Physicochemical Properties
| Molecular Formula | C22H30O6 |
| Molecular Weight | 390.4700 |
| Exact Mass | 390.204 |
| CAS # | 60857-08-1 |
| PubChem CID | 454217 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 550.5±50.0 °C at 760 mmHg |
| Melting Point | 216-219℃ |
| Flash Point | 188.7±23.6 °C |
| Vapour Pressure | 0.0±3.4 mmHg at 25°C |
| Index of Refraction | 1.600 |
| LogP | 1.84 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 28 |
| Complexity | 825 |
| Defined Atom Stereocenter Count | 7 |
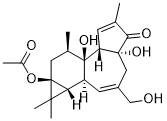
| SMILES | C[C@@H]1C[C@@]2([C@@H](C2(C)C)[C@H]3[C@]1([C@@H]4C=C(C(=O)[C@]4(CC(=C3)CO)O)C)O)OC(=O)C |
| InChi Key | BOJKFRKNLSCGHY-HXGSDTCMSA-N |
| InChi Code | InChI=1S/C22H30O6/c1-11-6-16-20(26,18(11)25)9-14(10-23)7-15-17-19(4,5)21(17,28-13(3)24)8-12(2)22(15,16)27/h6-7,12,15-17,23,26-27H,8-10H2,1-5H3/t12-,15+,16-,17-,20-,21+,22-/m1/s1 |
| Chemical Name | [(1R,2S,6R,10S,11R,13S,15R)-1,6-dihydroxy-8-(hydroxymethyl)-4,12,12,15-tetramethyl-5-oxo-13-tetracyclo[8.5.0.02,6.011,13]pentadeca-3,8-dienyl] acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Prostratin has a Ki of 210 nM and transcribes [3H]PDBu binding to CEM cells [1]. Transcriptional acute myeloid leukemia (AML) cell lines (HL-60, NB4, and U937 cells) are not able to proliferate at concentrations of prostratin (125-1000 nM). In HL-60 cells, prostatretin (125–100 nM) influences cell cycle-related molecules (pRb phosphorylation, CDK, and p21) and causes G1 arrest in AML cells. Prostratin also activates PKC, which causes AML cell lines to become active. Moreover, humidity produced by prostaglandin is necessary for PKC-driven activation of MEK/ERK/MAP signal load [2]. For HIV-1-induced activation, prostatin induction necessitates the active form of PKD3. Moreover, prostratein activates PKD3 via PKCε of the new PKC subfamily [2]. |
| Cell Assay |
Cell viability assay [2] Cell Types: HL-60, NB4 and U937 Cell Tested Concentrations: 125nM, 250nM, 500nM, 1000nM Incubation Duration: 24 hrs (hours), 48 hrs (hours), 72 hrs (hours) Experimental Results: dose-dependent inhibition 3]. Growth of acute myeloid leukemia (AML) cell lines. Cell cycle analysis[2] Cell Types: HL-60, NB4 and U937 Cell Tested Concentrations: 125 nM, 250 nM, 500 nM, 1000 nM Incubation Duration: 24 hrs (hours) Experimental Results: Induced G0/G1 phase accumulation in a concentration-dependent manner. Western Blot Analysis[2] Cell Types: HL-60 Cell Tested Concentrations: 125 nM, 250 nM, 500 nM, 1000 nM Incubation Duration: 24 hrs (hours) Experimental Results: Effects of cell cycle related molecules (pRb phosphorylation, CDK and p21) in HL- in 60 cells. |
| References |
[1]. A nonpromoting phorbol from the samoan medicinal plant Homalanthus nutans inhibits cell killing by HIV-1. J Med Chem. 1992 May 29;35(11):1978-86. [2]. The protein kinase C agonist prostratin induces differentiation of human myeloid leukemia cells and enhances cellular differentiation by chemotherapeutic agents. Cancer Lett. 2015 Jan 28;356(2 Pt B):686-96. [3]. Protein kinase D3 is essential for prostratin-activated transcription of integrated HIV-1 provirus promoter via NF-κB signaling pathway. Biomed Res Int. 2014;2014:968027. |
| Additional Infomation |
Prostratin is a phorbol ester. It has a role as a metabolite. Prostratin has been reported in Euphorbia triangularis, Euphorbia fischeriana, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~128.05 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1.25 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5610 mL | 12.8051 mL | 25.6102 mL | |
| 5 mM | 0.5122 mL | 2.5610 mL | 5.1220 mL | |
| 10 mM | 0.2561 mL | 1.2805 mL | 2.5610 mL |