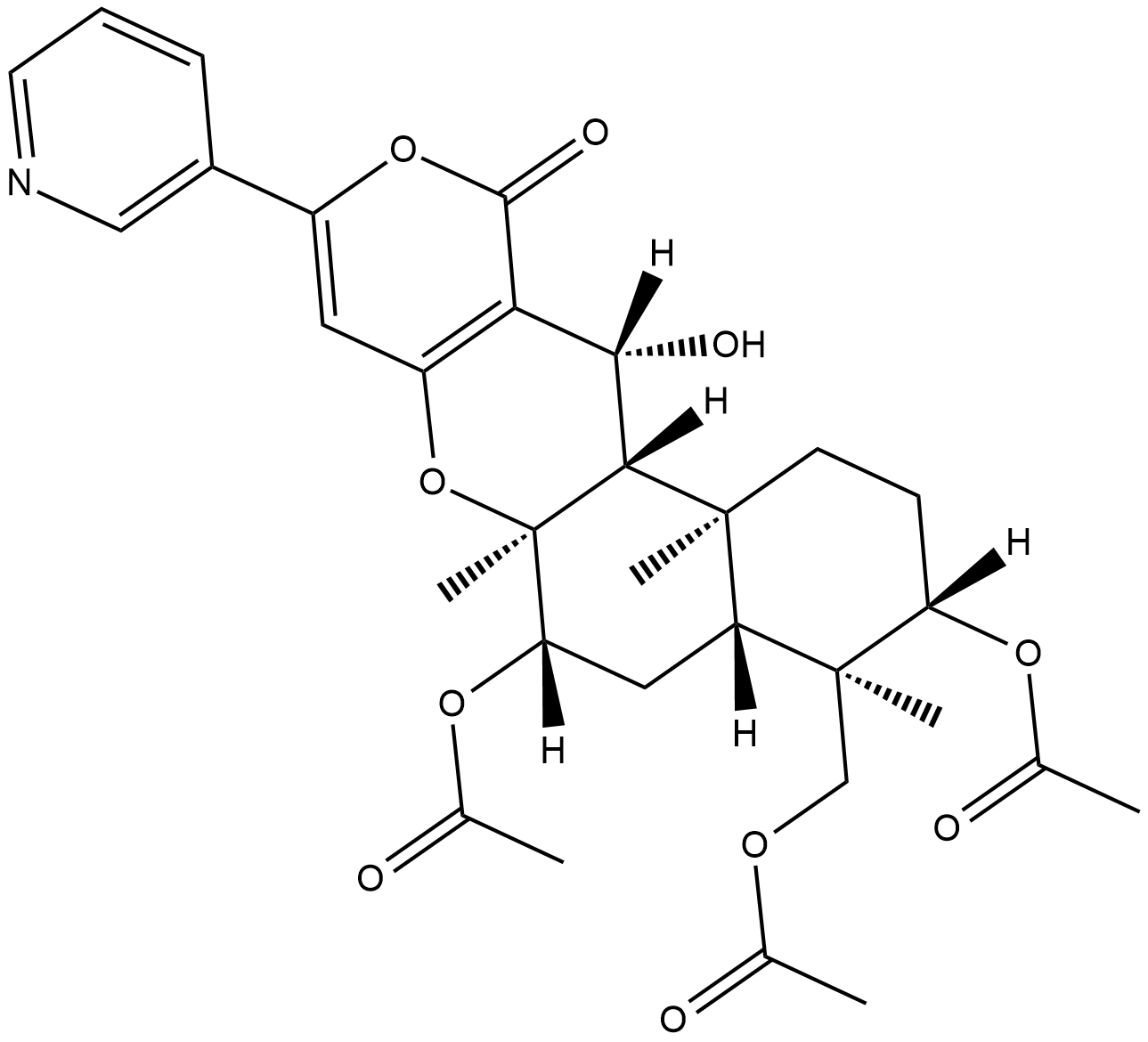
Pyripyropene A is a potent and specific inhibitor of sterol O-acyltransferase 2 (SOAT2) or acyl-coenzyme A:cholesterol acyltransferase 2 (ACAT2) with the potential to be used for atherosclerosis. ACAT is a key enzyme for cholesteryl ester accumulation in atherogenesis, lipoprotein formation in liver, and cholesterol absorption from intestines, all of which are events that contribute to the atherosclerotic process.
Physicochemical Properties
| Molecular Formula | C31H37NO10 |
| Molecular Weight | 583.62618 |
| Exact Mass | 583.241 |
| CAS # | 147444-03-9 |
| PubChem CID | 11828024 |
| Appearance | White to light yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 690.8±55.0 °C at 760 mmHg |
| Melting Point | 153-154°C (lit.) |
| Flash Point | 371.6±31.5 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.587 |
| LogP | 2.68 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 42 |
| Complexity | 1230 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | CC(=O)OC[C@@]1([C@H](CC[C@]2([C@H]1C[C@@H]([C@@]3([C@@H]2[C@H](C4=C(O3)C=C(OC4=O)C5=CN=CC=C5)O)C)OC(=O)C)C)OC(=O)C)C |
| InChi Key | PMMQOFWSZRQWEV-RVTXXDJVSA-N |
| InChi Code | InChI=1S/C31H37NO10/c1-16(33)38-15-30(5)22-13-24(40-18(3)35)31(6)27(29(22,4)10-9-23(30)39-17(2)34)26(36)25-21(42-31)12-20(41-28(25)37)19-8-7-11-32-14-19/h7-8,11-12,14,22-24,26-27,36H,9-10,13,15H2,1-6H3/t22-,23+,24+,26+,27-,29+,30+,31-/m1/s1 |
| Chemical Name | [(1S,2S,5S,6R,7R,9S,10S,18R)-5,9-diacetyloxy-18-hydroxy-2,6,10-trimethyl-16-oxo-14-pyridin-3-yl-11,15-dioxatetracyclo[8.8.0.02,7.012,17]octadeca-12(17),13-dien-6-yl]methyl acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | With an IC50 value of 1.8 μM, Pyripyropene A (0-100 μM; 72) has anti-proliferative activity against HUVEC [1]. Pyripyropene A (10 μM; 24) suppresses the induction of VEGF (20 ng/mL) in a dose-dependent manner. Pyripyropene A does not impede the proliferation of Neuro2A, K562, or KB3-1 cells [1]. |
| ln Vivo | Pyripyropene A (10-50 mg/kg daily; face; 12 weeks) Reduces cholesterol, very low-density lipoprotein (VLDL) and low-density lipoprotein (LDL) levels and liver cholesterol content in the lipoprotein E-loaded gene Pyripyropene A treated CLOCK showed area reduction of enzymatic atherosclerotic plaques in the aorta and heart [3]. Pyripyropene A Multilamellar phospholipidyl-CoA: multilamellar phospholipidyl transfer 2 (ACAT2) active internal [3] The half-life (t1/2) of Pyripyropene A is 0.693/λ, where λ represents the logarithmic component of the concentration-time curve [4 ]. |
| Cell Assay |
Cell proliferation assay[1] Cell Types: HUVEC Tested Concentrations: 0-100 µM Incubation Duration: HUVEC migration and tube formation[1]. 72 hour Experimental Results: demonstrated anti-proliferative activity against HUVEC with IC50 value of 1.8 µM. |
| Animal Protocol |
Animal/Disease Models: Male C57BL/6 mice[2] Doses: 0 mg/kg, 1]. mg/kg, 10 mg/kg, 50 mg/kg, 100 mg/kg Doses: Oral; daily; for 12 weeks Experimental Results: diminished areas of atherosclerotic disease in the aorta and heart. Animal/Disease Models: 9weeks old male ICR mice (pharmacokinetic/PK/PK analysis) [4] Doses: 5 mg/kg, 10 mg/kg Route of Administration: Oral Experimental Results:t1/2 = 0.693/λ |
| References |
[1]. Pyripyropenes, fungal sesquiterpenes conjugated with alpha-pyrone and pyridine moieties, exhibits anti-angiogenic activity against human umbilical vein endothelial cells. Biol Pharm Bull. 2009 Jul;32(7):1261-5. [2]. Design and Synthesis of A-Ring Simplified Pyripyropene A Analogues as Potent and Selective Synthetic SOAT2 Inhibitors. ChemMedChem. 2018 Mar 6;13(5):411-421. [3]. Pyripyropene A, an acyl-coenzyme A:cholesterol acyltransferase 2-selective inhibitor, attenuates hypercholesterolemia and atherosclerosis in murine models of hyperlipidemia. Arterioscler Thromb Vasc Biol. 2011 May;31(5):1108-15. [4]. Determination of Penicillium griseofulvum-oriented pyripyropene A, a selective inhibitor of acyl-coenzyme A:cholesterol acyltransferase 2, in mouse plasma using liquid chromatography-tandem mass spectrometry and its application to pharmacokinetic studies. Biomed Chromatogr. 2019 Feb;33(2):e4388. |
| Additional Infomation |
Pyripyropene A is a sesquiterpenoid that consists of (3S,4R,4aR,6S,6aS,12R,12aS,12bS)-4-(acetoxymethyl)-12-hydroxy-4,6a,12b-trimethyl-11-oxo-9-(pyridin-3-yl)-1,3,4,4a,5,6,6a,12,12a,12b-decahydro-2H,11H-benzo[f]pyrano[4,3-b]chromene-3,6-diol in which the hydrogens of the 3- and 6-hydroxy functions are substituted by acetyl groups. It has a role as a metabolite and an acyl-CoA:cholesterol acyltransferase 2 inhibitor. It is a sesquiterpenoid and an organic heterotetracyclic compound. Pyripyropene A has been reported in Aspergillus fischeri, Aspergillus fumigatus, and Aspergillus sydowii with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7134 mL | 8.5671 mL | 17.1341 mL | |
| 5 mM | 0.3427 mL | 1.7134 mL | 3.4268 mL | |
| 10 mM | 0.1713 mL | 0.8567 mL | 1.7134 mL |