RK-33 is a potent and first-in-class small molecule inhibitor of DDX3 (a RNA helicase) and causes G1 cell cycle arrest, induces apoptosis, and promotes radiation sensitization in DDX3-overexpressing cells. RK-33 was reported to bind to DDX3 and abrogated its activity. Inhibition of DDX3 by RK-33 resulted in G1 cell cycle arrest, induced apoptosis, and promoted radiation sensitization in DDX3-overexpressing cells. Moreover, the loss of DDX3 function caused by RK-33 impaired Wnt signaling via disruption of the DDX3-β-catenin axis. RK-33 binds specifically to DDX3, but not to the closely related proteins DDX5 and DDX17. RK-33 inhibits cancer growth and radiosensitizes lung cancer cells in a DDX3-dependent manner.
Physicochemical Properties
| Molecular Formula | C23H20N6O3 | |
| Molecular Weight | 428.44 | |
| Exact Mass | 428.159 | |
| CAS # | 1070773-09-9 | |
| Related CAS # |
|
|
| PubChem CID | 46184988 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 677.0±65.0 °C at 760 mmHg | |
| Flash Point | 363.3±34.3 °C | |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C | |
| Index of Refraction | 1.698 | |
| LogP | 1.35 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 32 | |
| Complexity | 783 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | COUMZXFUZDBRCZ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C23H20N6O3/c1-31-17-7-3-15(4-8-17)11-28-14-26-19-20-22(25-13-24-21(19)28)29(23(30)27-20)12-16-5-9-18(32-2)10-6-16/h3-10,13-14H,11-12H2,1-2H3 | |
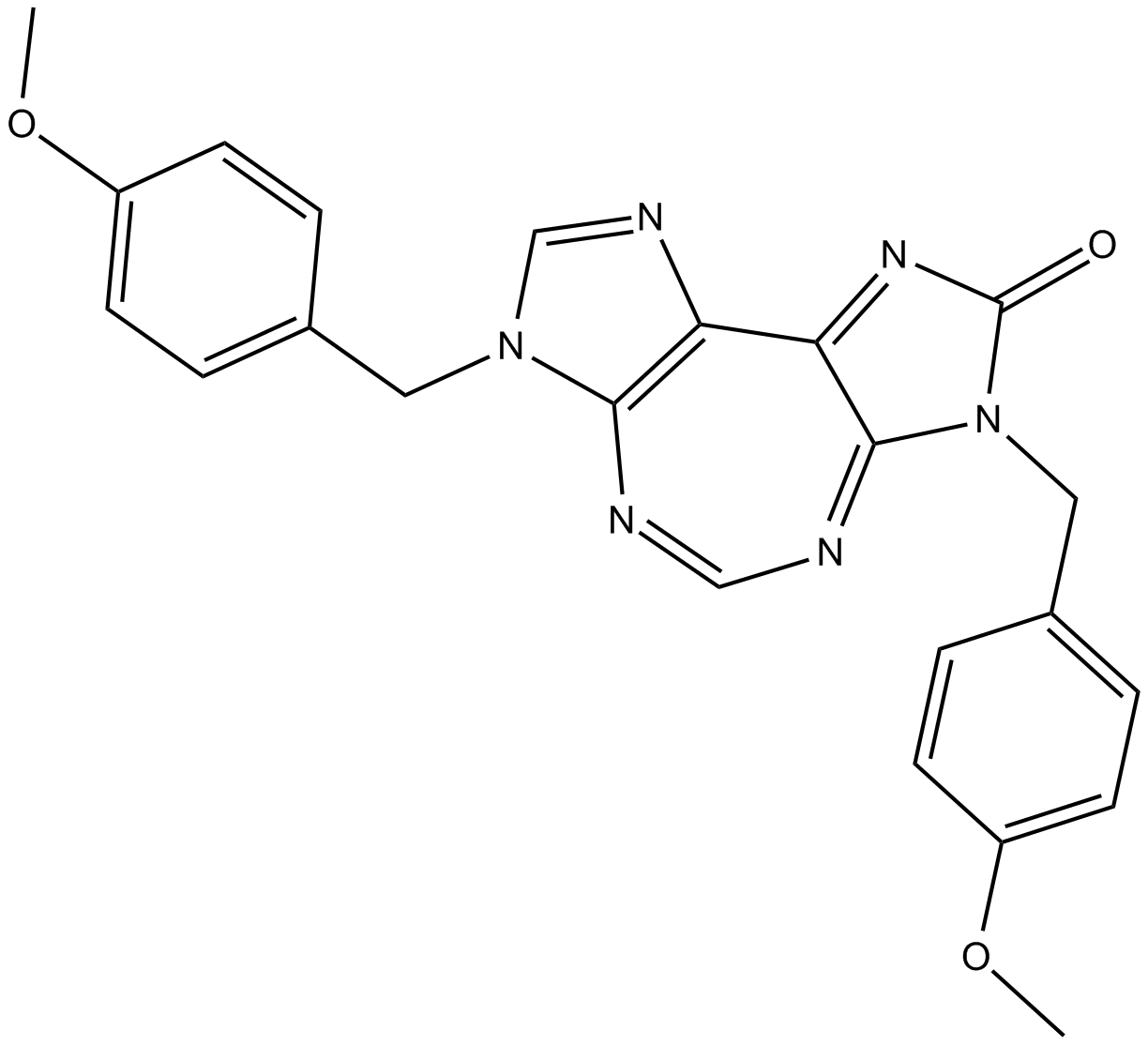
| Chemical Name | 3,7-dihydro-3,7-bis[(4-methoxyphenyl)methyl]-2H-diimidazo[4,5-d:4,5-f][1,3]diazepin-2-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
RK-33 targets RNA helicase DDX3 (IC50 = 4.6 μM for DDX3 ATPase activity; Ki = 3.2 μM for DDX3 binding) [1] |
| ln Vitro |
With an IC50 of 3-6 µM, RK-33 inhibits a wide variety of cancer cells, however PC3 is far less susceptible to its effects (IC50 >12 µM). While RK-33 treatment only moderately accumulates the G1 phase in 22Rv1, it drastically reduces the G2 phase in treated cells and induces a large accumulation in the G1 phase in DU145 and LNCaP. In 22Rv1, RK-33 therapy also results in 12 moderate G1 accumulation[1]. Equal doses of empty NPs had no killing impact on MCF-7 cells, while -loaded NPs exhibit dose-dependent cytotoxicity. 50 μg/mL is the IC50 value for 5% RK-33 loaded NPs and 25 μg/mL for 10% RK-33 loaded NPs[2]. In human prostate cancer cell lines (DU145, PC-3, LNCaP), RK-33 (1–20 μM) dose-dependently inhibits cell proliferation, with IC50 values of 5.8 μM (DU145), 7.2 μM (PC-3), and 9.5 μM (LNCaP) [1] - It blocks DDX3 ATPase activity and RNA helicase function: RK-33 (5 μM) reduces DDX3-mediated ATP hydrolysis by ~68% and inhibits DDX3-dependent RNA unwinding by ~72% [1] - In DU145 cells, RK-33 (5 μM) + radiation (2 Gy) synergistically inhibits cell proliferation (combination index = 0.48) and induces apoptosis (Annexin V-FITC/PI staining shows apoptotic rate ~65% vs. 22% for radiation alone) [1] - It downregulates DDX3 downstream signaling: Western blot shows reduced phosphorylation of AKT (Ser473) and ERK1/2 (Thr202/Tyr204), and decreased expression of Cyclin D1 and Bcl-2 in DU145 cells (5 μM treatment for 24 hours) [1] - PLGA nanoparticle-formulated RK-33 (NP-RK-33, 1–20 μM) shows enhanced cellular uptake in PC-3 cells (2.3-fold higher than free RK-33) and increased antiproliferative activity (IC50 = 3.1 μM for PC-3) [2] - It shows no significant cytotoxicity to normal human prostate epithelial cells (PrEC) at concentrations up to 20 μM (cell viability >85% vs. control) [1] - In clonogenic assay, RK-33 (5 μM) + radiation (4 Gy) reduces colony formation of DU145 cells by ~82% (vs. 35% for radiation alone) [1] |
| ln Vivo |
In comparison to the control or single treatment groups, the tumors from mice in the combination RK-33 and radiation group showed higher levels of interstitial edema and cell death (pyknotic or condensed nuclei mixed with fibrin). Radiation therapy plus RK-33 treatment has an advantage in slowing tumor growth[1]. RK-33 is found in the liver (28 μg/g) and plasma (34 μg/mL) of mice given RK-33-PLGA treatment, but not in the lungs[2]. In DU145 (prostate cancer) subcutaneous xenograft model (nude mice): Intraperitoneal administration of RK-33 (20 mg/kg/day) + radiation (2 Gy, 3 times/week) for 21 days inhibits tumor growth by ~78% vs. vehicle + radiation. Tumor tissues show reduced DDX3 expression, p-AKT, Ki-67, and increased cleaved caspase-3 levels (immunohistochemistry) [1] - In PC-3 (prostate cancer) subcutaneous xenograft model (nude mice): Intravenous injection of NP-RK-33 (15 mg/kg/day, PLGA nanoparticle formulation) for 14 days inhibits tumor growth by ~65% vs. free RK-33 (42% inhibition at the same dose). NP-RK-33 increases tumor tissue drug concentration by ~3.5-fold vs. free drug [2] - In DU145 xenografts, RK-33 + radiation prolongs median survival of mice from 45 days (vehicle + radiation) to 68 days [1] |
| Enzyme Assay |
DDX3 ATPase activity assay: Recombinant human DDX3 protein (10 nM) was incubated with ATP (1 mM) and reaction buffer (20 mM Tris-HCl pH 7.5, 10 mM MgCl2, 1 mM DTT) at 37°C for 60 minutes. RK-33 (0.1–50 μM) was added 10 minutes before ATP addition. Released inorganic phosphate (Pi) was detected by malachite green assay. Inhibition rate was calculated relative to vehicle control, and IC50 was determined by nonlinear regression [1] - DDX3 binding assay (SPR): Recombinant DDX3 protein was immobilized on a CM5 sensor chip. RK-33 (0.5–50 μM) was injected at a constant flow rate (30 μL/min) in running buffer (PBS pH 7.4, 0.05% Tween 20). Sensorgrams were recorded to measure binding affinity, and Ki value was calculated using steady-state affinity model [1] |
| Cell Assay |
Prostate cancer cell proliferation and radiosensitization assay: DU145/PC-3/LNCaP cells (5×10³ per well) were seeded in 96-well plates, pretreated with RK-33 (1–20 μM) for 1 hour, then exposed to radiation (0–8 Gy) for 72 hours. Cell viability was measured by MTT assay to determine IC50 and combination index [1] - DDX3 signaling and apoptosis assay: DU145 cells (1×10⁶ per well) were seeded in 6-well plates, treated with RK-33 (5–10 μM) + radiation (2 Gy) for 24 hours. Cells were lysed, and Western blot detected DDX3, p-AKT, AKT, p-ERK1/2, ERK1/2, Cyclin D1, Bcl-2, cleaved caspase-3, and GAPDH. Apoptosis was analyzed by Annexin V-FITC/PI staining and flow cytometry [1] - PLGA nanoparticle cellular uptake assay: PC-3 cells were incubated with fluorescently labeled NP-RK-33 or free RK-33 (10 μM) for 4 hours. Cellular uptake was quantified by flow cytometry and confocal microscopy [2] - Clonogenic assay: DU145 cells (1×10³ per well) were seeded in 6-well plates, pretreated with RK-33 (1–10 μM) for 1 hour, irradiated (2–8 Gy), and cultured for 14 days. Colonies were stained with crystal violet, and colonies with >50 cells were counted [1] |
| Animal Protocol |
The effect of RK-33 with a fractional dosing regimen was studied in the Twist1/KrasG12D lung cancer model. Results showed that during the 3 weeks treatment, a modest decrease in tumor growth with radiation and even more so with the combination of RK-33 and radiation. Therefore, these data indicated that RK-33 in combination with hypofractionated radiation was able to decrease lung tumor load effectively in preclinical lung cancer models and performed much better than the commonly used radiosensitizer carboplatin. Prostate cancer xenograft radiotherapy combination model (DU145): 6-week-old male nude mice were subcutaneously injected with DU145 cells (5×10⁶ cells/mouse). When tumors reached ~100 mm³, mice were randomized into control (vehicle), radiation alone (2 Gy, 3 times/week), RK-33 alone (20 mg/kg/day, i.p.), and combination groups (n = 6 per group). RK-33 was dissolved in DMSO (10%) + saline (90%), administered intraperitoneally once daily for 21 days. Radiation was delivered on days 1, 4, 7. Tumor volume (length×width²/2) and body weight were measured every 3 days; tumors were excised for immunohistochemistry [1] - PLGA nanoparticle efficacy model (PC-3): 6-week-old male nude mice were subcutaneously injected with PC-3 cells (5×10⁶ cells/mouse). When tumors reached ~120 mm³, mice were divided into free RK-33 (15 mg/kg/day, i.v.) and NP-RK-33 (15 mg/kg/day, i.v.) groups (n = 6 per group). NP-RK-33 was formulated with PLGA (50:50) via double emulsion method, suspended in saline. Drugs were administered intravenously once daily for 14 days. Tumor volume and body weight were measured every 2 days; tumor tissues were collected to quantify drug concentration [2] |
| ADME/Pharmacokinetics |
Oral bioavailability: Free RK-33 shows low oral bioavailability (~18%) in rats [2] - PLGA nanoparticle-improved PK: NP-RK-33 (15 mg/kg, i.v.) in rats shows a plasma half-life (t1/2) of 8.6 hours, vs. 2.3 hours for free RK-33 [2] - Tumor penetration: NP-RK-33 increases tumor tissue concentration of RK-33 to 4.8 μg/g, vs. 1.3 μg/g for free RK-33 (15 mg/kg, i.v.) [2] - Plasma protein binding rate: 89% in human plasma, 87% in rat plasma (equilibrium dialysis assay) [1] |
| Toxicity/Toxicokinetics |
In vitro toxicity: RK-33 at concentrations up to 20 μM shows no significant cytotoxicity to normal human PrEC or peripheral blood mononuclear cells (PBMCs) (cell viability >85% vs. control) [1] - Acute toxicity: LD50 > 200 mg/kg in rats (intraperitoneal administration); no mortality or severe toxic symptoms (lethargy, convulsions) observed at doses up to 200 mg/kg [1] - Repeat-dose toxicity: In a 21-day study in rats (intraperitoneal doses of 10, 20, 40 mg/kg/day), the drug was well-tolerated. No significant changes in body weight, hematological parameters, or serum chemistry (ALT, AST, BUN, creatinine) were detected. Histological examination of liver, kidney, and prostate revealed no abnormal lesions [1] - NP-RK-33 toxicity: Mice treated with NP-RK-33 (15 mg/kg/day, i.v.) for 14 days show no increased toxicity vs. free RK-33, with normal organ function and no inflammatory response in tumor tissues [2] |
| References |
[1]. RK-33 radiosensitizes prostate cancer cells by blocking the RNA helicase DDX3. Cancer Res. 2016 Sep 12. [2]. Bol GM, e tal. PLGA nanoparticle formulation of RK-33: an RNA helicase inhibitor against DDX3. Cancer Chemother Pharmacol. 2015 Oct;76(4):821-7. |
| Additional Infomation |
RK-33 is a small-molecule inhibitor of RNA helicase DDX3, with radiosensitizing activity in prostate cancer [1] - Its mechanism of action involves binding to DDX3’s ATP-binding pocket, inhibiting DDX3 ATPase and RNA helicase activity, thereby blocking DDX3-mediated downstream signaling (PI3K-AKT-ERK pathway) and inducing cell cycle arrest (G1 phase) and apoptosis [1] - It enhances radiotherapy efficacy by suppressing DDX3-dependent DNA damage repair, making cancer cells more sensitive to radiation-induced cell death [1] - PLGA nanoparticle formulation (NP-RK-33) improves RK-33 solubility, prolongs circulation time, enhances tumor targeting, and increases in vivo efficacy compared to free drug [2] - Preclinical data supports its potential as a radiosensitizer for the treatment of advanced prostate cancer, especially in combination with radiotherapy [1,2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3340 mL | 11.6702 mL | 23.3405 mL | |
| 5 mM | 0.4668 mL | 2.3340 mL | 4.6681 mL | |
| 10 mM | 0.2334 mL | 1.1670 mL | 2.3340 mL |