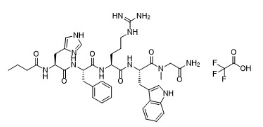
RO27-3225 TFA is a potent and specific melanocortin 4 receptor (MC4R) agonist with an EC50 of 1 nM and 8 nM for MC4R and MC1R, respectively. It shows ~30-fold selectivity for MC4R over MC3R. And it has neuroprotective and anti-inflammatory effects.
Physicochemical Properties
| Molecular Formula | C41H53F3N12O8 |
| Molecular Weight | 898.9 |
| Exact Mass | 898.4061411 |
| Elemental Analysis | C, 54.78; H, 5.94; F, 6.34; N, 18.70; O, 14.24 |
| CAS # | 1373926-49-8 |
| Related CAS # | 274682-89-2;1057258-86-2 (free base isomer);1373926-49-8 (TFA);1051970-60-5 (3 TFA); |
| PubChem CID | 146026285 |
| Sequence | butanoyl-His-Phe-Arg-Trp-Sar-NH2.TFA; N-butanoyl-L-histidyl-L-phenylalanyl-L-arginyl-L-tryptophyl-sarcosinamide trifluoroacetic acid |
| SequenceShortening | HFRWG |
| Appearance | Off-white to light yellow solid powder |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 22 |
| Heavy Atom Count | 64 |
| Complexity | 1470 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | CCCC(=O)N[C@@H](CC1=CN=CN1)C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC3=CNC4=CC=CC=C43)C(=O)N(C)CC(=O)N.C(=O)(C(F)(F)F)O |
| InChi Key | XBNXUPIBUGBMCO-WYDLTDSDSA-N |
| InChi Code | InChI=1S/C39H52N12O6.C2HF3O2/c1-3-10-34(53)47-31(19-26-21-43-23-46-26)37(56)49-30(17-24-11-5-4-6-12-24)36(55)48-29(15-9-16-44-39(41)42)35(54)50-32(38(57)51(2)22-33(40)52)18-25-20-45-28-14-8-7-13-27(25)28;3-2(4,5)1(6)7/h4-8,11-14,20-21,23,29-32,45H,3,9-10,15-19,22H2,1-2H3,(H2,40,52)(H,43,46)(H,47,53)(H,48,55)(H,49,56)(H,50,54)(H4,41,42,44);(H,6,7)/t29-,30-,31-,32-;/m0./s1 |
| Chemical Name | (2S)-N-[(2S)-1-[(2-amino-2-oxoethyl)-methylamino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]-2-[[(2S)-2-[[(2S)-2-(butanoylamino)-3-(1H-imidazol-5-yl)propanoyl]amino]-3-phenylpropanoyl]amino]-5-(diaminomethylideneamino)pentanamide;2,2,2-trifluoroacetic acid |
| Synonyms | RO27-3225 TFA; RO27-3225; RO27 3225; R-O27-3225 TFA; 1373926-49-8; R-O273225 TFA; R-O27-3225 (TFA); (2S)-N-[(2S)-1-[(2-amino-2-oxoethyl)-methylamino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]-2-[[(2S)-2-[[(2S)-2-(butanoylamino)-3-(1H-imidazol-5-yl)propanoyl]amino]-3-phenylpropanoyl]amino]-5-(diaminomethylideneamino)pentanamide;2,2,2-trifluoroacetic acid; CID 9962372; RO273225; RO 273225; RO-273225; Butir-His-D-Phe-Arg-Trp-Sar-NH2 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
RO27-3225 is a selective melanocortin-4 receptor (MC4R) agonist with EC₅₀ values of 0.6 nM at MC4R, 4.8 nM at MC3R, and >10,000 nM at MC1R and MC5R. [1] Functions as a potent and selective MC4R agonist. [2][3] |
| ln Vitro |
RO27-3225 is a cyclic heptapeptide (Ac-Cys¹⁰-[D-Nal⁷,Arg⁸]-Cys⁹-NH₂) with 1,000-fold selectivity for MC4R over MC1R/MC5R. [1] Mechanism: Activates MC4R to modulate central appetite regulation and peripheral anti-inflammatory pathways. [1][2] Confers neuroprotection by promoting angiogenesis and suppressing neuroinflammation post-stroke. [3] |
| ln Vivo |
The intravenous infusion of RO27-3225 (0.012-0.048 mg/kg) in Wistar rats cures hemorrhagic shock, minimizes damage to various organs, and increases survival. After circulatory shock, RO27-3225 may play a protective function against multiple organ failure[2].
In freely feeding rats, intraperitoneal injection of RO27-3225 (0.01-1 mg/kg) dose-dependently reduced food intake by up to 90% at 1 mg/kg within 4 hours without inducing conditioned taste aversion. This effect was absent in MC4R knockout mice. [1] In a rat hemorrhagic shock model, intravenous administration of RO27-3225 (90 μg/kg) restored mean arterial pressure (MAP) from 30±5 mmHg to 85±10 mmHg within 15 minutes, improved organ perfusion, and reduced plasma TNF-α and IL-6 levels by >50%. [2] In mice with middle cerebral artery occlusion (MCAO), daily intraperitoneal injection of RO27-3225 (0.5 mg/kg for 14 days) increased neurogenesis (BrdU⁺/DCX⁺ cells by 2.5-fold), enhanced PDGFRβ⁺ pericytes recruitment, and reduced microglial activation (Iba1⁺ cells decreased by 40%). [3] |
| Animal Protocol |
Animal/Disease Models: Wistar rats of both sexes (270-300 g) with haemorrhagic shock[2] Doses: 0.012 mg/kg, 0.024 mg/kg, 0.048 mg/kg Route of Administration: intravenous (iv) injection Experimental Results: Reversed haemorrhagic shock, decreased multiple organ damage and improved survival. Food intake study: Rats/mice received RO27-3225 (0.01, 0.1, 1 mg/kg) or vehicle via intraperitoneal (i.p.) injection. Food consumption was measured 1-4h post-dosing. Taste aversion assessed by sucrose preference after drug pairing. [1] Hemorrhagic shock model: Rats subjected to 45% blood loss received RO27-3225 (90 μg/kg) or vehicle intravenously at 60 min post-shock. Hemodynamics monitored for 6h; organs harvested for histology/cytokine analysis. [2] Stroke recovery model: MCAO mice treated with RO27-3225 (0.5 mg/kg i.p. daily for 14 days) starting 24h post-surgery. Brains analyzed by immunohistochemistry and ELISA at day 15. [3] |
| Toxicity/Toxicokinetics |
RO27-3225 (1 mg/kg i.p.) did not induce conditioned taste aversion or impair motor coordination in rotarod tests. [1] No acute toxicity observed at 90 μg/kg i.v. in hemorrhagic shock rats. [2] No behavioral abnormalities or mortality reported at 0.5 mg/kg i.p. during 14-day treatment in MCAO mice. [3] |
| References |
[1]. A novel selective melanocortin-4 receptor agonist reduces food intake in rats and mice without producing aversive consequences. J Neurosci. 2000 May 1;20(9):3442-8. [2]. Selective melanocortin MC4 receptor agonists reverse haemorrhagic shock and prevent multiple organ damage. Br J Pharmacol. 2007 Mar;150(5):595-603. [3]. Effects of RO27-3225 on neurogenesis, PDGFRβ+ cells and neuroinflammation after cerebral infarction. Int Immunopharmacol. 2020 Feb 11;81:106281. |
| Additional Infomation |
Background and purpose: In circulatory shock, melanocortins have life-saving effects likely to be mediated by MC4 receptors. To gain direct insight into the role of melanocortin MC4 receptors in haemorrhagic shock, we investigated the effects of two novel selective MC4 receptor agonists.
Experimental approach: Severe haemorrhagic shock was produced in rats under general anaesthesia. Rats were then treated with either the non-selective agonist [Nle4, D-Phe7]-melanocyte-stimulating hormone (NDP--MSH) or with the selective MC4 agonists RO27-3225 and PG-931. Cardiovascular and respiratory functions were continuously monitored for 2 h; survival rate was recorded up to 24 h. Free radicals in blood were measured using electron spin resonance spectrometry; tissue damage was evaluated histologically 25 min or 24 h after treatment.
Key results: All shocked rats treated with saline died within 30-35 min. Treatment with NDP--MSH, RO27-3225 and PG-931 produced a dose-dependent (13-108 nmol kg-1 i.v.) restoration of cardiovascular and respiratory functions, and improved survival. The three melanocortin agonists also markedly reduced circulating free radicals relative to saline-treated shocked rats. All these effects were prevented by i.p. pretreatment with the selective MC4 receptor antagonist HS024. Moreover, treatment with RO27-3225 prevented morphological and immunocytochemical changes in heart, lung, liver, and kidney, at both early (25 min) and late (24 h) intervals.
Conclusions and implications: Stimulation of MC4 receptors reversed haemorrhagic shock, reduced multiple organ damage and improved survival. Our findings suggest that selective MC4 receptor agonists could have a protective role against multiple organ failure following circulatory shock.[2] Cerebral infarction causes severe social and economic burdens to patients due to its high morbidity and mortality rates, and the available treatments are limited. RO27-3225 is a highly selective melanocortin receptor 4 agonist that alleviates damage in many nervous system diseases, such as cerebral hemorrhage, traumatic brain injury and chronic neurodegenerative diseases. However, the effect of RO27-3225 on cerebral infarction remains unclear. In this study, we used a mouse model of transient middle cerebral artery occlusion (tMCAO) and administered RO27-3225 or saline to the mice through intraperitoneal injection. RO27-3225 increased the number of Nestin+/BrdU+ cells and doublecortin (DCX)+/BrdU+ cells in the subventricular zone (SVZ) and the number of DCX+/BrdU+ cells in the peri-infarct area on day 7 after tMCAO. Furthermore, RO27-3225 decreased the number of activated microglia (Iba1+ cells with a specific morphology) and the expression levels of Iba1, TNFα, IL6, and iNOS proteins and increased the number of PDGFRβ+ cells in the peri-infarct region on day 3 after tMCAO. Finally, RO27-3225-treated mice exhibited significantly decreased infarct volumes, brain water contents, and neurological deficits after cerebral infarction. Thus, RO27-3225 can improve the outcomes following cerebral infarction, partially by regulating neurogenesis in the SVZ, PDGFRβ+ cell survival and neuroinflammation in the peri-infarct zone. Our research reveals that RO27-3225 is a potential new treatment for cerebral infarction.[3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1125 mL | 5.5624 mL | 11.1247 mL | |
| 5 mM | 0.2225 mL | 1.1125 mL | 2.2249 mL | |
| 10 mM | 0.1112 mL | 0.5562 mL | 1.1125 mL |