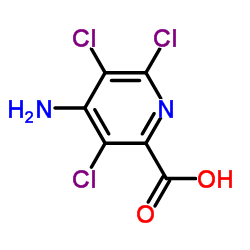
Picloram (Tordon; Grazon) is a auxinic and systemic herbicide used for general woody plant control such as broad-leaved weeds, but most grasses are resistant.
Physicochemical Properties
| Molecular Formula | C6H3CL3N2O2 |
| Molecular Weight | 241.45 |
| Exact Mass | 239.926 |
| CAS # | 1918-02-1 |
| PubChem CID | 15965 |
| Appearance | White to off-white solid powder |
| Density | 1.8±0.1 g/cm3 |
| Boiling Point | 420.5±45.0 °C at 760 mmHg |
| Melting Point | 200 °C (dec.)(lit.) |
| Flash Point | 208.1±28.7 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.677 |
| LogP | 2.94 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 13 |
| Complexity | 216 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | NQQVFXUMIDALNH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H3Cl3N2O2/c7-1-3(10)2(8)5(9)11-4(1)6(12)13/h(H2,10,11)(H,12,13) |
| Chemical Name | 4-amino-3,5,6-trichloropyridine-2-carboxylic acid |
| Synonyms | Grazon Tordon Picloram |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Disposition studies in young cattle confirm that tissue retention is minimal with dietary intakes of 2.6-23 mg/kg per day. At the highest dietary intake level studied (1600 ppm [mg/kg]), tissue concentrations were recorded as (ppm (mg/kg)): kidney, 15-18; blood, 1.4-2; liver, 1.1-1.6; and muscle and fat, 0.3-0.5. Clearance was rapid after cessation of intake. The fate of picloram (4-amino-3,5,6-trichloropicolinic acid), an active ingredient in TORDON brand herbicides, was defined in 6 healthy male volunteers following single /oral/ doses of 5.0 and 0.5 mg/kg, and a dermal dose of 2.0 mg/kg. Picloram was administered orally as the sodium salt in grape juice. The dermal dose was applied to the volunteers' backs as the free acid dissolved in ethanol. The data indicate picloram was rapidly absorbed from the gastrointestinal tract (t1/2 = 20 min) and rapidly excreted unchanged in the urine. Over 90% of the /oral/ dose was recovered as unchanged picloram in the urine excreted through 72 hr; most of the dose (>75%) was excreted within 6 hr and the remainder was excreted with an average half-life of 27 hr. By comparison picloram was slowly absorbed through the skin (t1/2 = 12 hr) and, based on the quantity of picloram excreted in the urine, only a small fraction (0.2%) of the picloram applied to the skin was absorbed. These data indicate that picloram because of its rapid excretion has a low potential to accumulate in man during repeated or prolonged exposure. In addition, picloram was poorly absorbed through human skin and it is unlikely that acutely toxic quatities will be absorbed by this route. Picloram is rapidly absorbed from the GI tract & is excreted virtually unchanged in the urine & feces of male Fischer 344 rats within 48 hr. Following a 10 mg/kg (14)C-picloram intravenous dose, the isotope was cleared biphasically & excreted in the urine. ... Balance studies in rats indicated that 98.4% of the dose was recovered. Urinary excretion resulted in an 80% to 84% recovery, fecal excretion resulted in approx 15% recovery, less than 0.5% was recovered in the bile, and virtually no radioactivity was recovered as trapped (14)CO2 or as other volatile compounds. Studies with (14)C-picloram showed that 90% of the compound fed in the diet to dogs was excreted within 48 hr in the urine, with small amounts appearing in the feces. For more Absorption, Distribution and Excretion (Complete) data for PICLORAM (6 total), please visit the HSDB record page. Metabolism / Metabolites The major metabolite was 2-ethyl-1,6-hexanoic acid /after administration of 14C-picloram isooctyl ester/. This study supports the fact that picloram ethylhexyl ester is hydrolyzed rapidly to picloram (free acid) and 2-ethyl hexanol... No measurable residues were found in milk samples from dairy cows fed 10-100 ppm picloram in the feed. Milk samples from cows fed 150-1,000 ppm picloram in the feed contained low levels (0.05-0.29 ppm) of residue which declined rapidly and were undetectable 58 hours after withdrawal from the feed. Biological Half-Life Following a 10 mg/kg (14)C-picloram intravenous dose /to rats/, the isotope was cleared biphasically and excreted in the urine. The half-time for rapid and slow clearance from plasma was 6.3 and 128 min, respectively. Oral admin of the same dose resulted in comparable half-times of 29 min and 3.8 hr, respectively. At higher (1400 mg/kg) oral doses, the plasma levels remained constant for 3 hr, then slowly declined. |
| Toxicity/Toxicokinetics |
Interactions Single oral doses of 720 mg/kg in sheep ... did not result in overt signs of toxicity. ... A toxic synergism between picloram & (2,4-dichlorophenoxy)acetic acid (2,4-D) appears to exist in sheep. A 36 mg/kg dose of picloram plus 2,4-D at 134 mg/kg resulted in death of livestock. Microsomes from male rats treated with picloram (100 mg/kg/day) for 7 days showed a 48% decrease in 16 alpha-hydroxylase activity when incubated with (4-14C) androstenedione. These data are consistent with the assertion that picloram decreases the titer of hepatic male specific cytochrome P-450h. ... First, SDS polyacrylamide gel electrophoresis revealed an intensified hepatic microsomal polypeptide (MW 54,000) following picloram pretreatment. This polypeptide co-migrated with protein bands which were correspondingly intensified after pretreatment with known inducers of cytochrome P-450d (3-methylcholanthrene and isosafrole). Second, no increase in the binding of metyrapone to picloram treated microsomes was noted compared with controls, suggesting no increase in phenobarbital-inducible forms of cytochrome P-450. Third, hepatic microsomes from picloram treated rats activated 2-amino-3-methylimidazo (4,5-f)quinoline (a cytochrome P-450d mediated catalysis) causing a 5-fold increase in the number of induced Salmonella typhimurium TA98 revertant colonies formed compared with control microsomes. Fourth, the binding of n-octylamine to hepatic microsomes from picloram-treated rats showed, like microsomes from 3-methylcholanthrene-treated rats, an increase in the proportion of high-spin cytochrome P-450 present. Cytochrome P-450d is known to be a high spin hemoprotein. Non-Human Toxicity Values LD50 Rat (male, Sprague-Dawley derived) oral 950 mg/kg (95% CI: 812-1120 mg/kg) LD50 Rat (female, Sprague-Dawley derived) oral 686 mg/kg (95% CI: 599-786 mg/kg) LD50 Rabbit oral 2000 mg/kg LD50 Guinea pig oral 1922 mg/kg For more Non-Human Toxicity Values (Complete) data for PICLORAM (11 total), please visit the HSDB record page. |
| References | Arabidopsis PIC30 Encodes a Major Facilitator Superfamily Transporter Responsible for the Uptake of Picolinate Herbicides. Plant J. 2020 Apr;102(1):18-33. |
| Additional Infomation |
Picloram appears as fine beige crystals or white powder. Odor of chlorine. (NTP, 1992) Picloram is a pyridinemonocarboxylic acid that is pyridine-2-carboxylic acid which is substituted by a chloro group at positions 3,5 and 6, and by an amino group at position 4. It is a systemic herbicide used to control deeply rooted herbaceous weeds and woody plants in rights-of-way, forestry, range lands, pastures, and small grain crops. It has a role as a herbicide and a synthetic auxin. It is an aminopyridine, a pyridinemonocarboxylic acid, a chloropyridine and an organochlorine pesticide. It is functionally related to a picolinic acid. Picloram is a persistent systemic herbicide used for general woody plant control and a broad-leaved weeds on non-crop and utility areas. It also controls a wide range of broad-leaved weeds, but most grasses are resistant. A chlorinated derivative of picolinic acid, picloram is in the pyridine family of herbicides. It is systemically absorbed by roots and leaves and translocated. A picolinic acid derivative that is used as a herbicide. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~414.15 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.1416 mL | 20.7082 mL | 41.4164 mL | |
| 5 mM | 0.8283 mL | 4.1416 mL | 8.2833 mL | |
| 10 mM | 0.4142 mL | 2.0708 mL | 4.1416 mL |