Pyrvinium is an FDA-approved antihelmintic drug for pinworms, it inhibits WNT pathway signaling. Pyrvinium pamoate (PP), inhibited proliferation of different subtypes of breast cancer cells (luminal: MCF-7, claudin-low: MDA-MB‑231, basal-like: MDA-MB‑468 and Her-2 enriched: SkBr-3) as a novel WNT pathway inhibitor. Additionally, PP was also shown to inhibit self-renewal of breast cancer stem cells (BCSCs) and decrease both CD44+CD24-/low and ALDH-positive BCSCs content in a panel of breast cancer cell lines. Besides, the metastatic potential and expression of EMT markers (such as N-cadherin, vimentin, Snail) were also found suppressed by PP. By using a xenograft model, we next tested the efficacy of PP on tumorigenicity of MDA-MB‑231, one of the most aggressive breast cancer cell lines, and we observed PP significantly delayed tumor growth in vivo. Moreover, in-depth analysis revealed that PP caused inhibition of WNT pathway activity and stemness regulator expression including NANOG, SOX2 and OCT4, which were inherently upregulated in the BCSCs as compared with the bulk of cells within the tumor. Collectively, our findings provide direct evidence for PP serving as a promising high-yield agent targeting BCSCs and cancer heterogeneity. Therefore, strategies combining PP with standard chemotherapy drugs which fail to eliminate the BCSCs hold promise to overcome BCSCs associated treatment resistance and achieve a better therapeutic outcome.
Physicochemical Properties
| Molecular Formula | C75H70N6O6 | |
| Molecular Weight | 1151.39 | |
| Exact Mass | 1150.535 | |
| CAS # | 3546-41-6 | |
| Related CAS # | 110-85-0 (cation);142-88-1 (adipate);3546-41-6 (pamoate); 548-84-5 (Cl); 35648-29-4 (Iodide); | |
| PubChem CID | 54680693 | |
| Appearance | Red to reddish brown solid powder | |
| Density | 1.0104 (rough estimate) | |
| Boiling Point | 829.27°C (rough estimate) | |
| Melting Point | 210-215° (softens at 190°) | |
| Index of Refraction | 1.6400 (estimate) | |
| LogP | 12.338 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 12 | |
| Heavy Atom Count | 87 | |
| Complexity | 1120 | |
| Defined Atom Stereocenter Count | 0 | |
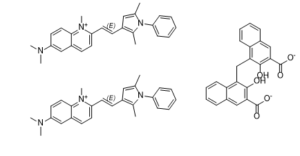
| SMILES | CC1=CC(=C(N1C2=CC=CC=C2)C)/C=C/C3=[N+](C4=C(C=C(C=C4)N(C)C)C=C3)C.CC1=CC(=C(N1C2=CC=CC=C2)C)/C=C/C3=[N+](C4=C(C=C(C=C4)N(C)C)C=C3)C.C1=CC=C2C(=C(C(=CC2=C1)C(=O)O)[O-])CC3=C(C(=CC4=CC=CC=C34)C(=O)O)[O-] |
|
| InChi Key | OOPDAHSJBRZRPH-UHFFFAOYSA-L | |
| InChi Code | InChI=1S/2C26H28N3.C23H16O6/c2*1-19-17-21(20(2)29(19)24-9-7-6-8-10-24)11-13-23-14-12-22-18-25(27(3)4)15-16-26(22)28(23)5;24-20-16(14-7-3-1-5-12(14)9-18(20)22(26)27)11-17-15-8-4-2-6-13(15)10-19(21(17)25)23(28)29/h2*6-18H,1-5H3;1-10,24-25H,11H2,(H,26,27)(H,28,29)/q2*+1;/p-2 | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light.(2). This product is not stable in solution, please use freshly prepared working solution for optimal results. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | MCF-7 (luminal), MDA-MB-231 (claudin-low), MDA-MB-468 (basal-like), and SkBr3 (HER2-OE) cells are all inhibited by pyrvinium pamoate (0-500 nM) in a dose-dependent manner. The MDA-MB-231 cell line's IC50 value is 1170±105.0 nM. Pividin pamoate produces BCSC populations with distinct characteristics by strongly inhibiting BCSC self-renewal and duplication. At the topological level, pividin pamoate dramatically decreased the average expression levels of FZD1, FZD10, WNT1, WNT7B, CTNNB1, MYC, and LRP5. Additionally, ALDH1, CD44, and ABCG2 expression are among the other stemness genes whose expression is successfully rearranged by pyrvinium pamoate[1]. Pyrvinium pamoate exhibits dose-dependent inhibition of the circular waveform (IC50), with a broad range of inhibitory concentrations (0.6 - 65 μM) observed for the WNT signal's circular waveform. While pyrvinium pamoate does not harm CK1, it eventually suppresses Wnt signaling by reducing the messenger RNA (mRNA) of known WNT target genes, such as c-MYC [3]. Cardiovascular heart-forming fibroblasts are susceptible to the amplitude toxicity of pamoic acid (IC50=9.5 nM). Piravinium pamoate's cytotoxic effects on cardiac fibroblasts in the presence of glutamine deficit and yogurt [4]. |
| ln Vitro |
MCF-7 (luminal), MDA-MB-231 (claudin-low), MDA-MB-468 (basal-like), and SkBr3 (HER2-OE) cells are all inhibited by pyrvinium pamoate (0-500 nM) in a dose-dependent manner. The MDA-MB-231 cell line's IC50 value is 1170±105.0 nM. Pividin pamoate produces BCSC populations with distinct characteristics by strongly inhibiting BCSC self-renewal and duplication. At the topological level, pividin pamoate dramatically decreased the average expression levels of FZD1, FZD10, WNT1, WNT7B, CTNNB1, MYC, and LRP5. Additionally, ALDH1, CD44, and ABCG2 expression are among the other stemness genes whose expression is successfully rearranged by pyrvinium pamoate[1]. Pyrvinium pamoate exhibits dose-dependent inhibition of the circular waveform (IC50), with a broad range of inhibitory concentrations (0.6 - 65 μM) observed for the WNT signal's circular waveform. While pyrvinium pamoate does not harm CK1, it eventually suppresses Wnt signaling by reducing the messenger RNA (mRNA) of known WNT target genes, such as c-MYC [3]. Cardiovascular heart-forming fibroblasts are susceptible to the amplitude toxicity of pamoic acid (IC50=9.5 nM). Piravinium pamoate's cytotoxic effects on cardiac fibroblasts in the presence of glutamine deficit and yogurt [4]. Pyrvinium pamoate (PP) exhibited specific cytotoxic effects on cardiac fibroblasts cultured under ischemic conditions (3% oxygen, 300 µM glucose, <100 µM glutamine) with an IC50 of 9.5 nM after 72 hours of treatment. Under normal culture conditions (20-21% oxygen, 25 mM glucose, 4 mM glutamine), PP showed minimal toxicity. [4] The cytotoxic effect of PP (87 nM) on cardiac fibroblasts under ischemic conditions was not rescued by supplementing normal levels of oxygen (20-21%) and glutamine (4 mM). [4] The cytotoxic effect of PP (87 nM) under ischemic conditions was partially rescued by supplementing normal levels of oxygen (20-21%) and glucose (25 mM). [4] The cytotoxic effect of PP (87 nM) under ischemic conditions was completely rescued by supplementing normal levels of glucose (25 mM) and glutamine (4 mM). This demonstrates that the cytotoxicity of PP is specifically dependent on glucose and, to a lesser extent, glutamine deficiency. [4] |
| ln Vivo |
Pyrvinium pamoate (500 nM) significantly reduced tumor volume, tumor size and weight, and severe tumor retardation in xenograft models [1]. In a mouse model of myocardial infarction (permanent left coronary artery ligation), daily oral administration of PP (initiated at 1 day post-ligation) significantly reduced the number of proliferating cells (Ki67-positive) in the infarcted zone at 4 days post-ligation compared to vehicle-treated controls. [4] PP treatment significantly reduced the number of α-smooth muscle actin (αSMA)-positive cardiac myofibroblasts in both the border and infarcted zones at 7 days post-ligation. [4] PP treatment significantly reduced the fibrosis area (as assessed by Masson's trichrome staining) and the hydroxyproline content (a quantitative indicator of mature collagen) in the left ventricle at 14 days post-ligation, without altering the initial infarct size at day 4. [4] PP treatment improved left ventricular systolic function, as indicated by a significantly increased ejection fraction measured by echocardiography at 14 days post-ligation compared to vehicle-treated infarcted mice. [4] PP treatment increased microvascular density (CD31-positive capillaries) in the scarred heart tissue at 14 days post-ligation. [4] |
| Animal Protocol |
For the myocardial infarction mouse model, pyrvinium pamoate (PP) was suspended at a final concentration of 400 µg/ml in a vehicle consisting of 4% dimethyl sulfoxide (DMSO) in saline. Mice were force-fed (intragastrically) with 0.5 ml of this suspension daily. Oral administration began at 1 day after coronary artery ligation (post-ligation) and continued daily until the endpoint at 14 days post-ligation. The control group received the vehicle (4% DMSO in saline) following the same schedule. [4] |
| ADME/Pharmacokinetics |
A previous study cited within the literature indicates that oral administration of PP at the same dosage used in this study resulted in clearance of the drug from the circulation within 12 hours. No further pharmacokinetic parameters (e.g., absorption, distribution, metabolism, excretion, half-life, bioavailability) are provided in this study. [4] |
| References |
[1]. WNT pathway inhibitor pyrvinium pamoate inhibits the self-renewal and metastasis of breast cancer stem cells. Int J Oncol. 2016 Mar;48(3):1175-86. [2]. The impact of pyrvinium pamoate on colon cancer cell viability. Int J Colorectal Dis. 2014 Oct;29(10):1189-98. [3]. Pyrvinium pamoate does not activate protein kinase CK1, but promotes Akt/PKB down-regulation and GSK3 activation. Biochem J. 2013 May 15;452(1):131-7. [4]. An anthelmintic drug, pyrvinium pamoate, thwarts fibrosis and ameliorates myocardial contractile dysfunction in a mouse model of myocardial infarction. PLoS One. 2013 Nov 4;8(11):e79374. |
| Additional Infomation |
Pyrvinium pamoate appears as odorless bright orange or orange-red to brownish red or almost black fluffy powder. Melting point 210-215 °C. Insoluble in water. Tasteless. Pyrvinium pamoate is a naphthoic acid. It has a role as an anticoronaviral agent. Pyrvinium Pamoate is the pamoate salt of pyrvinium, a quinoline-derived cyanine dye and an mitochondrial oxidative phosphorylation (OxPhos) inhibitor, with anthelmintic and potential antineoplastic activities. Upon administration, pyrvinium pamoate may inhibit mitochondrial OxPhos and mitochondrial respiration, decreases mitochondrial function, prevents tumor cell metabolism and deprives tumor cells of energy, thereby preventing tumor cell proliferation and leading to tumor cell death in some types of cancer cells. Mitochondrial OxPhos is overactivated in cancer cells and plays a key role in tumor cell proliferation. Drug resistant tumor cells are very susceptible to decreased mitochondrial OxPhos as they cannot easily compensate for the decrease in mitochondrial function by increasing glycolysis. See also: Pyrvinium (has active moiety). Pyrvinium pamoate is an FDA-approved anthelmintic drug. [4] The proposed mechanism in this study involves compromising the survival of cardiac fibroblasts specifically under conditions of glucose (and glutamine) deficiency, which mimics the ischemic microenvironment following myocardial infarction. This effect is similar to its previously reported activity against glucose-starved cancer cells. [4] The study suggests a novel therapeutic application of PP for reducing fibrosis and improving function in scarred hearts after myocardial infarction, and potentially in other fibrotic organs/tissues. [4] The timing of drug administration is critical. In this study, beneficial effects were observed when PP administration started one day after coronary ligation (after most cardiomyocyte death had occurred), rather than at the time of ligation. [4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8685 mL | 4.3426 mL | 8.6852 mL | |
| 5 mM | 0.1737 mL | 0.8685 mL | 1.7370 mL | |
| 10 mM | 0.0869 mL | 0.4343 mL | 0.8685 mL |