Physicochemical Properties
| Molecular Formula | C7H6O3 |
| Molecular Weight | 138.1207 |
| Exact Mass | 138.031 |
| CAS # | 139-85-5 |
| PubChem CID | 8768 |
| Appearance | Light yellow to brown solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 295.4±20.0 °C at 760 mmHg |
| Melting Point | 150-157 °C(lit.) |
| Flash Point | 146.7±18.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.674 |
| LogP | 1.14 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 10 |
| Complexity | 124 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | IBGBGRVKPALMCQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C7H6O3/c8-4-5-1-2-6(9)7(10)3-5/h1-4,9-10H |
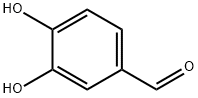
| Chemical Name | 3,4-dihydroxybenzaldehyde |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Protonacladehyde (PCA) treatment of MCF-7 cells at 50, 100 μM for 24 and 48 hours significantly decreased cell proliferation by 11% and 20% in the 24 hour period and by 22% and 27% in the 48 hour period, respectively. [2]. MCF-7 cells treated with 50 μM and 100 μM of protocatechuic aldehyde for 24 hours showed a 1.9- and 2.6-fold increase, respectively. PCA suppresses the enzymatic activity of HDAC and inhibits estrogen receptor (ER) active (MCF-7) mammary gland protocatechualdehyde (0, 100, and 200 μM, in HCT116 and SW480 cells, for 48 hours). The minimum inhibitory concentration (MIC) of Protocatechualdehyde (PCA) against Ralstonia solanacearum was 20 µg/mL, and the minimum bactericidal concentration (MBC) was 40 µg/mL[1]. In growth curve assays, PCA at concentrations of 30 and 40 µg/mL significantly inhibited the growth of R. solanacearum, with 40 µg/mL nearly completely stopping growth after 26 hours of incubation[1]. Scanning electron microscopy (SEM) revealed that PCA treatment (30 and 40 µg/mL) damaged the surface structure and cellular integrity of R. solanacearum cells, causing them to become longer and narrower compared to untreated cells[1]. PCA inhibited biofilm formation by R. solanacearum in a concentration-dependent manner; treatment with 30 µg/mL and 40 µg/mL PCA significantly reduced biofilm formation by 38.56%/37.27% and 48.11%/38.90% at 24 and 36 hours, respectively, compared to the control[1]. PCA significantly inhibited the swarming motility of R. solanacearum in a dose-dependent manner at concentrations ranging from 10 to 40 µg/mL[1]. |
| ln Vivo |
In a greenhouse pot experiment, soil drench application of Protocatechualdehyde (PCA) at 40 µg/mL effectively reduced the incidence of bacterial wilt on tobacco plants inoculated with R. solanacearum[1]. The control efficacy of PCA treatment reached 92.01% at nine days post-inoculation, which was significantly higher than the 47.31% efficacy achieved by streptomycin (40 µg/mL) treatment at the same time point[1]. The disease index for PCA-treated plants was 18.06 and 48.82 at 15 and 19 days post-inoculation, respectively, significantly lower than the control group (68.06 and 97.22) and comparable to or better than the streptomycin treatment group (51.22 and 78.65)[1]. |
| Cell Assay |
Cell proliferation assay [2] Cell Types: Human breast cancer cells (MCF-7 and MDA-MB-231) Tested Concentrations: 0, 5, 10, 25, PCA treatment was observed to inhibit HDAC activity in a dose-dependent manner [3]. , 50 and 100 μM Incubation Duration: 24, 48 hrs (hours) Experimental Results: Inhibition of MCF-7 cell growth. Apoptosis analysis [2] Cell Types: Human breast cancer cells (MCF-7 and MDA-MB-231) Tested Concentrations: 0, 5, 10, 25, 50 and 100 μM Incubation Duration: 24, 48 hrs (hours) Experimental Results: Apoptosis Increased apoptosis of MCF-7 cells. MIC/MBC Determination: The MIC and MBC of PCA against R. solanacearum were determined using the agar dilution method[1]. Bacterial suspensions were spread onto agar plates containing serial dilutions of PCA (10, 20, 30, 40 µg/mL)[1]. Plates were incubated at 30°C, and the MIC (no visible growth after 48 h) and MBC (no visible growth after 96 h) were recorded[1]. Antimicrobial Assay (Growth Curve): The antibacterial activity was evaluated by monitoring growth curves in liquid culture[1]. PCA was added to broth at final concentrations of 10, 20, 30, or 40 µg/mL, inoculated with R. solanacearum, and incubated with shaking at 30°C[1]. Optical density at 600 nm was measured at 2-hour intervals for 36 hours[1]. Bacterial Morphology (SEM): R. solanacearum cells in logarithmic phase were treated with PCA (30 or 40 µg/mL) for 12 hours with shaking[1]. Cells were collected, washed, fixed with glutaraldehyde, dehydrated through a graded ethanol series, and finally suspended in tert-butanol before being mounted and gold-coated for SEM observation[1]. Biofilm Formation Assay: Biofilm formation was quantified using a crystal violet staining method in 96-well plates[1]. R. solanacearum cultures containing different concentrations of PCA were added to wells and incubated statically at 30°C for 12, 24, or 36 hours[1]. After washing, biofilms were stained with crystal violet, dissolved in ethanol, and the absorbance was measured at 490 nm[1]. Swarming Motility Assay: Swarming motility was assessed on semisolid agar (0.35%) plates containing different concentrations of PCA[1]. Bacterial suspensions were spot-inoculated at the center of the plates[1]. The diameter of the migration zone was measured in two perpendicular directions after incubation at 28°C for 24 and 48 hours[1]. |
| Animal Protocol |
Greenhouse Efficacy Trial: Tobacco plants were inoculated by applying 5 mL of a R. solanacearum suspension to the rhizosphere[1]. Two days later, plants were treated by irrigating the roots with 10 mL of a PCA solution (40 µg/mL) or a streptomycin solution (40 µg/mL, positive control)[1]. Control plants received a water and solvent mixture[1]. Plants were grown in a greenhouse at 30°C and 85-90% relative humidity[1]. Disease incidence was assessed every two days from 7 to 19 days post-inoculation using a 0-4 disease rating scale[1]. |
| Toxicity/Toxicokinetics |
The solvent dimethyl sulfoxide (DMSO) used to dissolve PCA had no effect on the growth of R. solanacearum at the tested concentrations in control experiments[1]. No phytotoxicity or adverse effects on tobacco plants were reported from the greenhouse experiment following root irrigation with the PCA solution[1]. The study mentions that botanical compounds like PCA generally have advantages such as being easily decomposed, not significantly toxic to vertebrates and host plants, and causing less environmental pollution[1]. |
| References |
[1]. Evaluation of the Antibacterial Effects and Mechanism of Action of Protocatechualdehyde against Ralstonia solanacearum. Molecules. 2016 Jun 9;21(6). [2]. Anticancer activity of protocatechualdehyde in human breast cancer cells. J Med Food. 2014 Aug;17(8):842-8. [3]. Protocatechualdehyde possesses anti-cancer activity through downregulating cyclin D1 and HDAC2 in human colorectal cancer cells. Biochem Biophys Res Commun. 2013 Jan 4;430(1):381-6. |
| Additional Infomation |
3,4-dihydroxybenzaldehyde is a dihydroxybenzaldehyde. Also known as protocatechuic aldehyde, protocatechualdehyde is a naturally-occuring phenolic aldehyde that is found in barley, green cavendish bananas, grapevine leaves and root of the herb S. miltiorrhiza. Protocatechualdehyde possesses antiproliferative and pro-apoptotic properties against human breast cancer cells and colorectal cancer cells by reducing the expression of pro-oncogenes β-catenin and cyclin D1. 3,4-Dihydroxybenzaldehyde has been reported in Salvia miltiorrhiza, Hymenochaete xerantica, and other organisms with data available. See also: Black Cohosh (part of). Protocatechualdehyde (PCA) is a natural polyphenolic compound (phenolic aldehyde) isolated from the roots of Salvia miltiorrhiza[1]. The study suggests that the antibacterial mechanism of PCA against R. solanacearum involves damaging the bacterial cell structure and inhibiting biofilm formation and swarming motility[1]. This is reported as the first study concerning the antibacterial effects of PCA on plant-pathogenic bacteria[1]. PCA is proposed as a potential environmentally friendly antibacterial agent for controlling bacterial wilt caused by R. solanacearum[1]. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~362.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (18.10 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (18.10 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (18.10 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.2401 mL | 36.2004 mL | 72.4008 mL | |
| 5 mM | 1.4480 mL | 7.2401 mL | 14.4802 mL | |
| 10 mM | 0.7240 mL | 3.6200 mL | 7.2401 mL |