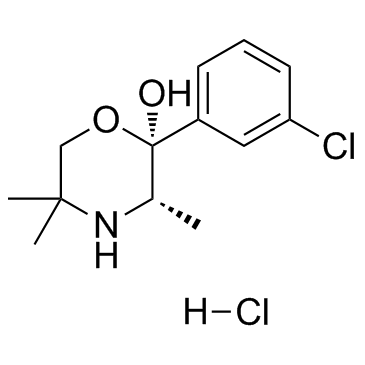
Radafaxine HCl (also known as GW-353162A) is a novel and potent DAT (dopamine transporter) and NET(norepinephrine transporter) transporters inhibitor, it is also an nAChR family modulator.
Physicochemical Properties
| Molecular Formula | C13H18NO2CL.HCL |
| Molecular Weight | 292.20146 |
| Exact Mass | 291.079 |
| CAS # | 106083-71-0 |
| Related CAS # | Radafaxine;192374-14-4 |
| PubChem CID | 9838996 |
| Appearance | White to off-white solid powder |
| Density | 1.144g/cm3 |
| Boiling Point | 386.6ºC at 760mmHg |
| Flash Point | 187.6ºC |
| Vapour Pressure | 0mmHg at 25°C |
| Index of Refraction | 1.523 |
| LogP | 3.402 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 18 |
| Complexity | 285 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | C[C@H]1[C@@](OCC(N1)(C)C)(C2=CC(=CC=C2)Cl)O.Cl |
| InChi Key | ORXTVTDGPVINDN-BTJVGWIPSA-N |
| InChi Code | InChI=1S/C13H18ClNO2.ClH/c1-9-13(16,17-8-12(2,3)15-9)10-5-4-6-11(14)7-10;/h4-7,9,15-16H,8H2,1-3H3;1H/t9-,13+;/m0./s1 |
| Chemical Name | (2S,3S)-2-(3-chlorophenyl)-3,5,5-trimethylmorpholin-2-ol;hydrochloride |
| Synonyms | Radafaxine hydrochloride; Radafaxine HCl; GW-353162A; GW353162A; GW 353162A; GW-353162-A; GW353162-A; GW 353162-A; GW-353162 A; GW353162 A; GW 353162 A; gw-353162A; gw353162 A; gw 353162-A; BW-306U; BW 306U; BW306U; BW-306 U; BW 306 U; BW306 U; BW-306-U; BW 306-U; BW306-U |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Dopamine transporter (DAT) and norepinephrine transporter (NET) – Radafaxine is the (+) isomer of hydroxybupropion and blocks both DAT and NET. [2] |
| ln Vitro |
It is doubtful that radafaxine hydrochloride (GW-353162A) will have potentiating effects because it blocks DAT slowly and with comparatively little potency [3]. In radioligand binding assays, S,S-hydroxybupropion (radafaxine) showed measurable affinity for the dopamine transporter (DAT) with a Ki of 1295 ± 280 nM and for the norepinephrine transporter (NET) with a Ki of 3850 ± 570 nM. It did not show significant affinity for the serotonin transporter (SERT) (Ki > 10,000 nM) or for various nicotinic acetylcholine receptor subtypes (α2β2, α2β4, α3β2, α3β4, α4β2, α4β4) at a concentration of 10,000 nM. [3] In a functional ⁸⁶Rb⁺ efflux assay using cells expressing the α3β4 nicotinic receptor subtype, S,S-hydroxybupropion acted as an antagonist, inhibiting nicotine-stimulated efflux with an IC₅₀ of 18 µM. It showed no agonist activity at a concentration of 100 µM. [3] |
| ln Vivo |
In healthy human subjects, a single oral dose of 40 mg Radafaxine induced a slow and long-lasting blockade of DAT in the striatum. Peak DAT occupancy was 20–22% at 4 hours post-dose, with occupancy remaining at 12–16% at 24 hours. No behavioral or cardiovascular effects were observed. [2] In rats trained to discriminate (-)nicotine from saline, S,S-hydroxybupropion (radafaxine) partially substituted for the nicotine stimulus. Doses of 11 and 11.5 mg/kg produced 55% and 66% nicotine-appropriate responding, respectively. [3] In rats trained to discriminate (+)amphetamine from saline, S,S-hydroxybupropion substituted completely for the amphetamine stimulus in a dose-related manner, with an ED₅₀ of 4.4 mg/kg (95% CL: 1.9 – 10.4 mg/kg), which was nearly equipotent to bupropion (ED₅₀ = 5.4 mg/kg). [3] |
| Enzyme Assay |
Radioligand binding assays for nicotinic acetylcholine receptors and monoamine transporters were performed by the NIMH Psychoactive Drug Screening Program (PDSP). Cell lines stably expressing specific rat nicotinic receptor subunit combinations (α2β2, α2β4, α3β2, α3β4, α4β2, α4β4) were established in HEK 293 cells. Binding of [³H]epibatidine (100 pM) to these receptors was measured. Initial screening was performed with a single concentration of test compound (10 µM), and compounds causing >25% inhibition were subjected to more detailed assays. For transporter binding, affinity at dopamine (DAT), norepinephrine (NET), and serotonin (SERT) transporters was assessed. [3] |
| Cell Assay |
Functional activity at the α3β4 nicotinic acetylcholine receptor was assessed using a ⁸⁶Rb⁺ efflux assay. KXα3β4R2 cells were loaded with ⁸⁶RbCl (2 µCi/mL). Agonist activity was evaluated by measuring efflux in the absence of other agents. Antagonist activity was assessed by measuring inhibition of efflux stimulated by 100 µM nicotine. For detailed analysis, concentration-response curves (8 concentrations) were generated in the presence (for inhibition/IC₅₀) or absence (for stimulation/EC₅₀) of nicotine. All assays were performed with four replicates. [3] |
| Animal Protocol |
Drug discrimination studies were conducted using male Sprague-Dawley rats. Animals were trained to discriminate either subcutaneous injections of (-)nicotine (0.6 mg/kg as free base) or intraperitoneal injections of (+)amphetamine sulfate (1.0 mg/kg) from saline vehicle (0.9% sterile saline) under a variable interval 15-s schedule of reward (sweetened milk) in a two-lever operant chamber. Training sessions lasted 15 minutes. Animals were used in stimulus generalization tests only after consistently making >80% of responses on the drug-appropriate lever after training drug and <20% after saline. For generalization tests, doses of test compounds (including S,S-hydroxybupropion) were administered subcutaneously (for nicotine-trained rats) or intraperitoneally (for amphetamine-trained rats) 15 minutes before a 2.5-minute non-reinforced (extinction) session. Doses were administered in random order. Drug solutions were prepared fresh daily. [3] |
| ADME/Pharmacokinetics |
Following a single oral dose of 40 mg Radafaxine in humans, peak plasma concentration occurred at approximately 4–6 hours, with an estimated value of 89.7 ng/mL. DAT occupancy in striatum correlated with plasma concentration (Spearman r = 0.51, p = 0.003). The drug showed slow brain pharmacokinetics with long-lasting DAT blockade. [2] |
| Toxicity/Toxicokinetics |
In the behavioral tests, the highest tested dose of S,S-hydroxybupropion (11.5 mg/kg) depressed response rates in rats by about 50%. [3] |
| References |
[1]. Stereoselective analysis of hydroxybupropion and application to drug interaction studies. Chirality. 2007 Mar;19(3):163-70. [2]. The slow and long-lasting blockade of dopamine transporters in human brain induced by the new antidepressant drug radafaxine predict poor reinforcing effects. Biol Psychiatry. 2005 Mar 15;57(6):640-6. [3]. Behavioral and biochemical investigations of bupropion metabolites. Eur J Pharmacol. 2003 Aug 1;474(1):85-93. |
| Additional Infomation |
Radafaxine is a new antidepressant drug that blocks both dopamine and norepinephrine transporters. It is the (+) isomer of hydroxybupropion. This study used PET with [¹¹C]cocaine to measure DAT occupancy in humans and predicted low abuse liability due to its slow kinetics and low (<50%) DAT blockade. Preclinical studies suggest no self-administration behavior. [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~25 mg/mL (~85.56 mM) H2O : ~25 mg/mL (~85.56 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.56 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.56 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.56 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 8.33 mg/mL (28.51 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4223 mL | 17.1116 mL | 34.2231 mL | |
| 5 mM | 0.6845 mL | 3.4223 mL | 6.8446 mL | |
| 10 mM | 0.3422 mL | 1.7112 mL | 3.4223 mL |