Physicochemical Properties
| Molecular Formula | C24H22CLF3N2O3S |
| Molecular Weight | 510.956294536591 |
| Exact Mass | 510.099 |
| CAS # | 1423077-49-9 |
| PubChem CID | 71263344 |
| Appearance | White to off-white solid powder |
| LogP | 7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 34 |
| Complexity | 756 |
| Defined Atom Stereocenter Count | 0 |
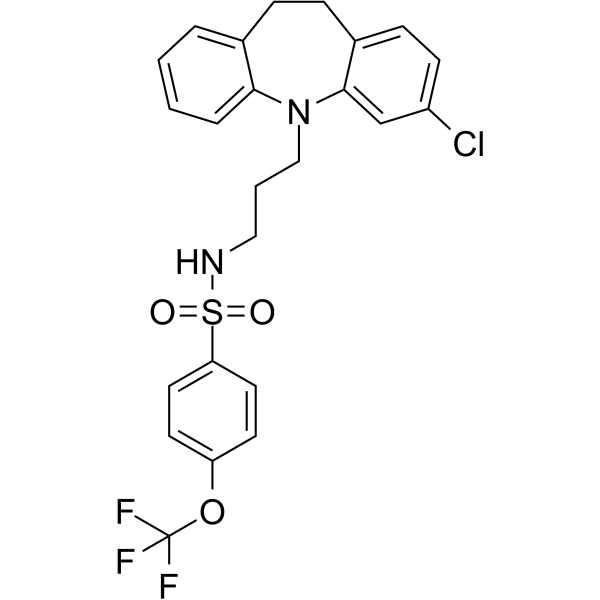
| SMILES | ClC1C=CC2CCC3C=CC=CC=3N(C=2C=1)CCCNS(C1C=CC(=CC=1)OC(F)(F)F)(=O)=O |
| InChi Key | QYOJMNDDVVEPFN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H22ClF3N2O3S/c25-19-9-8-18-7-6-17-4-1-2-5-22(17)30(23(18)16-19)15-3-14-29-34(31,32)21-12-10-20(11-13-21)33-24(26,27)28/h1-2,4-5,8-13,16,29H,3,6-7,14-15H2 |
| Chemical Name | N-[3-(2-chloro-5,6-dihydrobenzo[b][1]benzazepin-11-yl)propyl]-4-(trifluoromethoxy)benzenesulfonamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | The precise molecular target of RTC-5 is not a single kinase. It acts through a novel mechanism, putatively involving the activation of the tumor suppressor Protein Phosphatase 2A (PP2A). This leads to the concomitant negative regulation of the PI3K-AKT and RAS-ERK signaling pathways [1]. |
| ln Vitro |
RTC-5 (0-40 μM; 48 hours) blocks H1650 lung cancer cells from growing, having a GI50 of 12.6 μM [1]. By decreasing the expression of phospho-AKT and phospho-ERK levels, RTC-5 (20–40 μM; 24 hours) negatively affects the PI3K-AKT and RAS-ERK pathways [1]. RTC-5 exhibited anti-proliferative activity in an MTT cell viability assay using H1650 lung adenocarcinoma cells, with a GI50 of 12.6 µM [1]. Treatment with RTC-5 induced a dose-dependent accumulation of cells in the sub-G1 phase and cell cycle arrest, effects that were more pronounced than those induced by the parent compounds trifluoperazine (TFP) or clomipramine (CIP) [1]. RTC-5 induced apoptosis, as confirmed by an increase in Annexin V staining. This apoptotic effect was reversed by co-treatment with the pan-caspase inhibitor Z-VAD, indicating that apoptosis is caspase-mediated [1]. Western blot analysis demonstrated that RTC-5 treatment negatively regulates both the PI3K-AKT and RAS-ERK pathways, as indicated by decreased levels of phospho-AKT and phospho-ERK in H1650 cells [1]. A DiscoverRx kinome screen confirmed that RTC-5 does not inhibit any relevant kinase in an ATP-competitive manner [1]. In radioligand binding assays, RTC-5 showed negligible binding to the serotonin transporter (5-HTT, <5% inhibition at 0.1 mM). It exhibited weak binding to the dopamine transporter (DT) and norepinephrine transporter (NET) only at elevated concentrations (1.0 and 10 mM) [1]. RTC-5 did not show significant binding to a panel of dopamine receptor subtypes (D1-D5) or most serotonin receptor subtypes, except for some residual binding at 5-HT5A at high concentrations (24% at 1.0 mM, 68% at 10 mM) [1]. |
| ln Vivo | In a xenograft study using the H1650 lung cancer cell line in mice, treatment with RTC-5 at 100 mg/kg caused a statistically significant decrease in the mean fold change in tumor volume (1.49 ± 0.26, n=9) compared to the vehicle control group (3.46 ± 0.95, n=7) (p < 0.004, Student's t-test). In contrast, the parent compound TFP could not be dosed higher than 10 mg/kg in the same study due to marked central nervous system (CNS) side effects [1]. |
| Cell Assay |
Cell viability assay [1] Cell Types: H1650 lung adenocarcinoma cells Tested Concentrations: 0 μM, 1 μM, 10 μM, 20 μM, 30 μM, 40 μM Incubation Duration: 48 hrs (hours) Experimental Results: Inhibition of the growth of H1650 lung adenocarcinoma cells. Western Blot Analysis[1] Cell Types: H1650 lung adenocarcinoma cells Tested Concentrations: 20 μM, 40 μM Incubation Duration: 24 hrs (hours) Experimental Results: diminished expression of p-AKT, p-ERK. |
| ADME/Pharmacokinetics | In pharmacokinetic studies in mice, RTC-5 showed significant absorption via the intraperitoneal (IP) route (34-38%) and moderate oral absorption (15-18%). It exhibited moderate clearance (42 ml/min/kg) with a half-life (t1/2) of 0.61 hours [1]. |
| Toxicity/Toxicokinetics |
RTC-5 showed negligible binding to a panel of receptors (M2, H1, H2, hERG) localized to heart tissues and linked to QT interval prolongation [1]. RTC-5 did not bind to a panel of calcium and potassium channels associated with cardiovascular liabilities [1]. In a patch-clamp assay, RTC-5 exhibited negligible effects on the voltage-gated sodium channel Nav1.5 compared to clomipramine (CIP) [1]. |
| References |
[1]. Reengineered tricyclic anti-cancer agents. Bioorg Med Chem. 2015 Oct 1;23(19):6528-34. |
| Additional Infomation |
RTC-5 is a reengineered tricyclic compound, specifically a dibenzazepine derivative where the basic dimethylamine side chain of the parent neuroleptic drug has been replaced with a neutral, polar sulfonamide functional group (4-trifluoromethoxybenzenesulfonamide linked via a three-carbon chain). This key modification was designed to abrogate the CNS-related pharmacology (e.g., dopamine receptor antagonism) of the parent molecules while retaining and optimizing their anticancer "side-effect" [1]. The anticancer effect of RTC-5 is attributed to a novel mechanism involving the putative activation of the tumor suppressor protein phosphatase PP2A, leading to the dual negative regulation of the oncogenic PI3K-AKT and RAS-ERK signaling pathways. This distinguishes it from classical kinase inhibitors [1]. RTC-5 demonstrated in vivo efficacy in an EGFR-driven lung cancer xenograft model without exhibiting the neurotropic (sedative, extrapyramidal) effects that severely limit the dosing of the parent tricyclic drugs in animal models and humans [1]. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~15 mg/mL (~29.36 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.5 mg/mL (2.94 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 15.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.5 mg/mL (2.94 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 15.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9571 mL | 9.7855 mL | 19.5710 mL | |
| 5 mM | 0.3914 mL | 1.9571 mL | 3.9142 mL | |
| 10 mM | 0.1957 mL | 0.9786 mL | 1.9571 mL |