Physicochemical Properties
| Molecular Formula | C24H32O6 |
| Molecular Weight | 416.5073 |
| Exact Mass | 416.219 |
| CAS # | 17008-69-4 |
| PubChem CID | 204810 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 635.8±55.0 °C at 760 mmHg |
| Flash Point | 218.7±25.0 °C |
| Vapour Pressure | 0.0±4.2 mmHg at 25°C |
| Index of Refraction | 1.622 |
| LogP | 0.71 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 30 |
| Complexity | 847 |
| Defined Atom Stereocenter Count | 9 |
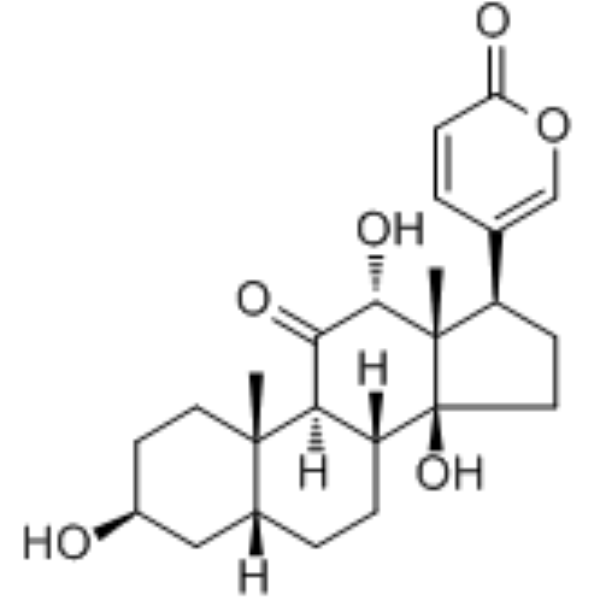
| SMILES | C[C@]12CC[C@@H](C[C@H]1CC[C@@H]3[C@@H]2C(=O)[C@@H]([C@]4([C@@]3(CC[C@@H]4C5=COC(=O)C=C5)O)C)O)O |
| InChi Key | SOGONHOGEFLVPE-BHZHDSHXSA-N |
| InChi Code | InChI=1S/C24H32O6/c1-22-9-7-15(25)11-14(22)4-5-17-19(22)20(27)21(28)23(2)16(8-10-24(17,23)29)13-3-6-18(26)30-12-13/h3,6,12,14-17,19,21,25,28-29H,4-5,7-11H2,1-2H3/t14-,15+,16-,17-,19-,21+,22+,23+,24+/m1/s1 |
| Chemical Name | 5-[(3S,5R,8R,9S,10S,12R,13S,14S,17R)-3,12,14-trihydroxy-10,13-dimethyl-11-oxo-2,3,4,5,6,7,8,9,12,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]pyran-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
ψ-Bufarenogin suppresses the proliferation of seven hepatocellular carcinoma (HCC) cell lines in a dose-dependent manner. The HCC cell line SMMC-7721 was among the most sensitive. [1] ψ-Bufarenogin inhibits cell cycle progression in HCC cells, leading to a decreased G1/S transition and a marked G2/M arrest. This was associated with reduced cyclin E expression and accumulation of cyclin B1. [1] ψ-Bufarenogin (50 nM) facilitates apoptosis in hepatoma cells after 48 hours of treatment. This pro-apoptotic effect is mediated through the downregulation of the anti-apoptotic protein Mcl-1, while Bax and Bcl-2 levels remain unchanged. Reconstitution of Mcl-1 expression attenuates ψ-Bufarenogin-triggered apoptosis. [1] ψ-Bufarenogin represses the expansion of hepatic tumor-initiating cells (T-ICs). It inhibits the expression of T-IC biomarkers (EpCAM, CD133, CD90), significantly reduces spheroid formation in HCC cell lines and primary patient cells, and diminishes the proportion of T-ICs as shown by limiting dilution assays. This effect is associated with the downregulation of the stemness-associated transcription factor Sox2. [1] ψ-Bufarenogin exhibits a synergistic effect with cisplatin in suppressing spheroid formation and in triggering apoptosis of patient-derived primary hepatoma cells. [1] ψ-Bufarenogin inhibits the activation of the Raf/MEK/ERK signaling pathway and the PI3-K/Akt cascade in HCC cells stimulated by EGF, in a dose-dependent manner. It slightly inhibits JNK phosphorylation but does not affect p38 activation. The inhibition of the PI3-K/Akt pathway is involved in the ψ-Bufarenogin-mediated reduction of Mcl-1 and Sox2. [1] ψ-Bufarenogin inhibits the auto-phosphorylation of EGFR and c-Met in HCC cells stimulated by EGF or HGF, respectively, and subsequently suppresses their downstream MEK/ERK and PI3-K/Akt signaling. [1] ψ-Bufarenogin exhibits potent cytotoxicity against hepatoma cells but has modest activity in normal hepatocytes. [1] Molecular docking studies predict that ψ-Bufarenogin binds to the kinase domains of EGFR and c-Met, forming specific hydrogen bond interactions. [1] |
| ln Vivo |
Intravenous (i.v.) administration of ψ-Bufarenogin (0.2 mg/kg and 0.4 mg/kg, once daily for 16 days) significantly inhibits the growth of SMMC-7721 cell-derived xenograft tumors in nude mice, reducing tumor volume by up to 60% compared to the control group, without causing obvious side effects as assessed by H&E staining of vital organs. [1] Intratumoral injection of ψ-Bufarenogin (0.5 mg/kg and 1.0 mg/kg, every other day) also significantly inhibits xenografted HCC growth in mice. [1] Intratumoral injection of ψ-Bufarenogin (1.0 mg/kg, every other day for 24 days) inhibits the growth of a patient-derived xenograft (PDX) HCC model characterized by high expression of EGFR and c-Met. [1] Tumors from ψ-Bufarenogin-treated mice show lower Ki67 antigen expression (indicating reduced proliferation), increased TUNEL-positive cells (indicating increased apoptosis), and reduced phosphorylation of MEK and Akt compared to controls. [1] |
| Enzyme Assay |
PI3-K Kinase Activity Assay: The PI3-K enzyme was immunoprecipitated from EGF-stimulated SMMC-7721 cells pretreated with or without ψ-Bufarenogin. The kinase activity in the immunoprecipitates was measured using a competitive enzyme-linked immunosorbent assay kit according to the manufacturer's instructions. The assay demonstrated that ψ-Bufarenogin significantly represses the kinase activity of PI3-K. [1] c-MET Tyrosine Phosphorylation Assay: SMMC-7721 cells preincubated with ψ-Bufarenogin for 4 hours were stimulated with a mitogen cocktail containing HGF. Cell lysates were extracted, and tyrosine phosphorylation of c-MET was detected using a bead-based flow cytometric assay according to the manufacturer's protocol. [1] |
| Cell Assay |
Cell Viability/Proliferation Assay (CCK-8): HCC cells were exposed to ψ-Bufarenogin at indicated concentrations for 48 hours. Cell viability was measured using a CCK-8 assay, and the inhibition rate was calculated. IC50 values were determined for different cell lines. [1] Cell Cycle Analysis: Hepatoma cells treated with ψ-Bufarenogin were collected, fixed, stained with propidium iodide, and analyzed by flow cytometry to determine cell cycle distribution. [1] Apoptosis Assay: Apoptosis in hepatoma cells triggered by ψ-Bufarenogin was examined using a Vybrant Apoptosis Kit and analyzed by flow cytometry. [1] Anchorage-Independent Growth Assay (Soft Agar): Hepatoma cells were plated in culture dishes with medium containing Matrigel in the presence or absence of ψ-Bufarenogin. After 2 weeks, multicellular colonies were counted under a microscope. [1] Spheroid Formation Assay: Primary hepatoma cells from patients were plated in ultra-low attachment culture dishes and treated with ψ-Bufarenogin. After one week, the number of spheroids formed was counted under a microscope. [1] Limiting Dilution Assay: Hepatoma cells were seeded into 96-well ultra-low attachment plates. After 7 days, spheroid formation was assessed visually. The frequency of wells without colonies was used to estimate the proportion of stem cells using Poisson distribution statistics. [1] Western Blot Analysis: Cell lysates from treated hepatoma cells or xenograft tumor tissues were subjected to SDS-PAGE, transferred to membranes, and probed with specific primary antibodies (e.g., against p-EGFR, p-c-MET, p-MEK, p-ERK, p-Akt, Mcl-1, Sox2, cyclins). Signals were detected using IRDye-conjugated secondary antibodies. [1] Real-time PCR: Total RNA was extracted from treated cells, reverse transcribed, and amplified using gene-specific primers (e.g., for cyclin E, cyclin B1, Sox2) on a real-time PCR system to measure relative mRNA expression levels. [1] |
| Animal Protocol |
Xenograft Model (Cell Line-Derived): Fragments of SMMC-7721 xenografts were implanted subcutaneously into the flanks of nude mice. When tumors reached approximately 300 mm³, mice were randomly assigned to treatment or control groups. ψ-Bufarenogin was dissolved in DMSO and diluted with normal saline. For intravenous administration, ψ-Bufarenogin (0.2 or 0.4 mg/kg) or vehicle was injected once daily for 16 days. For intratumoral injection, ψ-Bufarenogin (0.5 or 1.0 mg/kg) or vehicle was injected every other day. Tumor dimensions were measured periodically with calipers to calculate volume. [1] Patient-Derived Xenograft (PDX) Model: HCC tissue from a patient with high EGFR/c-MET expression was implanted into mice. When tumors were established, mice received intratumoral injections of ψ-Bufarenogin (1.0 mg/kg) or vehicle every other day for 24 days. Tumor growth was monitored as above. [1] Tissue Analysis: At the endpoint, tumors and vital organs were harvested. Parts were fixed for H&E staining, immunohistochemistry (e.g., for Ki-67, p-MEK), and TUNEL assay. Other parts were snap-frozen for protein extraction and Western blot analysis. [1] |
| Toxicity/Toxicokinetics | In the xenograft mouse models, intravenous administration of ψ-Bufarenogin at doses of 0.2 mg/kg and 0.4 mg/kg daily for 16 days did not cause obvious side effects. Histopathological examination (H&E staining) of vital organs (heart, liver, spleen, lung, kidney) from treated mice showed no notable abnormalities compared to the control group. [1] |
| References |
[1]. ψ-Bufarenogin, a novel anti-tumor compound, suppresses liver cancer growth by inhibiting receptor tyrosine kinase-mediated signaling. Oncotarget. 2015 May 10; 6(13): 11627–11639. |
| Additional Infomation |
ψ-Bufarenogin (molecular formula C₂₄H₃₂O₆) is a bufadienolide compound isolated and purified from the skin of the toad Bufo bufo gargarizans Cantor using a two-dimensional reversed-phase liquid chromatography/hydrophilic interaction chromatography system. [1] It is an epimer of Bufarenogin at the C-12 position and is reported to be nearly 100-fold more active against cancer cells than Bufarenogin. [1] The proposed mechanism of action involves dual inhibition of EGFR and c-MET receptor tyrosine kinases, leading to the suppression of their downstream Raf/MEK/ERK and PI3-K/Akt signaling pathways. This results in inhibited cell proliferation (via cell cycle arrest), promoted apoptosis (via Mcl-1 downregulation), and repressed cancer stem cell expansion (via Sox2 downregulation). [1] ψ-Bufarenogin demonstrates synergistic effects with conventional chemotherapeutics like cisplatin. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~240.09 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4009 mL | 12.0045 mL | 24.0090 mL | |
| 5 mM | 0.4802 mL | 2.4009 mL | 4.8018 mL | |
| 10 mM | 0.2401 mL | 1.2005 mL | 2.4009 mL |