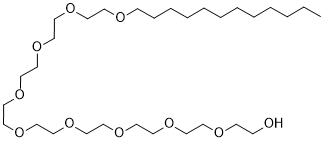
Polidocanol (Nonaethylene glycol monododecyl ether; Nonaoxyethylene monododecyl ether) is a non-ionic surfactant and polyethylene glycol (PEG) detergent used to form initial coalesced O/W emulsion droplets, as well as for protein separation and purification. It is also a local anaesthetic agent and antipruritic agent, and a component of ointments and bath additives. It relieves itching caused by eczema and dry skin.
Physicochemical Properties
| Molecular Formula | C30H62O10 |
| Molecular Weight | 582.816 |
| Exact Mass | 582.434 |
| CAS # | 3055-99-0 |
| Related CAS # | 9043-30-5 |
| PubChem CID | 656641 |
| Appearance | White to off-white oil |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 615.9±50.0 °C at 760 mmHg |
| Melting Point | 33-36ºC |
| Flash Point | 326.3±30.1 °C |
| Vapour Pressure | 0.0±4.0 mmHg at 25°C |
| Index of Refraction | 1.459 |
| LogP | 2.18 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 37 |
| Heavy Atom Count | 40 |
| Complexity | 431 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCCCCCCCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO |
| InChi Key | ONJQDTZCDSESIW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C30H62O10/c1-2-3-4-5-6-7-8-9-10-11-13-32-15-17-34-19-21-36-23-25-38-27-29-40-30-28-39-26-24-37-22-20-35-18-16-33-14-12-31/h31H,2-30H2,1H3 |
| Chemical Name | 2-[2-[2-[2-[2-[2-[2-[2-(2-dodecoxyethoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol |
| Synonyms | Asclera, Aethoxysklerol and Varithena C12E9, Dodecyl nonaethylene glycol ether, Dodecylnonaglycol, Polidocanol, Polyoxyethylene (9) lauryl ether Polidocanol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Research using many long-chain E-PDS from various organisms and a range of nonionic PEG detergents revealed that in vitro treatment with nonaethylene glycol monododecyl ether often produced respiratory quinones formed in vivo. The isoprenoid moiety's equivalent chain length. 2]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion When given intravenously, the maximum blood concentrations were reached in 15 mins. Route of elimination was not indicated. When given intravenously, the volume of distribution was 35-82L. Sytemic clearance was 0.2-0.4 L/min. Metabolism / Metabolites Metabolism was not measured. Biological Half-Life The half-life is approximately 1.5 h. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of polidocanol during breastfeeding. Although polidocanol is unlikely to adversely affect the breastfed infant, international guidelines recommend that breastfeeding be withheld for 2 days after sclerotherapy. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Plasma protein binding was not measured. |
| References |
[1]. Oil-in-Water Emulsion Templated and Crystallization-Driven Self-Assembly Formation of Poly(l-lactide)-Polyoxyethylene-Poly(l-lactide) Fibers. Langmuir. 2017 Nov 14;33(45):13060-13067. [2]. Dependence of the product chain-length on detergents for long-chain E-polyprenyl diphosphate synthases. Biochemistry. 2013 Jul 23;52(29):5002-8. [3]. Comparison of the different types of surfactants for the effect on activity and structure of soybean peroxidase. Langmuir. 2009 Feb 17;25(4):2363-8. |
| Additional Infomation |
Polidocanol is a hydroxypolyether that is nonaethylene glycol in which one of the terminal hydroxy functions is substituted by a lauryl (dodecyl) group. It has a role as a nonionic surfactant, a hepatotoxic agent and a sclerotherapy agent. It is functionally related to a nonaethylene glycol. Polidocanol is a sclerosing agent indicated to treat uncomplicated spider veins (varicose veins ≤1 mm in diameter) and uncomplicated reticular veins (varicose veins 1 to 3 mm in diameter) in the lower extremity. It is marketed under the brand names Asclera and Varithena. The formula for Polidocanol has the structural formula C12H25(OCH2CH2)nOH, a mean extent of polymerization (n) of approximately 9 and a mean molecular weight of approximately 600. Polidocanol is an alkyl polyglycol ether of lauryl alcohol with sclerosing and potential antineoplastic activities. Upon intralesional administration, polidocanol induces endothelial cell injury by disrupting calcium signaling and nitric oxide pathways. Following endothelial damage, platelets aggregate at the site of injury and attach to the venous wall, resulting in a dense network of platelets, cellular debris, and fibrin that occludes the vessel. Inducing endothelial cell damage within melanoma metastases may incite an antitumor response in untreated bystander lesions and inhibit the growth of in transit metastases and other cutaneous lesions. See also: Ethylene Oxide (has monomer) ... View More ... Drug Indication Polidocanol is a sclerosing agent indicated to treat uncomplicated spider veins and uncomplicated reticular veins in the lower extremity. FDA Label Mechanism of Action When administered, polidocanol locally damages blood vessel endothelium. Following the endothelial damage, platelets aggregate at the site and attach to the venous wall eventually resulting in a dense network of platelets, cellular debris, and fibrin that occludes the vessel. Eventually the vessel is replaced by connective fibrous tissue. Pharmacodynamics Polidocanol has a concentration and volume dependent damaging effect on the blood vessel endothelium. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~250 mg/mL (~428.96 mM) Ethanol : ~100 mg/mL (~171.58 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.57 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.57 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.57 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7158 mL | 8.5790 mL | 17.1580 mL | |
| 5 mM | 0.3432 mL | 1.7158 mL | 3.4316 mL | |
| 10 mM | 0.1716 mL | 0.8579 mL | 1.7158 mL |