Physicochemical Properties
| Molecular Formula | C5H11NO2S |
| Molecular Weight | 149.21 |
| Exact Mass | 149.051 |
| CAS # | 52-67-5 |
| Related CAS # | DL-Penicillamine;52-66-4;Penicillamine-d3 |
| PubChem CID | 5852 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 251.8±35.0 °C at 760 mmHg |
| Melting Point | 210 °C (dec.)(lit.) |
| Flash Point | 106.1±25.9 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.528 |
| LogP | 0.93 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 9 |
| Complexity | 124 |
| Defined Atom Stereocenter Count | 1 |
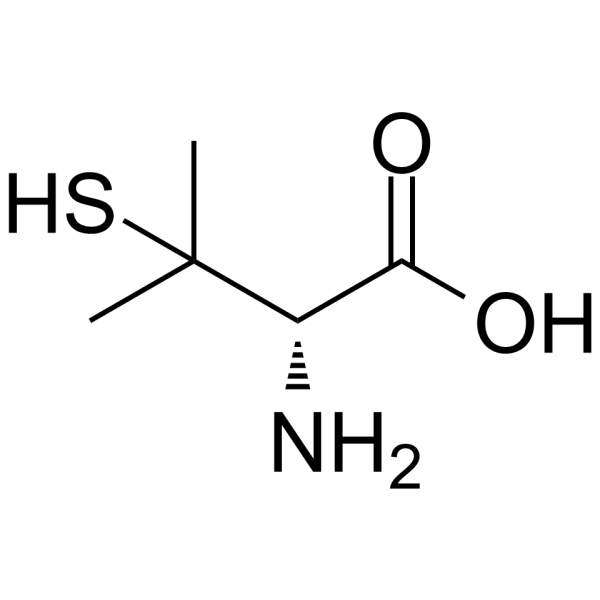
| SMILES | CC(C)([C@H](C(=O)O)N)S |
| InChi Key | VVNCNSJFMMFHPL-VKHMYHEASA-N |
| InChi Code | InChI=1S/C5H11NO2S/c1-5(2,9)3(6)4(7)8/h3,9H,6H2,1-2H3,(H,7,8)/t3-/m0/s1 |
| Chemical Name | (2S)-2-amino-3-methyl-3-sulfanylbutanoic acid |
| Synonyms | Penicillamine; D-Penicillamine; Penicillamine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Cellular immunological responses are stimulated by penicillamine (D-(-)-penicillamine) (5 mg; 7 days; CD4+ and CD+ splenocytes) [3]. |
| ln Vivo | Serum free copper concentrations are raised by penicillamine (D-(-)-penicillamine) (200 mg/kg; ig; daily for 3, 10, and 14 days; tx mice and DL mice) [1]. In the brains of tx mice and DL mice, penicillamine (200 mg/kg; ig; daily for 3, 10, and 14 days) enhances the expression of CTR1 and ATP7A mRNA [1]. Penicillamine causes oxidative stress in the central nervous system when administered intraperitoneally (i.g., 200 mg/kg; tx mice and DL mice) for 3, 10, and 14 days [1]. Penicillamine has a binaural impact on epileptic convulsions when administered intraperitoneally (0.1–250 mg/kg) to male NMRI mice once for 90 minutes [2]. Small dosages of penicillamine (5 mg/kg; intravenous injection; once daily for 8 weeks; male BN rats) can stop autoimmunity from developing [3]. |
| Cell Assay |
Western Blot Analysis[3] Cell Types: CD4+ and CD+ splenocytes Tested Concentrations: 5 mg Incubation Duration: 7 days Experimental Results: IL-4 and IFN-γ mRNA expression increased after high-dose treatment, and expression in CD4+ and CD+ splenocytes remained Keep it high. |
| Animal Protocol |
Animal/Disease Models: Toxic lactation mutant mice (tx mice) and DL mice [1] Doses: 200 mg/kg Route of Administration: po (oral gavage); one time/day for 3, 10 and 14 days Experimental Results: tx on day 3 Free copper concentrations increased in mouse serum. Animal/Disease Models: Toxic lactation mutant mice (tx mice) and DL mice [1] Doses: 200 mg/kg Route of Administration: po (oral gavage); one time/day for 3, 10 and 14 days Experimental Results: ATP7A mRNA expression Increased by 4 times. CTR1 mRNA expression increased 6.9-fold in the cortex and 9.1-fold in the basal ganglia of tx mice. Animal/Disease Models: Toxic lactation mutant mice (tx mice) and DL mice [1] Doses: 200 mg/kg Route of Administration: po (oral gavage); one time/day for 3, 10 and 14 days Experimental Results: Via nitric oxide /NMDA pathway increases MDA concentration and decreases GSH/GSSG ratio. Animal/Disease Models: Male NMRI mouse [2] Doses: 0.1, 0.5, 1, 10, 100, 150 and 250 mg/kg Route of Administration: intraperitoneal (ip) injection; once for 90 minutes Experimental Results: Low dose (0.5 mg/kg) It has anticonvulsant effects, and high dos |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion rapidly but incompletely Excretion is mainly renal, mainly as disulfides. HUMAN SUBJECTS SUFFERING FROM WILSON'S DISEASE, RAPIDLY ABSORBED ORAL DOSE OF (35)S DL-PENICILLAMINE. PLASMA CONCN OF (35)S PEAKED WITHIN 60 MIN. (35)S WAS RAPIDLY EXCRETED, ALMOST COMPLETELY IN 24 HR URINE WHERe 73% OF ADMIN (35)S ... RECOVERED. THERE WAS INTERSUBJECT VARIATION IN EXTENT OF BINDING ... BY PLASMA PROTEINS. /DL-PENICILLAMINE/ FROM METABOLIC POINT OF VIEW, D-PENICILLAMINE IS VIRTUALLY INERT, & THIS OBSERVATION IS COMPATIBLE WITH FACT THAT EXTRACELLULAR WATER MAKES UP MAIN DISTRIBUTION VOL FOR D-PENICILLAMINE. PENICILLAMINE IS WELL ABSORBED (40% to70%) FROM GI TRACT &, THEREFORE, HAS DECIDED ADVANTAGE OVER OTHER CHELATING AGENTS. PEAK CONCN IN BLOOD ARE OBTAINED BETWEEN 1 AND 3 HR AFTER ADMINISTRATION. ... /IT/ IS SOMEWHAT RESISTANT TO ATTACK BY CYSTEINE DESULFHYDRASE OR L-AMINO ACID OXIDASE. AS A RESULT ... IS RELATIVELY STABLE IN VIVO. ... HEPATIC BIOTRANSFORMATION IS RESPONSIBLE FOR MOST OF THE DEGRADATION OF PENICILLAMINE, AND VERY LITTLE IS EXCRETED UNCHANGED. METABOLITES ARE FOUND IN BOTH URINE AND FECES. The effects of chelating agents (citric acid, tartaric acid, penicillamine and ethylenediaminetetraacetic acid) and cysteine on the absorption of mercuric chloride were investigated in rats. Perfusion of the small intestine showed that the chelating agents and cysteine decreased the absorption of mercuric chloride depending on their stability of constants wtih Hg2+, under the predominant conditions of water absorption and secretion. The difference in absorption of mercuric chloride between both conditions was inversely correlated with their logarithmic stability constant values. These agents decreased the transport of mercuric chloride through the everted intestinal wall and the uptake of mercuric chloride by the intestinal brush border membrane in a similar manner. From these results, it is suggested that the chelating agents and cysteine decrease the absorption of mercuric chloride through the pores of the brush border membrane due to th solvent drag effect. For more Absorption, Distribution and Excretion (Complete) data for (D)-PENICILLAMINE (7 total), please visit the HSDB record page. Metabolism / Metabolites Hepatic The transformation of D-penicillamine was studied in orally and iv dosed rats and in human plasma in vitro. In each case, low molecular weight metabolites (previously identified as disulfides) and a mixed disulfide between D-penicillamine and albumin (D-penicillamine-protein) formed. The rates of D-penicillamine elimination, other than through protein conjugation, were comparable in the rat groups to the rate of oxidation to low molecula weight metabolites in vitro. The rates of transformation to D-penicillamine protein were also comparable in the in vitro preparations and in orally treated rats. These qualitative and quantitative similarities suggest blood plasma may be an important site of transformation in vivo. Extracellular oxidation of D-penicillamine may be linked to its antirheumatic action, either through reduction of oxygen species or through formation of D-penicillamine protein disulfides at surfaces of mononuclear leukocytes. Biological Half-Life 1 hour |
| Toxicity/Toxicokinetics |
Interactions DOWNWARD ADJUSTMENT OF DOSAGE OF OTHER ANTI-INFLAMMATORY DRUGS, ESP CORTICOSTEROIDS, IS OFTEN POSSIBLE AFTER ADDN OF PENICILLAMINE TO DRUG REGIMEN. Pyridoxine, 10-25 mg/day, should be administered concomitantly because penicillamine inhibits pyridoxal-dependent enzymes. Concurrent use /of 4-aminoquinolines or bone marrow depressants ... or gold compounds or immunosuppressants, excepting glucocorticoids/ with penicillamine may increase the potential for serious hematologic and/or renal adverse reactions. Concurrent use with 4-aminoquinolines may ... increase the risk of severe dermatologic reactions. For more Interactions (Complete) data for (D)-PENICILLAMINE (8 total), please visit the HSDB record page. |
| References |
[1]. Penicillamine increases free copper and enhances oxidative stress in the brain of toxic milk mice. PLoS One. 2012;7(5):e37709. [2]. Effects of D-penicillamine on pentylenetetrazole-induced seizures in mice: involvement of nitric oxide/NMDA pathways. Epilepsy Behav. 2014 Oct;39:42-7. [3]. Tolerance induced by low dose D-penicillamine in the brown Norway rat model of drug-induced autoimmunity is immune-mediated. Chem Res Toxicol. 2004 Jan;17(1):82-94. [4]. Penicillamine revisited: historic overview and review of the clinical uses and cutaneous adverse effects. Am J Clin Dermatol. 2013 Jun;14(3):223-33. |
| Additional Infomation |
Therapeutic Uses Antidotes; Antirheumatic Agents; Chelating Agents THE D ISOMER IS USED CLINICALLY, ALTHOUGH THE L ISOMER ALSO FORMS CHELATION COMPLEXES. PENICILLAMINE IS EFFECTIVE CHELATOR OF COPPER, MERCURY, ZINC, & LEAD & PROMOTES EXCRETION OF THESE METALS IN URINE. THERE IS SOME PROMISE IN TREATMENT OF RHEUMATOID ARTHRITIS WITH PENICILLAMINE. ... BENEFICIAL EFFECT IS SEEN ONLY AFTER SEVERAL WK OF TREATMENT, & ARTHRITIC SYMPTOMS RETURN IF DRUG IS WITHDRAWN PREMATURELY. PENICILLAMINE HAS BECOME ESTABLISHED IN TREATMENT OF CYSTINURIA & ASSOC NEPHROLITHIASIS. IN DOSE OF 30 MG/KG/DAY, IT LOWERS OR ELIMINATES URINARY CYSTINE & PREVENTS FURTHER STONE DEVELOPMENT. For more Therapeutic Uses (Complete) data for (D)-PENICILLAMINE (11 total), please visit the HSDB record page. Drug Warnings VET: USE IN PREGNANCY IS CONTRAINDICATED BECAUSE OF ITS CHELATING EFFECT ON TRACE METALS. CROSS SENSITIVITY BETWEEN PENICILLIN & PENICILLAMINE DOES NOT ALWAYS OCCUR; THEREFORE, PENICILLAMINE CAN BE GIVEN CAUTIOUSLY TO PATIENTS WHO ARE HYPERSENSITIVE TO PENICILLIN. CAREFUL EXAM OF SKIN, AS WELL AS URINALYSIS, DIFFERENTIAL & WHITE BLOOD CELL COUNTS, DIRECT PLATELET COUNTS, & HEMOGLOBIN DETERMINATION SHOULD BE PERFORMED. EXPTL, TOXIC EFFECTS IN RATS GIVEN HIGH DOSES OF PENICILLAMINE RESEMBLE THOSE SEEN IN PYRIDOXINE DEFICIENCY, & EFFECTS ARE REVERSED BY FEEDING PYRIDOXINE. IN HUMAN BEINGS, PYRIDOXINE ANTAGONISM IS READILY DEMONSTRATED WITH L & DL FORMS, BUT RARELY WITH D FORM. ... INCR URINARY EXCRETION OF XANTHURENIC ACID & KYNURENINE. For more Drug Warnings (Complete) data for (D)-PENICILLAMINE (11 total), please visit the HSDB record page. Pharmacodynamics Penicillamine is a chelating agent used in the treatment of Wilson's disease. It is also used to reduce cystine excretion in cystinuria and to treat patients with severe, active rheumatoid arthritis unresponsive to conventional therapy. Penicillamine is used as a form of immunosuppression to treat rheumatoid arthritis. Penicillamine inhibits macrophages, decreases IL-1 and the number of T-lymphocytes, and prevents collagen cross linkage. In Wilson's disease it binds copper, allowing it to be eliminated in the urine. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~125 mg/mL (~837.75 mM) DMSO : ~1.43 mg/mL (~9.58 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (670.20 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.7020 mL | 33.5098 mL | 67.0196 mL | |
| 5 mM | 1.3404 mL | 6.7020 mL | 13.4039 mL | |
| 10 mM | 0.6702 mL | 3.3510 mL | 6.7020 mL |