Physicochemical Properties
| Molecular Formula | C11H10FNO3S2 |
| Molecular Weight | 287.3304 |
| Exact Mass | 287.008 |
| CAS # | 190967-35-2 |
| PubChem CID | 9882374 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 403.4±55.0 °C at 760 mmHg |
| Flash Point | 197.8±31.5 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.624 |
| LogP | 4.22 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 18 |
| Complexity | 363 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | BEIZIEZPGSIQGR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H10FNO3S2/c1-18(14,15)13-10-6-7-11(17-10)16-9-4-2-8(12)3-5-9/h2-7,13H,1H3 |
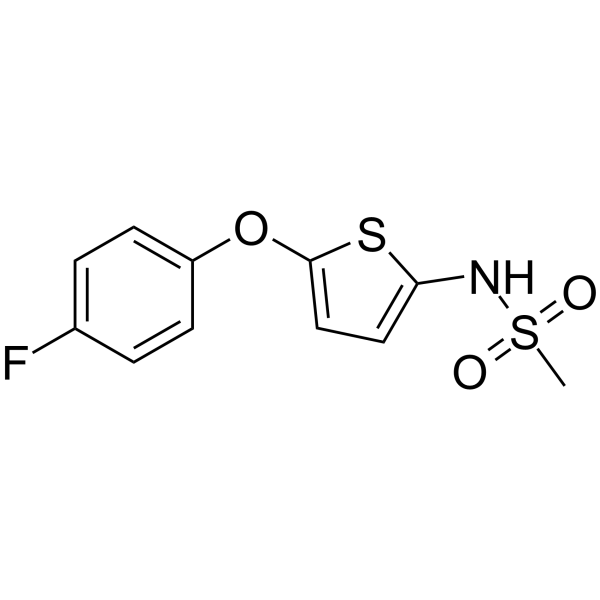
| Chemical Name | N-[5-(4-fluorophenoxy)thiophen-2-yl]methanesulfonamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
1. Cyclooxygenase-2 (COX-2, selective inhibitor, IC50 = 0.12 μM for human recombinant COX-2; >100-fold selectivity over COX-1, COX-1 IC50 > 15 μM) [1] 2. 5-Lipoxygenase (5-LOX, inhibitor, IC50 = 0.8 μM for human recombinant 5-LOX) [1] |
| ln Vitro |
RWJ 63556 retains its eicosanoid characteristics in vitro and is a strong inhibitor of leukocyte entry within 24 hours [1]. 1. Enzymatic inhibitory activity: RWJ 63556 exhibited potent and selective inhibition of COX-2 and 5-LOX in recombinant enzyme assays. It inhibited human COX-2 with an IC50 of 0.12 μM, while showing minimal activity against COX-1 (IC50 > 15 μM), resulting in a COX-2/COX-1 selectivity ratio of >125. For 5-LOX, the compound had an IC50 of 0.8 μM, with no significant inhibition of other lipoxygenase isoforms (12-LOX/15-LOX IC50 > 20 μM) at concentrations up to 20 μM [1] 2. Inflammatory mediator suppression in cell models: In canine peripheral blood mononuclear cells (PBMCs) stimulated with lipopolysaccharide (LPS), RWJ 63556 (0.1-5 μM) dose-dependently suppressed the production of prostaglandin E₂ (PGE₂, a COX-2-derived mediator) and leukotriene B₄ (LTB₄, a 5-LOX-derived mediator). At 1 μM, PGE₂ levels were reduced by 78% and LTB₄ levels by 65% relative to LPS-treated controls, with no significant effect on cell viability (viability > 92% at 5 μM) [1] |
| ln Vivo |
The development of SP-induced edema is considerably inhibited by intravenous administration of RWJ 63556 (1 mg/kg) [2]. 1. Anti-inflammatory activity in canine carrageenan-induced paw edema model: In beagle dogs, oral administration of RWJ 63556 (1 mg/kg, 3 mg/kg, 10 mg/kg) 1 h prior to carrageenan injection into the hind paw significantly reduced paw edema in a dose-dependent manner. At 10 mg/kg, the maximal edema volume at 4 h post-carrageenan was reduced by 62% compared with vehicle control; the 3 mg/kg and 1 mg/kg doses achieved 45% and 22% edema reduction respectively. The compound also suppressed local inflammatory mediator levels: paw tissue PGE₂ was reduced by 71% and LTB₄ by 58% at 10 mg/kg, with plasma PGE₂/LTB₄ levels also decreased by 52% and 48% respectively [1] 2. Anti-hyperalgesic effect in canine inflammation model: RWJ 63556 (10 mg/kg, oral) reduced carrageenan-induced mechanical hyperalgesia in dogs (pain threshold increased from 2.1 N to 5.8 N at 4 h post-carrageenan, vs 2.3 N in vehicle group), confirming its anti-inflammatory and analgesic efficacy in vivo [1] |
| Enzyme Assay |
1. Recombinant COX-2/COX-1 activity assay: Purified human recombinant COX-2 and COX-1 enzymes were incubated with serial dilutions of RWJ 63556 (0.01-20 μM) in a buffer system containing arachidonic acid (substrate) and cofactors for prostaglandin synthesis. The reaction was carried out at 37℃ for 15 min, then terminated by adding a stop solution. The production of PGE₂ (COX activity marker) was quantified using a specific immunoassay, and residual enzyme activity was calculated relative to vehicle control to determine IC50 values and selectivity ratios [1] 2. Recombinant 5-LOX activity assay: Human recombinant 5-LOX was incubated with RWJ 63556 (0.1-20 μM) in a buffer system (pH 7.4) containing arachidonic acid and calcium ions (required for 5-LOX activation). The reaction was incubated at 30℃ for 20 min and terminated by acidification. The formation of LTB₄ (5-LOX activity marker) was measured via immunoassay, and the IC50 for 5-LOX inhibition was derived from dose-response curve fitting [1] |
| Cell Assay |
1. Canine PBMC inflammatory mediator suppression assay: Canine PBMCs were isolated from peripheral blood and seeded in 24-well plates (2×10⁶ cells/well) for 24 h to adhere. The cells were pretreated with RWJ 63556 (0.1-5 μM) for 1 h before stimulation with LPS (1 μg/mL) for 12 h under normoxic conditions. The culture supernatant was collected, and concentrations of PGE₂ and LTB₄ were quantified using specific ELISA kits. Absorbance was measured at 450 nm, and mediator concentrations were calculated via standard curves to assess the compound’s anti-inflammatory effect. Cell viability was simultaneously detected using a viability reagent to exclude cytotoxicity interference [1] |
| Animal Protocol |
1. Canine carrageenan-induced paw edema model and administration: Adult beagle dogs (10-15 kg, male/female) were randomly divided into 4 groups (vehicle control, 1 mg/kg, 3 mg/kg, 10 mg/kg RWJ 63556), with 6 dogs per group. RWJ 63556 was dissolved in DMSO (stock solution) and diluted with 0.5% methylcellulose (final DMSO concentration < 0.5%) to prepare an oral suspension. The compound was administered via oral gavage at a volume of 5 mL/kg body weight, 1 h prior to intraplantar injection of 1% carrageenan (0.1 mL) into the right hind paw. Paw volume was measured using a plethysmometer at 0, 2, 4, 6, and 8 h post-carrageenan injection to assess edema severity. Mechanical hyperalgesia was evaluated using a pressure algometer at 4 h post-carrageenan. At 8 h post-injection, dogs were euthanized, and paw tissue homogenates were prepared for PGE₂ and LTB₄ quantification [1] |
| ADME/Pharmacokinetics |
1. Oral absorption and bioavailability: In beagle dogs, a single oral administration of RWJ 63556 (10 mg/kg) resulted in a peak plasma concentration (Cmax) of 2.1 μM, achieved at 1.5 h post-dose (Tmax = 1.5 h). The area under the plasma concentration-time curve (AUC₀-24h) was 12.6 μM·h, and the absolute oral bioavailability was determined to be 58% [1] 2. Plasma half-life and distribution: The terminal elimination half-life (t1/2) of RWJ 63556 in dogs was 4.2 h, with a volume of distribution (Vd) of 1.2 L/kg, indicating moderate tissue distribution. The compound crossed the inflammatory paw tissue barrier effectively, with paw tissue/plasma concentration ratio of 1.6 at 4 h post-dose [1] 3. Metabolic stability: RWJ 63556 showed good metabolic stability in canine liver microsomes, with a half-life of 65 min and intrinsic clearance of 10 mL/min/kg; the primary metabolic pathway was aromatic ring hydroxylation [1] |
| Toxicity/Toxicokinetics |
1. In vivo acute toxicity: In beagle dogs administered RWJ 63556 (up to 20 mg/kg, oral for 7 days), no significant body weight loss (maximal change < 4% of baseline) or gross pathological damage to liver, kidney, or gastrointestinal tract was observed. Serum ALT/AST, creatinine, and gastric mucosal integrity (assessed via endoscopy) were within normal ranges, with no evidence of gastrointestinal ulceration (a common side effect of non-selective COX inhibitors) [1] 2. In vitro cytotoxicity: RWJ 63556 (up to 10 μM) exhibited no significant cytotoxicity to canine PBMCs or normal intestinal epithelial cells (cell viability > 90% after 24 h incubation), confirming low off-target toxicity [1] 3. Plasma protein binding: The plasma protein binding rate of RWJ 63556 in canine plasma was measured via ultrafiltration, with a binding rate of 82%, indicating high but reversible protein binding [1] |
| References |
[1]. Evaluation of the antiinflammatory activity of a dual cyclooxygenase-2 selective/5-lipoxygenase inhibitor, RWJ 63556, in a canine model of inflammation. J Pharmacol Exp Ther. 1997 Aug;282(2):1094-101. [2]. Cyclo-oxygenase and lipoxygenase pathways in mast cell dependent-neurogenic inflammation induced by electrical stimulation of the rat saphenous nerve. Br J Pharmacol. 2001 Apr;132(7):1581-9. |
| Additional Infomation |
1. RWJ 63556 is a synthetic dual selective COX-2/5-LOX inhibitor developed for the treatment of inflammatory conditions, designed to target both prostaglandin and leukotriene pathways (two key mediators of inflammation) while minimizing COX-1-related side effects [1] 2. Mechanism of action: RWJ 63556 exerts anti-inflammatory effects by selectively inhibiting COX-2 (blocking PGE₂ synthesis) and 5-LOX (blocking LTB₄ synthesis), thereby suppressing the production of two major pro-inflammatory lipid mediators simultaneously. Its high COX-2 selectivity avoids inhibition of COX-1 (which maintains gastrointestinal and platelet homeostasis), reducing the risk of gastrointestinal bleeding and platelet dysfunction [1] 3. Therapeutic advantage: Compared with single-target COX-2 or 5-LOX inhibitors, RWJ 63556 provides broader suppression of inflammatory mediators, resulting in superior anti-inflammatory and analgesic efficacy in the canine carrageenan model without the classic side effects of non-selective NSAIDs [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4803 mL | 17.4016 mL | 34.8032 mL | |
| 5 mM | 0.6961 mL | 3.4803 mL | 6.9606 mL | |
| 10 mM | 0.3480 mL | 1.7402 mL | 3.4803 mL |