Physicochemical Properties
| Molecular Formula | C6H11NO2 |
| Molecular Weight | 129.1570 |
| Exact Mass | 129.078 |
| CAS # | 535-75-1 |
| Related CAS # | L-Pipecolic acid;3105-95-1;Pipecolic acid-d9;790612-94-1 |
| PubChem CID | 849 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 265.8±33.0 °C at 760 mmHg |
| Melting Point | 268-278ºC |
| Flash Point | 114.5±25.4 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.479 |
| LogP | -2.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 9 |
| Complexity | 114 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | HXEACLLIILLPRG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H11NO2/c8-6(9)5-3-1-2-4-7-5/h5,7H,1-4H2,(H,8,9) |
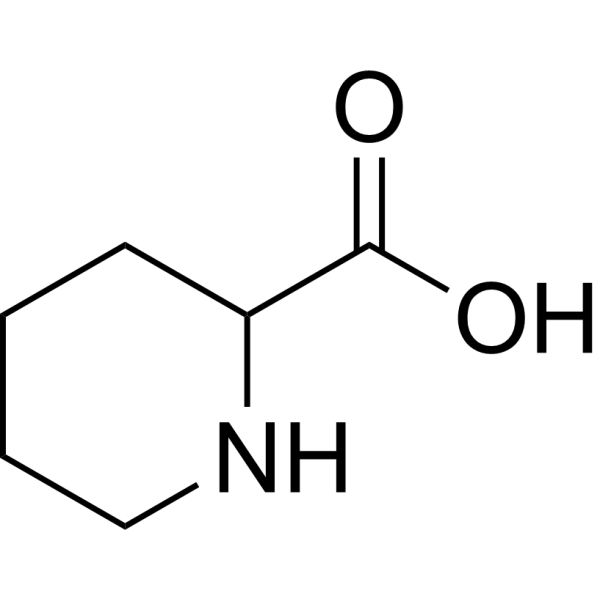
| Chemical Name | piperidine-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
- In microbial cultures (e.g., Pseudomonas putida, Bacillus subtilis), Pipecolic acid biosynthesis was detected via enzymatic reactions. When lysine (10 mM) was used as the substrate, the combination of lysine 6-dehydrogenase and Δ¹-piperideine-2-carboxylate reductase produced Pipecolic acid at a yield of 65–78% after 24 hours of incubation at 37°C (pH 7.2). The product was quantified by high-performance liquid chromatography (HPLC) with a detection limit of 0.1 μM [1] - In clinical sample analysis: Cerebrospinal fluid (CSF) and plasma samples from patients with pyridoxine-dependent epilepsy (PDE) were analyzed. Pipecolic acid concentrations in PDE patients’ CSF (12.5–98.3 μmol/L) were significantly higher than those in healthy controls (<1.0 μmol/L), and plasma concentrations (8.2–45.6 μmol/L) were also elevated compared to controls (<0.8 μmol/L). Detection was performed using gas chromatography-mass spectrometry (GC-MS) with a coefficient of variation (CV) <5% [2] |
| Enzyme Assay |
- Assay for lysine 6-dehydrogenase (involved in Pipecolic acid biosynthesis): The reaction system (1 mL) contained 50 mM Tris-HCl buffer (pH 8.0), 10 mM L-lysine (substrate), 0.5 mM NAD+ (cofactor), and 10 μg purified lysine 6-dehydrogenase. The mixture was incubated at 30°C for 30 minutes, then the reaction was stopped by adding 0.1 mL 10% trichloroacetic acid. The amount of Δ¹-piperideine-2-carboxylate (intermediate product) was measured by HPLC at 245 nm, and enzyme activity was calculated as nmol product/min per mg protein [1] - Assay for Δ¹-piperideine-2-carboxylate reductase (subsequent enzyme in biosynthesis): The reaction system (1 mL) included 50 mM phosphate buffer (pH 7.5), 5 mM Δ¹-piperideine-2-carboxylate (substrate), 0.2 mM NADPH (cofactor), and 8 μg purified reductase. Incubation was at 37°C for 20 minutes, and the reaction was terminated by heating at 95°C for 5 minutes. The amount of Pipecolic acid (product) was quantified by HPLC at 210 nm, with enzyme activity expressed as μmol product/h per mg protein [1] |
| References |
[1]. Pipecolic acid in microbes: biosynthetic routes and enzymes. J Ind Microbiol Biotechnol. 2006 Jun;33(6):401-7. [2]. Pipecolic acid as a diagnostic marker of pyridoxine-dependent epilepsy. Neuropediatrics. 2005 Jun;36(3):200-5. |
| Additional Infomation |
Pipecolic acid is a piperidinemonocarboxylic acid in which the carboxy group is located at position C-2. It is a conjugate acid of a pipecolate. Pipecolic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). Pipecolic acid has been reported in Angelica gigas, Slafractonia leguminicola, and other organisms with data available. - Pipecolic acid is a cyclic amino acid and a metabolite of L-lysine. In microbes, its biosynthesis mainly involves two key enzymes: lysine 6-dehydrogenase (catalyzes lysine to Δ¹-piperideine-2-carboxylate) and Δ¹-piperideine-2-carboxylate reductase (catalyzes the intermediate to Pipecolic acid) [1] - Pipecolic acid serves as a specific diagnostic marker for pyridoxine-dependent epilepsy (PDE), an autosomal recessive disorder caused by mutations in the ALDH7A1 gene. PDE patients have impaired Pipecolic acid catabolism, leading to its accumulation in CSF, plasma, and urine [2] - In PDE diagnosis, measuring Pipecolic acid levels in CSF has higher diagnostic value than plasma (sensitivity 98%, specificity 96% for CSF vs. sensitivity 90%, specificity 92% for plasma). Administration of pyridoxine (vitamin B6) to PDE patients reduces Pipecolic acid concentrations by 40–60% within 1–2 weeks [2] - Some microbes (e.g., Rhizobium leguminosarum) use Pipecolic acid as a signaling molecule to regulate symbiotic nitrogen fixation, while others (e.g., Aspergillus niger) produce it as a secondary metabolite with potential antimicrobial activity [1] |
Solubility Data
| Solubility (In Vitro) | H2O : ~50 mg/mL (~387.12 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.7423 mL | 38.7117 mL | 77.4234 mL | |
| 5 mM | 1.5485 mL | 7.7423 mL | 15.4847 mL | |
| 10 mM | 0.7742 mL | 3.8712 mL | 7.7423 mL |