Physicochemical Properties
| Molecular Formula | C20H24N2O2.H2O4S |
| Molecular Weight | 422.49524 |
| Exact Mass | 746.335 |
| CAS # | 549-56-4 |
| Related CAS # | Quinine;130-95-0;Quinine hydrobromide;549-49-5 |
| PubChem CID | 11949689 |
| Appearance | White to off-white solid powder |
| Boiling Point | 1136.7ºC at 760mmHg |
| Flash Point | 641.2ºC |
| LogP | 6.65 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 538 |
| Defined Atom Stereocenter Count | 4 |
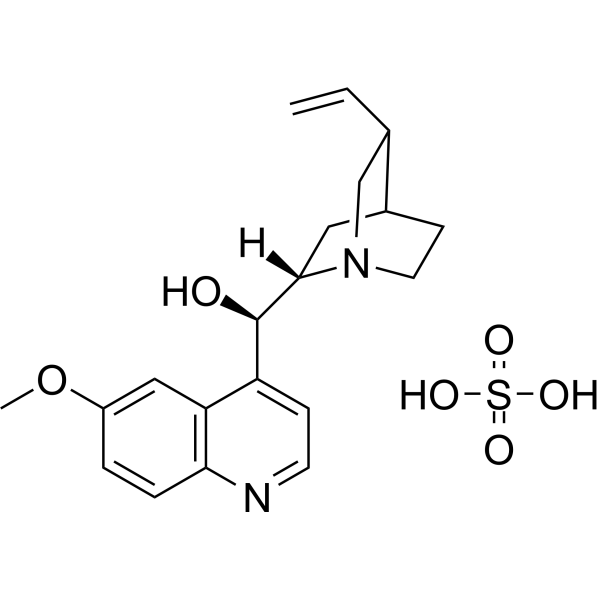
| SMILES | C=C[C@H]1C[N@](CC[C@H]1C2)[C@@H]2[C@H](O)C3=CC=NC4=CC=C(OC)C=C34.O=S(O)(O)=O |
| InChi Key | AKYHKWQPZHDOBW-DSXUQNDKSA-N |
| InChi Code | InChI=1S/C20H24N2O2.H2O4S/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18;1-5(2,3)4/h3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3;(H2,1,2,3,4)/t13-,14-,19-,20+;/m0./s1 |
| Chemical Name | (R)-[(2S,4S,5R)-5-ethenyl-1-azabicyclo[2.2.2]octan-2-yl]-(6-methoxyquinolin-4-yl)methanol;sulfuric acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | It has been demonstrated that quinidine sulfate, an antiarrhythmic medication, effectively blocks numerous classes of K+ channels in a variety of cell types via influencing ionic currents in the myocardium [1]. Ik peak amplitude decreases in a dose-dependent manner when quinidine sulfate is applied in a bath. It is calculated that at 0 mV, the Kd for Ik blockage is 41 μM [1]. The rate of Ik decay is increased by quinidine sulfate in a dose-dependent manner, and membrane depolarization amplifies this action. Quinidine sulfate also lengthens the half-life of recovery from inactivation and induces a 5 mV hyperpolarizing change in the steady-state inactivation curve. When evaluated at -30 mV, quinidine sulfate had no effect on the beginning of inactivation [1]. |
| ln Vivo | Quinidine sulfate is quickly absorbed; 60 to 90 minutes after oral dosing, the plasma concentration reaches its peak. Other salts, such as polygalacturonate and gluconate, have lower peak concentrations and are absorbed more slowly [2]. About 70–90% of quinidine sulfate is linked to plasma proteins. In the liver, it goes through oxidative metabolism to produce N-oxide, 3-hydroxy, O-desmethyl, and 2'-quinidinone [2]. Rats' metabolism of amphetamine is inhibited by quinidine sulfate. Quinidine sulfate pretreatment led to a large rise in amphetamine excretion to 542% of control levels during 24 and 48 hours, coupled by a significant decrease in parahydroxyamphetamine excretion to 7.2 and 24.1% of vehicle control levels at 24 and 48 hours, respectively. [3]. |
| References |
[1]. Quinidine-induced inhibition of the fast transient outward K+ current in rat melanotrophs. Br J Pharmacol. 1991 Jul;103(3):1807-13. [2]. Class I antiarrhythmic agents: quinidine, procainamide and N-acetylprocainamide, disopyramide. [3]. Quinidine inhibits in vivo metabolism of amphetamine in rats: impact upon correlation between GC/MS and immunoassay findings in rat urine. J Anal Toxicol. 1990 Sep-Oct;14(5):311-7. |
| Additional Infomation |
Quinine Sulfate is the sulfate salt form of the quinidine alkaloid isolate quinine. Quinine has many mechanisms of action, including reduction of oxygen intake and carbohydrate metabolism; disruption of DNA replication and transcription via DNA intercalation; and reduction of the excitability of muscle fibers via alteration of calcium distribution. This agent also inhibits the drug efflux pump P-glycoprotein which is overexpressed in multi-drug resistant tumors and may improve the efficacy of some antineoplastic agents. (NCI04) An alkaloid derived from the bark of the cinchona tree. It is used as an antimalarial drug, and is the active ingredient in extracts of the cinchona that have been used for that purpose since before 1633. Quinine is also a mild antipyretic and analgesic and has been used in common cold preparations for that purpose. It was used commonly and as a bitter and flavoring agent, and is still useful for the treatment of babesiosis. Quinine is also useful in some muscular disorders, especially nocturnal leg cramps and myotonia congenita, because of its direct effects on muscle membrane and sodium channels. The mechanisms of its antimalarial effects are not well understood. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3669 mL | 11.8343 mL | 23.6686 mL | |
| 5 mM | 0.4734 mL | 2.3669 mL | 4.7337 mL | |
| 10 mM | 0.2367 mL | 1.1834 mL | 2.3669 mL |