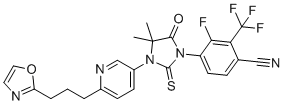
Proxalutamide (GT0918; GT-0918) is a novel, oral and potent androgen receptor/AR antagonist or nonsteroidal antiandrogen (NSAA). It is being investigated in phase 1/2/3 clinical trials for treating prostate cancer, breast cancer and COVID-19 pandemic. It was approved in Paraguay for the treatment of COVID-19 in July 2021, but has not been approved at this time in other countries.
Physicochemical Properties
| Molecular Formula | C24H19F4N5O2S |
| Molecular Weight | 517.4986 |
| Exact Mass | 517.119 |
| Elemental Analysis | C, 55.70; H, 3.70; F, 14.68; N, 13.53; O, 6.18; S, 6.20 |
| CAS # | 1398046-21-3 |
| PubChem CID | 60194102 |
| Appearance | Solid powder |
| LogP | 4.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 36 |
| Complexity | 894 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | KCBJGVDOSBKVKP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H19F4N5O2S/c1-23(2)21(34)32(17-9-6-14(12-29)19(20(17)25)24(26,27)28)22(36)33(23)16-8-7-15(31-13-16)4-3-5-18-30-10-11-35-18/h6-11,13H,3-5H2,1-2H3 |
| Chemical Name | 4-(4,4-dimethyl-3-(6-(3-(oxazol-2-yl)propyl)pyridin-3-yl)-5-oxo-2-thioxoimidazolidin-1-yl)-3-fluoro-2-(trifluoromethyl)benzonitrile |
| Synonyms | Proxalutamide GT 0918 GT0918GT-0918 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
|
|
| ln Vitro |
|
|
| ln Vivo |
|
|
| Animal Protocol |
|
|
| References |
[1].Abstract 614: Proxalutamide (GT0918), a potent androgen receptor pathway inhibitor. Cancer Research. AACR Annual Meeting 2014; April 5-9, 2014; San Diego, CA. [2].Microenvironmental pH-modified solid dispersions to enhance the dissolution and bioavailability of poorly water-soluble weakly basic GT0918, a developing anti-prostate cancer drug: preparation, characterization and evaluation in vivo. Int J Pharm. 2014 Nov 20;475(1-2):97-109. [3].Proxalutamide Significantly Accelerates Viral Clearance and Reduces Time to Clinical Remission in Patients with Mild to Moderate COVID-19: Results from a Randomized, Double-Blinded, Placebo-Controlled Trial. Cureus. 2021 Feb; 13(2): e13492. [4].Quantitative determination of proxalutamide in rat plasma and tissues using liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom. 2021 Feb 15;35(3):e9003. [5].Metabolomic profiling to evaluate the efficacy of proxalutamide, a novel androgen receptor antagonist, in prostate cancer cells. Invest New Drugs. 2020 Oct;38(5):1292-1302. |
|
| Additional Infomation |
Proxalutamide is under investigation in clinical trial NCT03899467 (The Safety and Tolerability of Proxalutamide (GT0918) in Subjects With Metastatic Castrate Resistant Prostate Cancer). Proxalutamide is an orally bioavailable androgen receptor (AR) antagonist with potential antineoplastic activity. Upon oral administration, proxalutamide binds to AR in target tissues, inhibits androgen-induced receptor activation, and facilitates the formation of inactive complexes that cannot translocate to the nucleus. This prevents binding to and transcription of AR-responsive genes that regulate prostate cancer cell proliferation. In addition, proxalutamide induces AR downregulation, thereby further preventing AR-mediated signaling. This ultimately leads to an inhibition of growth in AR-expressing prostate cancer cells. AR is overexpressed in prostate cancer and plays a key role in prostate cancer cell proliferation. Mechanism of Action Proxalutamide is an antagonist at the androgen receptor which leads to inhibition of androgen-induced receptor activation and results in formation of inactive complexes that are not able to translocate to the nucleus. It also induces androgen receptor downregulation, furthering adrogen-induced receptor activation. This drug is being investigated for its potential antineoplastic activity and is being investigated specifically against prostate cancer and COVID-19. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9324 mL | 9.6618 mL | 19.3237 mL | |
| 5 mM | 0.3865 mL | 1.9324 mL | 3.8647 mL | |
| 10 mM | 0.1932 mL | 0.9662 mL | 1.9324 mL |