Physicochemical Properties
| Molecular Formula | C16H18N4O2.HCL |
| Molecular Weight | 334.80066 |
| Exact Mass | 334.12 |
| CAS # | 78213-63-5 |
| Related CAS # | Piribedil;3605-01-4;Piribedil-d8;1398044-45-5;Piribedil dihydrochloride;1451048-94-4 |
| PubChem CID | 4850 |
| Appearance | Solid powder |
| Boiling Point | 469.4ºC at 760 mmHg |
| Flash Point | 237.7ºC |
| LogP | 2.332 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 22 |
| Complexity | 356 |
| Defined Atom Stereocenter Count | 0 |
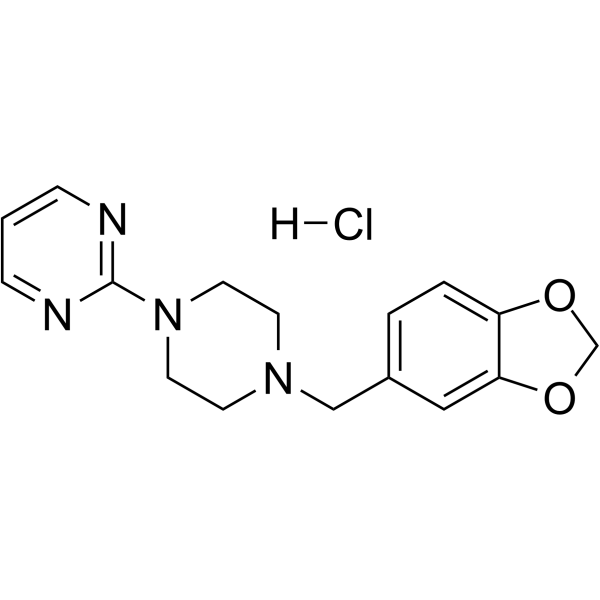
| SMILES | C1CN(CCN1CC2=CC3=C(C=C2)OCO3)C4=NC=CC=N4.Cl |
| InChi Key | VRSRBPOCDOKYKT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H18N4O2.ClH/c1-4-17-16(18-5-1)20-8-6-19(7-9-20)11-13-2-3-14-15(10-13)22-12-21-14;/h1-5,10H,6-9,11-12H2;1H |
| Chemical Name | 2-[4-(1,3-benzodioxol-5-ylmethyl)piperazin-1-yl]pyrimidine;hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | D2/D3 Receptor |
| ln Vitro | MLL1 methyltransferase activity is specifically inhibited and MLL-r cell proliferation is selectively inhibited by piribedil hydrochloride (0-160 μM, 7 days) [4]. By disrupting the MLL1-WDR5 interaction, piribedil hydrochronide (0-160 μM, 4 days) specifically lowers H3K4 methylation in MLL-r cells (THP-1 and MV4; 11). Cell cycle arrest, apoptosis, and differentiation of MLL-r cells (THP-1 and MV4; 11) are induced by piribedil hydrochronide (0-160 μM, 4 days) [4]. |
| ln Vivo | In a rat model of Parkinson's disease, piribedil hydrochloride (intraperitoneal injection, 5, 15, 40 mg/kg) can reduce levodopa-induced dyskinesia [2]. In adult common marmosets, piribedil hydrochloride (oral gavage, 4-5 mg/kg daily for 2 weeks) reverses motor deficits and increases locomotor activity [3]. In MV4;11 tumor xenografts, piribedil hydrochloride (150 mg/kg orally, daily for 21 days) inhibits MLL-r tumor growth and decreases MLL1 target gene expression [4]. |
| Enzyme Assay | Targeting WT MLL for the treatment of MLL-r leukemia, which is highly aggressive and resistant to chemotherapy, has been shown to be a promising strategy. However, drug treatments targeting WT MLL are lacking. We used an in vitro histone methyltransferase assay to screen a library consists of 592 FDA-approved drugs for MLL1 inhibitors by measuring alterations in HTRF signal and found that Piribedil represented a potent activity. Piribedil specifically inhibited the proliferation of MLL-r cells by inducing cell-cycle arrest, apoptosis and myeloid differentiation with little toxicity to the non-MLL cells. Mechanism study showed Piribedil blocked the MLL1-WDR5 interaction and thus selectively reduced MLL1-dependent H3K4 methylation. Importantly, MLL1 depletion induced gene expression that was similar to that induced by Piribedil and rendered the MLL-r cells resistant to Piribedil-induced toxicity, revealing Piribedil exerted anti-leukemia effects by targeting MLL1. Furthermore, both the Piribedil treatment and MLL1 depletion sensitized the MLL-r cells to doxorubicin-induced apoptosis. Our study support the hypothesis that Piribedil could serve as a new drug for the treatment of MLL-r AML and provide new insight for further optimization of targeting MLL1 HMT activity.[4] |
| Cell Assay |
Cell proliferation assay[4] Cell Types: MLL-r AML cells (THP-1 and MV4;11), non-MLL leukemia cell line (K562) Tested Concentrations: 0, 20, 40, 80 and 160 μM Incubation Duration: 0- 7 Experimental Results: Inhibited the growth rate of THP-1 and MV4;11 cells in a time-dependent manner. Western Blot Analysis[4] Cell Types: THP-1 and MV4; 11 cells Tested Concentrations: 0, 20, 40, 80 and 160 μM Incubation Duration: 4 days Experimental Results: H3K4me2 and H3K4me3 levels were diminished, but did not affect other histones Methylation, such as H3K79, H3K36 and H3K27. |
| Animal Protocol |
Animal/Disease Models: Parkinson's disease rat model [2] Doses: 5, 15, 40 mg/kg Route of Administration: intraperitoneal (ip) injection, 5 minutes before levodopa administration. Experimental Results: Rotational behavior as well as AD (axial dystonia), OD (oral dyskinesia) and FD (forelimb dyskinesia) were diminished at doses of 5 and 40 mg/kg. LD (dyskinesia) increased at 40 mg/kg. Animal/Disease Models: Adult common marmoset [3] Doses: 4-5 mg/kg Route of Administration: po (oral gavage), one time/day for 2 weeks Experimental Results: Improved alertness and vigilance, reversed MPTP in rostral and caudal stripes Down-regulation of protachykinin mRNA induced in the cyst. |
| References |
[1]. Piribedil, a dopamine agonist, in Parkinson's disease. Clin Pharmacol Ther. 1974 Dec;16(6):1077-82. [2]. The effect of piribedil on L-DOPA-induced dyskinesias in a rat model of Parkinson's disease: differential role of α(2) adrenergic mechanisms. J Neural Transm (Vienna). 2013 Jan;120(1):31-6. [3]. Repeated administration of piribedil induces less dyskinesia than L-dopa in MPTP-treated common marmosets: a behavioural and biochemical investigation. Mov Disord. 2002 Sep;17(5):887-901. [4]. Piribedil disrupts the MLL1-WDR5 interaction and sensitizes MLL-rearranged acute myeloid leukemia (AML) to doxorubicin-induced apoptosis. Cancer Lett. 2018 Sep 1;431:150-160. |
| Additional Infomation |
2-[4-(1,3-benzodioxol-5-ylmethyl)-1-piperazinyl]pyrimidine is a N-arylpiperazine. Piribedil has been investigated in Parkinson's Disease. A dopamine D2 agonist. It is used in the treatment of parkinson disease, particularly for alleviation of tremor. It has also been used for circulatory disorders and in other applications as a D2 agonist. |
Solubility Data
| Solubility (In Vitro) | DMSO: > 10 mM |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9869 mL | 14.9343 mL | 29.8686 mL | |
| 5 mM | 0.5974 mL | 2.9869 mL | 5.9737 mL | |
| 10 mM | 0.2987 mL | 1.4934 mL | 2.9869 mL |