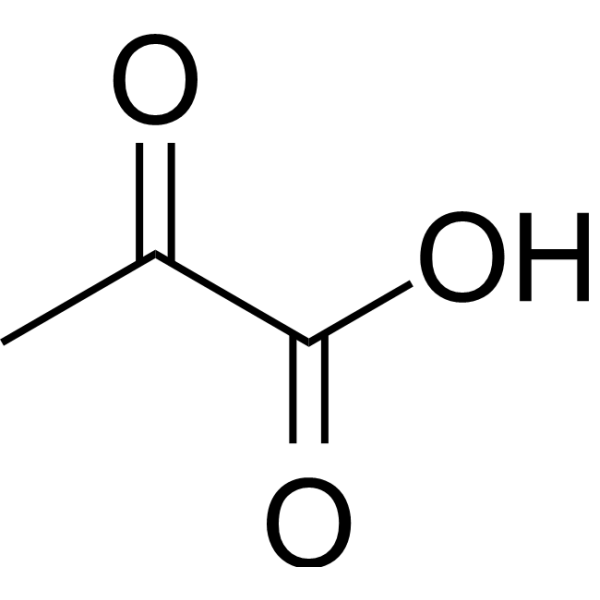
Physicochemical Properties
| Molecular Formula | C3H4O3 |
| Molecular Weight | 88.0621 |
| Exact Mass | 88.016 |
| CAS # | 127-17-3 |
| PubChem CID | 1060 |
| Appearance | Colorless to light yellow <11°C solid powder,>12°C liquid |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 165.0±0.0 °C at 760 mmHg |
| Melting Point | 11-12 °C(lit.) |
| Flash Point | 54.3±15.2 °C |
| Vapour Pressure | 1.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.417 |
| LogP | -1.24 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 6 |
| Complexity | 84 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O([H])C(C(C([H])([H])[H])=O)=O |
| InChi Key | LCTONWCANYUPML-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C3H4O3/c1-2(4)3(5)6/h1H3,(H,5,6) |
| Chemical Name | 2-oxopropanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Pyruvate is absorbed from the gastrointestinal tract from whence it is transported to the liver via the portal circulation. Metabolism / Metabolites In the liver, pyruvate is metabolized via several pathways. In the liver, pyruvate is metabolized via several pathways. |
| Additional Infomation |
Pyruvic acid is a 2-oxo monocarboxylic acid that is the 2-keto derivative of propionic acid. It is a metabolite obtained during glycolysis. It has a role as a fundamental metabolite and a cofactor. It is functionally related to a propionic acid. It is a conjugate acid of a pyruvate. An intermediate compound in the metabolism of carbohydrates, proteins, and fats. In thiamine deficiency, its oxidation is retarded and it accumulates in the tissues, especially in nervous structures. (From Stedman, 26th ed) Pyruvic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). Pyruvic acid has been reported in Cnidium monnieri, Drosophila melanogaster, and other organisms with data available. Pyruvic acid is an intermediate compound in the metabolism of carbohydrates, proteins, and fats. In thiamine deficiency, its oxidation is retarded and it accumulates in the tissues, especially in nervous structures. (From Stedman, 26th ed.) Biological Source: Intermediate in primary metabolism including fermentation processes. Present in muscle in redox equilibrium with Lactic acid. A common constituent, as a chiral cyclic acetal linked to saccharide residues, of bacterial polysaccharides. Isolated from cane sugar fermentation broth and peppermint. Constituent of Bauhinia purpurea, Cicer arietinum (chickpea), Delonix regia, Pisum sativum (pea) and Trigonella caerulea (sweet trefoil) Use/Importance: Reagent for regeneration of carbonyl compdounds from semicarbazones, phenylhydrazones and oximes. Pyruvic acid is a metabolite found in or produced by Saccharomyces cerevisiae. An intermediate compound in the metabolism of carbohydrates, proteins, and fats. In thiamine deficiency, its oxidation is retarded and it accumulates in the tissues, especially in nervous structures. (From Stedman, 26th ed) Drug Indication For nutritional supplementation, also for treating dietary shortage or imbalance Mechanism of Action Pyruvate serves as a biological fuel by being converted to acetyl coenzyme A, which enters the tricarboxylic acid or Krebs cycle where it is metabolized to produce ATP aerobically. Energy can also be obtained anaerobically from pyruvate via its conversion to lactate. Pyruvate injections or perfusions increase contractile function of hearts when metabolizing glucose or fatty acids. This inotropic effect is striking in hearts stunned by ischemia/reperfusion. The inotropic effect of pyruvate requires intracoronary infusion. Among possible mechanisms for this effect are increased generation of ATP and an increase in ATP phosphorylation potential. Another is activation of pyruvate dehydrogenase, promoting its own oxidation by inhibiting pyruvate dehydrogenase kinase. Pyruvate dehydrogenase is inactivated in ischemia myocardium. Yet another is reduction of cytosolic inorganic phosphate concentration. Pyruvate, as an antioxidant, is known to scavenge such reactive oxygen species as hydrogen peroxide and lipid peroxides. Indirectly, supraphysiological levels of pyruvate may increase cellular reduced glutathione. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~1135.59 mM) DMSO : ≥ 50 mg/mL (~567.79 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.63 mg/mL (29.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 26.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (28.39 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (28.39 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (1135.59 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 11.3559 mL | 56.7795 mL | 113.5589 mL | |
| 5 mM | 2.2712 mL | 11.3559 mL | 22.7118 mL | |
| 10 mM | 1.1356 mL | 5.6779 mL | 11.3559 mL |