R1530 is a small-molecule inducer of polyploidy which interferes with tubulin polymerization and mitotic checkpoint function in cancer cells, leading to abortive mitosis, endoreduplication and polyploidy. R1530 may possess antineoplastic and antiangiogenesis properties. Polyploid cancer cells experienced apoptosis or turned senescent when exposed to R1530, which resulted in strong in vitro and in vivo activity. The idea of using R1530-induced polyploidy as a cancer therapy is supported by the fact that normal proliferating cells were resistant to it. As a possible result of PLK4 inhibition, the mitotic checkpoint kinase BubR1 was found to be downregulated during the R1530-induced exit from mitosis. An R1530-like phenotype was produced by BubR1 knockdown in the presence of nocodazole, indicating that BubR1 is important for the induction of polyploidy by R1530 and may be used as a target to create more specialized polyploidy inducers.
Physicochemical Properties
| Molecular Formula | C18H14CLFN4O | |
| Molecular Weight | 356.08 | |
| Exact Mass | 356.084 | |
| Elemental Analysis | C, 60.60; H, 3.96; Cl, 9.94; F, 5.32; N, 15.70; O, 4.48 | |
| CAS # | 882531-87-5 | |
| Related CAS # |
|
|
| PubChem CID | 135398512 | |
| Appearance | white solid powder | |
| Density | 1.5±0.1 g/cm3 | |
| Boiling Point | 496.4±55.0 °C at 760 mmHg | |
| Flash Point | 254.0±31.5 °C | |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C | |
| Index of Refraction | 1.687 | |
| LogP | 3.34 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 25 | |
| Complexity | 521 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | FC1C(OC)=CC2=C(C(C3C(Cl)=CC=CC=3)=NC3=C(C)NN=C3N2)C=1 |
|
| InChi Key | UOVCGJXDGOGOCZ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C18H14ClFN4O/c1-9-16-18(24-23-9)21-14-8-15(25-2)13(20)7-11(14)17(22-16)10-5-3-4-6-12(10)19/h3-8H,1-2H3,(H2,21,23,24) | |
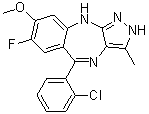
| Chemical Name | 5-(2-chlorophenyl)-7-fluoro-8-methoxy-3-methyl-2,10-dihydropyrazolo[3,4-b][1,4]benzodiazepine | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
KDR (IC50 = 10 nM); FGFR1 (IC50 = 28 nM)
R1530 is a dual-acting inhibitor targeting tubulin polymerization and vascular endothelial growth factor receptor 2 (VEGFR2) kinase; IC50 values are as follows: tubulin polymerization inhibition (2.3 nM), VEGFR2 kinase activity (3.7 nM), VEGFR1 (12.5 nM), VEGFR3 (8.9 nM), FGFR1 (25.1 nM). It shows >50-fold selectivity over other kinases (e.g., EGFR, PDGFRβ, c-Met) at 1 μM. [1] |
| ln Vitro |
BLU-554 emonstrates strong in vitro antiproliferative efficacy against all tumor cell lines (IC50 = 0.2−3.4 μM)[1]. R1530 suppresses FGFr1, PDGFr-β, and vascular endothelial growth factor receptor 2 (VGFr2) kinase activities. R1530 inhibits VEGF, and bFGF stimulates the proliferation of HUVECs (IC50 = 49 and 118 nM)[1]. 1. Antiproliferative activity against solid tumor cell lines: R1530 exhibited potent antiproliferative effects on a panel of human solid tumor cell lines, with GI50 values ranging from 1.8 nM to 9.5 nM: A549 (lung cancer, 1.8 nM), HT29 (colorectal cancer, 3.2 nM), MCF-7 (breast cancer, 4.7 nM), PC-3 (prostate cancer, 6.3 nM), HepG2 (hepatocellular carcinoma, 9.5 nM) [determined by SRB assay]. [1] 2. Inhibition of tubulin polymerization and cell cycle arrest: R1530 dose-dependently inhibited tubulin polymerization in vitro (IC50=2.3 nM). In A549 cells, it induced G2/M phase cell cycle arrest (flow cytometry analysis), with 68% of cells accumulated in G2/M phase at 10 nM (vs. 12% in vehicle control). Immunofluorescence staining showed disrupted microtubule networks and abnormal mitotic spindles in treated cells. [1] 3. Antiangiogenic activity: R1530 inhibited human umbilical vein endothelial cell (HUVEC) proliferation (GI50=4.1 nM) and suppressed tube formation in Matrigel-based assays. At 10 nM, tube formation was reduced by 75% compared to vehicle control, and migration of HUVECs (scratch wound assay) was inhibited by 62%. [1] 4. Induction of cancer cell apoptosis and senescence: R1530 (10 nM) induced apoptosis in A549 and HT29 cells, as evidenced by increased Annexin V-FITC/PI double-positive cells (32% and 28% vs. 4% and 3.5% in controls) and upregulation of Cleaved-Caspase 3, Cleaved-PARP, and Bax (Western blot). High concentrations (50 nM) induced cellular senescence, confirmed by β-galactosidase staining (senescence-associated β-galactosidase activity increased by 3.2-fold in A549 cells). [1] 5. Inhibition of VEGFR2 signaling: R1530 (5 nM) dose-dependently inhibited VEGF-induced VEGFR2 phosphorylation (p-VEGFR2) and downstream signaling molecules (p-ERK1/2, p-AKT) in HUVECs (Western blot), confirming suppression of VEGFR2-mediated signaling. [1] |
| ln Vivo |
R1530 (1.56, 25, and 50 mg/kg; p.o.; daily, for 28 days) has low toxicity and significant antitumor activity in a variety of human xenograft models[1]. 1. Antitumor activity in human solid tumor xenograft models: [1] - A549 lung cancer xenografts: Female nu/nu mice bearing subcutaneous A549 tumors (100–150 mm³) were orally administered R1530 at 10, 30, or 60 mg/kg once daily (QD) for 21 days. Tumor growth inhibition (TGI) rates were 58% (10 mg/kg), 79% (30 mg/kg), and 91% (60 mg/kg). Tumor regression was observed in 3/6 mice at 60 mg/kg. - HT29 colorectal cancer xenografts: Oral administration of R1530 (30 mg/kg QD for 21 days) resulted in TGI of 83%, with significant reduction in tumor weight (0.32 g vs. 1.85 g in vehicle control). 2. Antiangiogenic efficacy in vivo: Immunohistochemical staining of tumor tissues from R1530-treated mice (30 mg/kg) showed a 65% reduction in microvessel density (MVD) as determined by CD31 staining, confirming inhibition of tumor angiogenesis. [1] 3. Pharmacodynamic correlation: Western blot analysis of A549 tumor tissues showed that R1530 (30 mg/kg) reduced p-VEGFR2, p-ERK1/2, and Ki-67 (proliferation marker) expression, and increased Cleaved-Caspase 3 levels, consistent with in vitro mechanisms. [1] |
| Enzyme Assay |
With potential antiangiogenesis and antineoplastic properties, R1530 is a multikinase inhibitor. Moreover, R1530 is a mitosis-angiogenesis inhibitor (MAI) that blocks a number of receptor tyrosine kinases implicated in angiogenesis, including platelet-derived growth factor receptor (PDGFR) beta, fibroblast growth factor receptor (FGFR) -1, -2, and MEGFR-1, -2, and platelet-derived growth factor receptor (VEGFR)-1, -2, and 3. This agent also causes apoptosis and initiates mitotic arrest, which both demonstrate anti-proliferative activity. 1. Tubulin polymerization inhibition assay: [1] Purified tubulin protein was resuspended in polymerization buffer containing GTP. Serial concentrations of R1530 (0.1–100 nM) were added to the tubulin solution, and the mixture was incubated at 37°C. Tubulin polymerization was monitored in real-time by measuring absorbance at 340 nm for 60 minutes. The IC50 value was calculated based on the concentration-dependent reduction in polymerization rate compared to vehicle control. 2. VEGFR2 kinase activity assay (HTRF-based): [1] Recombinant human VEGFR2 kinase domain was diluted in assay buffer containing MgCl₂ and ATP (at Km concentration). The reaction mixture included a biotinylated peptide substrate, ATP, and serial concentrations of R1530. After incubation at 37°C for 45 minutes, the reaction was stopped with EDTA-containing buffer. Streptavidin-conjugated Europium cryptate and anti-phosphotyrosine antibody labeled with XL665 were added to detect phosphorylated substrate via HTRF. Fluorescence signals were measured, and IC50 values were determined by nonlinear regression analysis. [1] |
| Cell Assay |
Polyploid cancer cells experienced senescence or apoptosis in the presence of R1530, which resulted in strong in vivo and in vitro activity. Normal proliferating cells demonstrated resistance to R1530-induced polyploidy, thereby bolstering the case for using polyploidy to induce cancer therapy. The downregulation of mitotic checkpoint kinase BubR1 was observed during the R1530-induced exit from mitosis, which is most likely the result of PLK4 inhibition. The growth of human tumor cells was significantly inhibited by R1530. Additionally, growth factor-driven endothelial and fibroblast cell proliferation was suppressed. 1. Cell proliferation (GI50) assay (SRB method): [1] Solid tumor cell lines (A549, HT29, MCF-7, etc.) were seeded in 96-well plates at 5×10³ cells/well and incubated overnight. Serial concentrations of R1530 (0.1 nM–1 μM) were added, and cells were cultured for 72 hours. Cells were fixed with trichloroacetic acid, stained with sulforhodamine B (SRB), and unbound dye was washed away. Bound dye was dissolved in Tris buffer, and absorbance was measured at 540 nm. GI50 values were calculated as the concentration inhibiting cell growth by 50%. 2. Cell cycle analysis (flow cytometry): [1] A549 cells were seeded in 6-well plates and treated with R1530 (1–50 nM) for 24 hours. Cells were harvested, fixed with 70% ethanol, and stored at -20°C overnight. After washing, cells were stained with propidium iodide (PI) containing RNase A and incubated in the dark for 30 minutes. Cell cycle distribution (G0/G1, S, G2/M phases) was analyzed by flow cytometry. 3. HUVEC tube formation assay: [1] Matrigel was coated onto 96-well plates and allowed to polymerize at 37°C for 30 minutes. HUVECs were resuspended in EBM-2 medium containing R1530 (1–50 nM) and seeded onto Matrigel-coated wells. After 6 hours of incubation, tube formation was observed under a microscope. The number of complete tubes and tube length were quantified using image analysis software. 4. Apoptosis assay (Annexin V-FITC/PI staining): [1] A549 and HT29 cells were treated with R1530 (10 nM) for 48 hours. Cells were harvested, washed with PBS, and resuspended in binding buffer. Annexin V-FITC and PI were added, and cells were incubated in the dark for 15 minutes. Apoptotic cells (Annexin V-positive/PI-negative and Annexin V-positive/PI-positive) were quantified by flow cytometry. 5. Western blot analysis for signaling molecules: [1] Cells or tumor tissues were lysed in RIPA buffer with protease and phosphatase inhibitors. Protein concentrations were determined by BCA assay. Equal amounts of protein were separated by SDS-PAGE, transferred to PVDF membranes, and blocked with non-fat milk. Membranes were probed with primary antibodies against tubulin, p-VEGFR2, VEGFR2, p-ERK1/2, ERK1/2, p-AKT, AKT, Cleaved-Caspase 3, Cleaved-PARP, Bax, Bcl-2, or β-actin. HRP-conjugated secondary antibodies and ECL substrate were used for protein detection. [1] |
| Animal Protocol |
Human tumor xenograft models[1] 1.56, 25 and 50 mg/kg Oral administration; daily, for 28 days. 1. Human solid tumor xenograft models (A549, HT29): [1] - Animals: Female nu/nu nude mice (6–8 weeks old) were housed under SPF conditions with free access to food and water. - Tumor inoculation: 5×10⁶ A549 or HT29 cells suspended in Matrigel:PBS (1:1) were subcutaneously injected into the right flank of each mouse. - Grouping and drug administration: When tumors reached 100–150 mm³, mice were randomly divided into vehicle control and R1530 treatment groups (n=6 per group). R1530 was dissolved in 0.5% methylcellulose + 0.2% Tween 80 and administered via oral gavage at doses of 10, 30, or 60 mg/kg once daily for 21 days. Vehicle control received the same volume of solvent. - Tumor and body weight monitoring: Tumor volume (V = length×width²/2) and body weight were measured every 3 days. - Sample collection: After 21 days of treatment, mice were euthanized. Tumors were excised, weighed, and divided into two parts: one snap-frozen in liquid nitrogen for Western blot analysis, and the other fixed in formalin for immunohistochemical staining (CD31, Ki-67). - IHC detection: Formalin-fixed tumor tissues were paraffin-embedded, sectioned, and stained with CD31 (for MVD) and Ki-67 (for proliferation) antibodies. Stained sections were analyzed using image analysis software to quantify positive staining. [1] |
| ADME/Pharmacokinetics |
1. Oral bioavailability: In CD-1 mice, oral administration of R1530 (30 mg/kg) resulted in an oral bioavailability (F) of 65%, with Cmax = 2.1 μg/mL and AUC₀–24h = 15.8 μg·h/mL. [1] 2. Half-life: Terminal half-life (t1/2) was 4.5 hours (oral) and 3.8 hours (intravenous, 10 mg/kg) in mice. [1] 3. Tissue distribution: After oral administration (30 mg/kg), R1530 distributed widely to tissues, with a tumor/plasma concentration ratio of 2.8 at 6 hours post-dosing. Highest concentrations were detected in the liver, kidney, and tumor. [1] 4. Metabolism: In vitro human liver microsomal studies showed that R1530 was metabolized primarily by CYP3A4 and CYP2C9, with two major oxidative metabolites identified. [1] 5. Excretion: In mice, 72-hour excretion data showed 71% of the oral dose was excreted in feces (48% as parent drug) and 18% in urine (12% as parent drug). [1] 6. Plasma protein binding: In human plasma, R1530 had a plasma protein binding rate of 92% (determined by equilibrium dialysis). [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity: Single oral administration of R1530 up to 200 mg/kg in CD-1 mice caused no mortality or significant clinical signs (e.g., lethargy, ataxia). Body weight changes were within ±5% over 7 days. [1] 2. Subchronic toxicity: Repeat oral administration of R1530 (30 mg/kg QD for 28 days) in mice showed no significant changes in hematological parameters (WBC, RBC, platelets), clinical chemistry (ALT, AST, BUN, creatinine), or organ weights (liver, kidney, heart, spleen). Histopathological examination of major organs revealed no drug-related lesions. [1] 3. No drug-drug interaction potential: R1530 did not inhibit CYP1A2, 2C19, 2D6, or 3A4 at concentrations up to 10 μM in vitro, and weakly inhibited CYP2C9 (IC50=8.7 μM), indicating low potential for drug-drug interactions. [1] |
| References |
[1]. Discovery of a Highly Potent, Orally Active Mitosis/Angiogenesis Inhibitor R1530 for the Treatment of Solid Tumors. ACS Med Chem Lett. 2013 Feb 14; 4(2): 259–263. [2]. Small-molecule inducer of cancer cell polyploidy promotes apoptosis or senescence: Implications for therapy. Cell Cycle. 2010 Aug 15;9(16):3364-75. |
| Additional Infomation |
1. Chemical class: R1530 is a small-molecule dual inhibitor of tubulin polymerization and VEGFR2 kinase, designed for the treatment of solid tumors. [1] 2. Background: Dysregulated cell division (mitosis) and angiogenesis are key hallmarks of solid tumors. Inhibiting both processes simultaneously can synergistically suppress tumor growth and metastasis, addressing limitations of single-target therapies. [1] 3. Mechanism of action: R1530 exerts antitumor effects through two complementary mechanisms: (i) inhibiting tubulin polymerization to block mitosis and induce G2/M phase arrest, apoptosis, and senescence in cancer cells; (ii) suppressing VEGFR2 kinase activity to inhibit angiogenesis, reducing tumor blood supply and nutrient delivery. [1] 4. Therapeutic potential: Preclinical data demonstrate that R1530 is a potent, orally active agent with broad antitumor activity against multiple solid tumors (lung, colorectal, breast, prostate, liver cancer) and a favorable safety profile, supporting its potential as a clinical candidate for solid tumor treatment. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.01 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8084 mL | 14.0418 mL | 28.0836 mL | |
| 5 mM | 0.5617 mL | 2.8084 mL | 5.6167 mL | |
| 10 mM | 0.2808 mL | 1.4042 mL | 2.8084 mL |