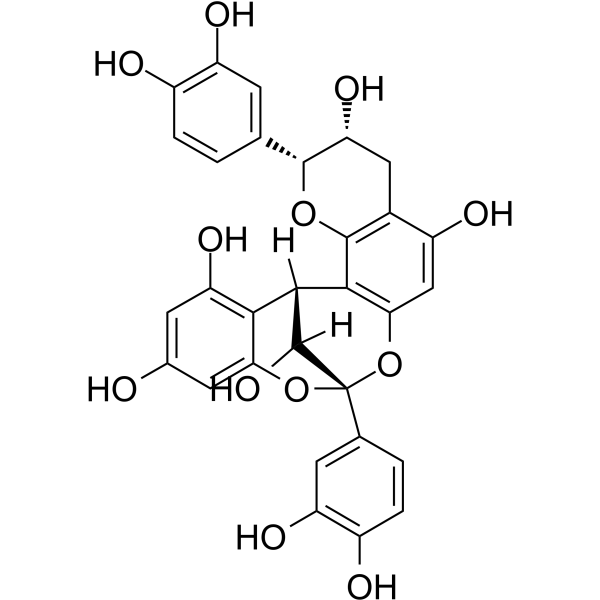
Physicochemical Properties
| Molecular Formula | C30H24O12 |
| Molecular Weight | 576.5044 |
| Exact Mass | 576.127 |
| CAS # | 41743-41-3 |
| Related CAS # | Cyanidin Chloride;528-58-5;Procyanidin B1;20315-25-7;Procyanidin B2;29106-49-8;Procyanidin C1;37064-30-5;Procyanidin A1;103883-03-0;Procyanidin B3;23567-23-9 |
| PubChem CID | 124025 |
| Appearance | White to pink solid powder |
| Density | 1.766g/cm3 |
| Boiling Point | 946ºC at 760 mmHg |
| Melting Point | 300 °C |
| Flash Point | 525.9ºC |
| Index of Refraction | 1.829 |
| LogP | 2.793 |
| Hydrogen Bond Donor Count | 9 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 42 |
| Complexity | 986 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | C1[C@H]([C@H](OC2=C1C(=CC3=C2[C@@H]4[C@H]([C@](O3)(OC5=CC(=CC(=C45)O)O)C6=CC(=C(C=C6)O)O)O)O)C7=CC(=C(C=C7)O)O)O |
| InChi Key | NSEWTSAADLNHNH-LSBOWGMISA-N |
| InChi Code | InChI=1S/C30H24O12/c31-13-7-20(37)24-22(8-13)41-30(12-2-4-16(33)19(36)6-12)29(39)26(24)25-23(42-30)10-17(34)14-9-21(38)27(40-28(14)25)11-1-3-15(32)18(35)5-11/h1-8,10,21,26-27,29,31-39H,9H2/t21-,26-,27-,29-,30+/m1/s1 |
| Chemical Name | (1R,5R,6R,13S,21R)-5,13-bis(3,4-dihydroxyphenyl)-4,12,14-trioxapentacyclo[11.7.1.02,11.03,8.015,20]henicosa-2(11),3(8),9,15,17,19-hexaene-6,9,17,19,21-pentol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Procyanidin A2 exerts antiproliferative effects on human cancer cells [1] - Procyanidin A2 modulates the IL-4-mediated signaling pathway in human alveolar epithelial cells, specifically inhibiting IL-4-induced CCL26 production (targets related to IL-4 signaling, e.g., STAT6)[2] |
| ln Vitro |
The DPPH scavenging activity of proanthocyanidin A2 is 5.08 ± 0.37 μM [1]. - Antiproliferative activity against human cancer cells: Procyanidin A2 inhibited the proliferation of three human cancer cell lines in a concentration-dependent manner. For HepG2 (hepatocellular carcinoma cells), 100 μM reduced cell viability by 52%, 50 μM by 38%, and 25 μM by 15% (vs. control) after 48-hour incubation. For MCF-7 (breast cancer cells), 100 μM caused a 48% viability reduction, and for A549 (lung cancer cells), 100 μM caused a 45% viability reduction (MTT assay)[1] - Inhibition of IL-4-induced CCL26 production in alveolar epithelial cells: In human alveolar epithelial cells (A549), pretreatment with Procyanidin A2 (25, 50 μM) for 2 hours prior to IL-4 (10 ng/mL) stimulation inhibited CCL26 expression. At 50 μM, it reduced CCL26 mRNA levels by 65% (qPCR) and CCL26 protein secretion by 58% (ELISA) compared to the IL-4-only group. At 25 μM, the reductions were 42% (mRNA) and 35% (protein), respectively. Mechanistically, it suppressed IL-4-induced STAT6 phosphorylation (western blot analysis)[2] |
| Cell Assay |
- Cancer cell proliferation assay (MTT method): Human cancer cells (HepG2, MCF-7, A549) were seeded into 96-well plates at a density of 5×10³ cells/well and cultured overnight. Procyanidin A2 (dissolved in DMSO, diluted in medium to final concentrations of 25, 50, 100 μM) was added, and the cells were incubated at 37°C in a 5% CO₂ atmosphere for 48 hours. After incubation, MTT reagent was added to each well and incubated for another 4 hours. The formazan crystals formed were dissolved with DMSO, and absorbance was measured at 570 nm. Cell viability was calculated as (absorbance of drug group/absorbance of control group) × 100%[1] - CCL26 expression assay in alveolar epithelial cells: Human alveolar epithelial cells (A549) were seeded into 6-well plates (for qPCR) or 24-well plates (for ELISA) and cultured to 80% confluence. The cells were pretreated with Procyanidin A2 (25, 50 μM) for 2 hours, then stimulated with IL-4 (10 ng/mL) for 24 hours. For qPCR: total RNA was extracted, reverse-transcribed into cDNA, and CCL26 mRNA levels were quantified using specific primers (normalized to GAPDH). For ELISA: culture supernatant was collected, and CCL26 protein concentration was measured using a specific antibody kit. For western blot: cell lysates were prepared, and STAT6 phosphorylation levels were detected using phospho-STAT6 and total STAT6 antibodies[2] |
| References |
[1]. Identification of flavonoids in litchi (Litchi chinensis Sonn.) leaf and evaluation of anticancer activities. Journal of Functional Foods. January 2014, 6, P: 555-563. [2]. Procyanidin A2 Modulates IL-4-Induced CCL26 Production in Human Alveolar Epithelial Cells. Int J Mol Sci. 2016 Nov 12;17(11). |
| Additional Infomation |
Proanthocyanidin A2 is a proanthocyanidin obtained by the condensation of (-)-epicatechin units. It has a role as an antioxidant, an anti-HIV agent, a metabolite and an angiogenesis modulating agent. It is a hydroxyflavan and a proanthocyanidin. It is functionally related to a (-)-epicatechin. Proanthocyanidin A2 has been reported in Cinnamomum iners, Litchi chinensis, and other organisms with data available. See also: Litchi fruit (part of). - Procyanidin A2 is a flavonoid compound isolated from the leaves of Litchi chinensis Sonn. (litchi)[1] - The antiproliferative effect of Procyanidin A2 on cancer cells is concentration-dependent, with higher efficacy at higher concentrations (≥50 μM) but no significant toxicity to normal human peripheral blood mononuclear cells (PBMCs) at 100 μM[1] - The inhibitory effect of Procyanidin A2 on IL-4-induced CCL26 production is mediated by blocking the phosphorylation of STAT6, a key transcription factor in the IL-4 signaling pathway that regulates CCL26 gene expression[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~173.46 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.34 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.34 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7346 mL | 8.6730 mL | 17.3461 mL | |
| 5 mM | 0.3469 mL | 1.7346 mL | 3.4692 mL | |
| 10 mM | 0.1735 mL | 0.8673 mL | 1.7346 mL |