, and NH4OH (40:40:15). The TLC plate is dried before autoradiography is performed. |
| Cell Assay | For cytotoxicity tests, cells (1.0×105 cells/mL HPGM) are exposed to different concentrations of the drug Recilisib Sodium or a control substance for two or twenty-four hours. The cells are cleaned and plate into gridded dishes for methocult. Using an Olympus IMT-2 microscope, 14 days after plating, the total number of colony forming units (CFUs) was counted. |
| References |
[1]. ON01210.Na (Ex-RAD) mitigates radiation damage through activation of the AKT pathway. PLoS One. 2013;8(3):e58355. |
Solubility Data
| Solubility (In Vitro) | DMSO: 35.7~67 mg/mL (106 mM~198.9 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9692 mL | 14.8460 mL | 29.6921 mL | |
| 5 mM | 0.5938 mL | 2.9692 mL | 5.9384 mL | |
| 10 mM | 0.2969 mL | 1.4846 mL | 2.9692 mL |