Paritaprevir (formerly ABT-450; ABT450; Veruprevir; Viekirax; Viekira Pak; Technivie) is an acylsulfonamide analog and a component of the anti-HCV drug combination therapies: Viekira Pak (Paritaprevir+ombitasvir, ritonavir and dasabuvir) and Technivie (Paritaprevir+ombitasvir and ritonavir).This non-structural protein 3/4A (NS3/4A) protease inhibitor was authorized in 2015 as a component of combination therapy for the treatment of chronic Hepatitis C. The combination therapy included two versions: Technivie/Viekirax, which included ombitasvir and ritonavir, and Viekira Pak, which included ombitasvir, dasabuvir, and ritonavir. With EC50 values of 1 and 0.21 nM for HCV 1a and 1b, respectively, it inhibits NS3/4A. Abbott Laboratories made the discovery and developed the treatment, which appears to be effective in treating hepatitis C. With an EC50 of 0.09 nM against GT4a, paritaprevir exhibits antiviral activity against HCV GT1-4 and GT6 in vitro (EC50 range: 0.09 to 19 nM). For individuals with hepatitis C virus genotype 1, this combination of ribavirin and ritonavir given for 12 weeks has been estimated to have a 95% rate of sustained virologic response 24 weeks after treatment. Since paritaprevir targets the binding site, treatment resistance is rare. However, mutations at positions 155 and 168 in NS3 have been observed to cause resistance to the medication.
Physicochemical Properties
| Molecular Formula | C40H43N7O7S |
| Molecular Weight | 765.88 |
| Exact Mass | 765.294 |
| Elemental Analysis | C, 62.73; H, 5.66; N, 12.80; O, 14.62; S, 4.19 |
| CAS # | 1216941-48-8 |
| Related CAS # | Paritaprevir dihydrate;1456607-71-8 |
| PubChem CID | 45110509 |
| Appearance | White to off-white powder |
| LogP | 6.346 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 55 |
| Complexity | 1600 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | S(C1([H])C([H])([H])C1([H])[H])(N([H])C([C@@]12C([H])([H])[C@@]1([H])C([H])=C([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[C@@]([H])(C(N1C([H])([H])[C@@]([H])(C([H])([H])[C@]1([H])C(N2[H])=O)OC1C2=C([H])C([H])=C([H])C([H])=C2C2=C([H])C([H])=C([H])C([H])=C2N=1)=O)N([H])C(C1C([H])=NC(C([H])([H])[H])=C([H])N=1)=O)=O)(=O)=O |
| InChi Key | UAUIUKWPKRJZJV-QPLHLKROSA-N |
| InChi Code | InChI=1S/C40H43N7O7S/c1-24-21-42-33(22-41-24)35(48)43-32-16-6-4-2-3-5-11-25-20-40(25,39(51)46-55(52,53)27-17-18-27)45-36(49)34-19-26(23-47(34)38(32)50)54-37-30-14-8-7-12-28(30)29-13-9-10-15-31(29)44-37/h5,7-15,21-22,25-27,32,34H,2-4,6,16-20,23H2,1H3,(H,43,48)(H,45,49)(H,46,51)/b11-5-/t25-,26-,32+,34+,40-/m1/s1 |
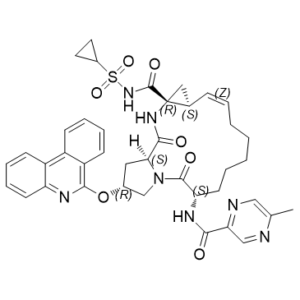
| Chemical Name | (1S,4R,6S,7Z,14S,18R)-N-cyclopropylsulfonyl-14-[(5-methylpyrazine-2-carbonyl)amino]-2,15-dioxo-18-phenanthridin-6-yloxy-3,16-diazatricyclo[14.3.0.04,6]nonadec-7-ene-4-carboxamide |
| Synonyms | ATB450; ABT 450; ABT-450; Veruprevir; Paritaprevir; Brand name: VIEKIRA PAK; 1216941-48-8; Veruprevir; ABT-450; Veruprevir anhydrous; ABT450; 1221573-85-8; Paritaprevir(ABT-450); |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
HCV 1a (EC50 = 1 nM); HCV 1b (EC50 = 0.21 nM); SARS-CoV 3CLpro (IC50 = 1.31 μM)
Paritaprevir (also named ABT-450) targets hepatitis C virus (HCV) NS3/4A protease; EC50 values are 1.0 nM (genotype 1a), 0.21 nM (genotype 1b), 5.3 nM (genotype 2a), 19 nM (genotype 3a), 0.09 nM (genotype 4a), 0.69 nM (genotype 6a) against stable HCV replicons with NS3 protease [2] Paritaprevir is an NS3/4A protease inhibitor [1] |
| ln Vitro |
Paritaprevir inhibits in vitro the p-glycoprotein (p-gp)[1]. With 50% effective concentration values of 1.0, 0.21, 5.3, 19, 0.09, and 0.69 nM against stable HCV replicons with NS3 protease from genotypes 1a, 1b, 2a, 3a, 4a, and 6a, respectively, ABT-450 is an effective inhibitor of HCV NS3/4A protease. With a CC50 greater than 37 μM, ABT-450 exhibits an in vitro selectivity index of approximately 37,000 times. ABT-450 has an EC50 of 5.3 nM against the genotype 2a JFH-1 subgenomic replicon, indicating activity against various HCV genotypes[2]. 1. Paritaprevir (ABT-450) exhibited potent inhibitory activity against HCV NS3/4A protease from different genotypes in stable HCV replicon assays: EC50 values were 1.0 nM (1a), 0.21 nM (1b), 5.3 nM (2a), 19 nM (3a), 0.09 nM (4a), and 0.69 nM (6a) [2] 2. In vitro resistance selection assays of Paritaprevir (ABT-450) in genotype 1 HCV replicons showed that the most common amino acid variants were located in NS3 at positions 155, 156, and 168; the D168Y variant conferred the highest resistance (219-fold for 1a, 337-fold for 1b) [2] |
| ln Vivo |
Paritaprevir achieves maximum concentrations after oral administration in a mean of 4-5 hours, with exposure increases exceeding dose proportionality. Approximately 50% of the total bioavailability occurs when food is administered. It exhibits high plasma protein binding (between 97-99.9%) and 16.7 liters of apparent volume of distribution. Paritaprevir is metabolized primarily by CYP3A4 and CYP3A5[1]. Phenotypic evaluation of in vivo resistance development. [2] Phenotypic analyses of viral isolates from baseline (before the first dose was administered) and at the end of 3 days of ABT-450/r monotherapy were performed in order to characterize the selection of resistant variants. The development of phenotypic resistance to ABT-450 during 3 days of dosing was assessed by calculating the fold change in EC50 at the end of the 3-day monotherapy compared to baseline (Table 4). Thirteen of the 24 patients (12 of 19 infected with genotype 1a and 1 of 5 infected with genotype 1b) had a viral load level sufficient (≥500 IU/ml) to allow amplification of the target gene at the end of the 3-day dosing period (5, 3, and 5 patients in the ABT-450/r 200/100-, 100/100-, and 50/100-mg treatment groups, respectively). [2] Administration of 30 mg/kg paritaprevir for 3 days protected rats from LPS-induced ALI, as reflected by the changes in the lung coefficient (from 0.75 to 0.64) and lung pathology scores (from 5.17 to 5.20). Furthermore, the levels of the protective adhesion protein VE-cadherin and tight junction protein claudin-5 increased, and the cytoplasmic p-FOX-O1 and nuclear β-catenin and FOX-O1 levels decreased[3]. 1. In a 3-day monotherapy clinical study (Phase II) of HCV genotype 1-infected patients, Paritaprevir (ABT-450) was coadministered with ritonavir (a CYP3A4 inhibitor); a mean maximum HCV RNA decline of 4.02 log10 was observed at the end of the 3-day dosing period across all doses [2] 2. In phase II/III clinical trials, the fixed-dose combination of Paritaprevir/ombitasvir/ritonavir plus dasabuvir (± ribavirin) achieved high rates of sustained virological response (SVR12) in adults with chronic HCV genotype 1 infection (including 1a/1b, compensated cirrhosis, liver transplant recipients, HIV-1 co-infection) [1] 3. Resistant variants selected in Paritaprevir (ABT-450)-treated patients were R155K and D168V (genotype 1a) and D168V (genotype 1b); selection of resistant variants was significantly reduced at the highest Paritaprevir dose compared to lower doses [2] |
| Enzyme Assay |
Paritaprevir exhibits antiviral activity in vitro against HCV GT1-4 and GT6 (EC50 range: 0.09 to 19 nM), while its EC50 against GT4a is 0.09 nM. Antiviral activity against a panel of resistant mutants. [2] The 1a-H77 and 1b-Con1 subgenomic replicon shuttle vector constructs used for introduction of mutations of interest in the NS3 gene were similar to the replicon cell line constructs described above, but in both cases the Neo gene was not present, and the HCV NS2 gene was inserted between the EMCV IRES and the NS3 gene (Fig. 2B). In addition, the 1a-H77 replicon construct had the adaptive mutation in NS3 protease encoding E1202G replaced with one encoding P1496L in NS3 helicase. An AscI restriction site was introduced into the NS2 gene 62 nucleotides upstream of the 5′ end of the NS3 gene, and a BstBI restriction site was introduced within the helicase domain of NS3 after the NS3 amino acid 251 codon. The introduction of these restriction sites did not result in an amino acid insertion or change in either the genotype 1a or 1b replicon. Mutations encoding resistance-associated variants were introduced by site-directed mutagenesis and confirmed by sequence analysis. Subgenomic replicon RNA was generated by linearization of plasmid DNA followed by in vitro transcription. Replicon RNA was transfected into Huh7-derived cells, and inhibition of replication of the HCV replicon by ABT-450 was measured using the luciferase assay as described above, except that cells were incubated for 4 days rather than 3 days prior to lysis. Replication efficiency was calculated as a percentage of wild-type replication using the following equation: 100 ×[(mutant 4-day luciferase activity/wild-type 4-day luciferase activity)/(mutant 4-h luciferase activity/wild-type 4-h luciferase activity)][2]. 1. HCV NS3/4A protease inhibition assay: Stable HCV replicon cell lines (with NS3 protease from genotypes 1a, 1b, 2a, 3a, 4a, 6a) and transient replicon assays were used to evaluate the inhibitory activity of Paritaprevir (ABT-450); the 50% effective concentration (EC50) values for each genotype were determined by measuring viral replication inhibition at different drug concentrations [2] 2. Resistance profile assay of Paritaprevir: In vitro resistance selection experiments were conducted on genotype 1 HCV replicons treated with Paritaprevir (ABT-450); amino acid variations in NS3 protease were identified, and the resistance level of key variants (e.g., D168Y) was quantified by measuring EC50 fold changes [2] |
| Cell Assay |
Antiviral activity in cell culture.[2] Replicon cell lines were maintained in Dulbecco's modified Eagle medium (DMEM) supplemented with 100 IU/ml penicillin, 100 μg/ml streptomycin, and 200 μg/ml G418, all of which were from Invitrogen, as well as 10% (vol/vol) fetal bovine serum (FBS). The inhibitory effect of ABT-450 was evaluated by incubating replicon-containing cells in the presence of a series of ABT-450 dilutions for 3 days in the same medium containing 5% FBS, followed by measurement of firefly luciferase activity using the luciferase assay system. In assays measuring inhibitory activity in the presence of human plasma, the medium contained 40% human plasma and 5% FBS. The percent inhibition of HCV RNA replication was calculated for each compound concentration, and the 50% effective concentration (EC50) was calculated using nonlinear regression sigmoidal dose-response variable slope curve fitting to the 4-parameter logistic equation and GraphPad Prism 4 software. The cytotoxicity of ABT-450 was determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide colorimetric assay. The 50% cytotoxicity concentration (CC50) was calculated using nonlinear regression sigmoidal dose-response variable slope curve fitting as described above. 1. HCV replicon cell assay: Stable HCV replicon cell lines expressing NS3 protease from different genotypes (1a, 1b, 2a, 3a, 4a, 6a) were cultured with different concentrations of Paritaprevir (ABT-450); viral replication levels were detected to calculate EC50 values; transient replicon assays were also used to confirm the inhibitory activity [2] 2. Resistance selection cell assay: Genotype 1 HCV replicon cells were exposed to Paritaprevir (ABT-450) to induce resistant variants; NS3 protease gene sequencing was performed to identify amino acid mutations, and the replication capacity of variant-containing replicons was assessed to determine resistance levels [2] |
| Animal Protocol |
In this study, 75 patients meeting all eligibility criteria and none of the exclusion criteria were randomized to receive various doses of ABT-450/r, dasabuvir, or ABT-072. Only data from the 24 patients treated with ABT-450/r are discussed in this report. Eligibility criteria for study M11-602 included the following: age of 18 to 65 years, body mass index (BMI) of ≥18 and <35 kg/m2, chronic HCV genotype 1 infection for at least 6 months prior to study enrollment, plasma HCV RNA level of ≥100,000 IU/ml at screening, liver biopsy within the past 3 years with histology consistent with HCV-induced liver damage, and no evidence of cirrhosis. Exclusion criteria included the following: liver biopsy with a METAVIR fibrosis score of 3 or 4, positive test result for hepatitis B surface antigen or anti-HIV antibodies, history of major depression within the 2 years prior to enrollment, history of disease precluding the use of pegIFN or RBV, and unresolved clinically significant diseases other than HCV.[2] Patients were randomized to receive 1 of 3 doses of ABT-450/r (50/100 mg, 100/100 mg, or 200/100 mg) or placebo once daily (QD). Following 3 days of monotherapy, pegIFN alfa-2a at 180 μg/week and weight-based RBV at 1,000 to 1,200 mg/day were added, and the same dose of ABT-450/r or placebo was continued to complete a total of 12 weeks. At week 12, ABT-450/r or placebo was discontinued, and patients received pegIFN/RBV alone for up to 36 additional weeks.[2] 1. Clinical trial protocol (3-day monotherapy): HCV genotype 1-infected patients received Paritaprevir (ABT-450) coadministered with ritonavir at different doses for 3 days; plasma trough concentrations of Paritaprevir were measured, and HCV RNA levels were monitored to evaluate antiviral efficacy and resistance selection [2] 2. Phase II/III clinical trial protocol: Adults with chronic HCV genotype 1 infection were treated with Paritaprevir/ombitasvir/ritonavir (fixed-dose tablet) plus dasabuvir (± ribavirin); SVR12 (sustained virological response 12 weeks post-treatment) was the primary efficacy endpoint, and safety/tolerability was monitored [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Tmax of approximately 4 to 5 hours with a maximum concentration (Cmax) of 194 ng/mL. Following a single dose administration of 14C-paritaprevir co-dosed with 100 mg of ritonavir, approximately 88% of the radioactivity was recovered in feces with limited radioactivity (8.8%) in urine; unchanged paritaprevir accounted for 1.1% of the radioactivity in the feces and 0.05% in the urine. Volume of distribution at steady state is approximately 103 L. Metabolism / Metabolites Paritaprevir is predominantly metabolized by CYP3A4 and to a lesser extent by CYP3A5. Biological Half-Life 5.5 hr 1. Ritonavir (a cytochrome P450 3A4 inhibitor) markedly increased peak, trough, and overall plasma exposures of Paritaprevir (ABT-450) when coadministered [2] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Paritaprevir has not been studied in nursing mothers being treated for hepatitis C infection. Because it is greater than 97% bound to maternal plasma proteins, amounts in breastmilk are likely to be very low. Some sources recommend against breastfeeding when paritaprevir is used with ribavirin. Ritonavir used as a booster has been studied in several studies of breastfeeding mothers. It is excreted into milk in measurable concentrations and low levels can be found in the blood of some breastfed infants. No reports of adverse reactions in breastfed infants have been reported. For more information, refer to the LactMed record on ritonavir. Hepatitis C is not transmitted through breastmilk and breastmilk has been shown to inactivate hepatitis C virus (HCV). However, the Centers for Disease Control recommends that mothers with HCV infection should consider abstaining from breastfeeding if their nipples are cracked or bleeding. It is not clear if this warning would apply to mothers who are being treated for hepatitis C. Infants born to mothers with HCV infection should be tested for HCV infection; because maternal antibody is present for the first 18 months of life and before the infant mounts an immunologic response, nucleic acid testing is recommended. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding 97 to 98.6% bound to human plasma proteins. 1. The combination regimen of Paritaprevir/ombitasvir/ritonavir plus dasabuvir was generally well tolerated in clinical trials; the most common adverse events included nausea, insomnia, asthenia, pruritus, other skin reactions, and fatigue [1] |
| References |
[1]. Drugs . 2015 Jun;75(9):1027-38. [2].Antimicrob Agents Chemother . 2015 Feb;59(2):988-97. [3]. Arch Pharm Res. 2023 Jun;46(6):564-572. |
| Additional Infomation |
Paritaprevir is a direct acting antiviral medication used as part of combination therapy to treat chronic Hepatitis C, an infectious liver disease caused by infection with Hepatitis C Virus (HCV). HCV is a single-stranded RNA virus that is categorized into nine distinct genotypes, with genotype 1 being the most common in the United States, and affecting 72% of all chronic HCV patients. Treatment options for chronic Hepatitis C have advanced significantly since 2011, with the development of Direct Acting Antivirals (DAAs) such as paritaprevir. As a newer generation and directly acting HCV antiviral, paritaprevir products have better Sustained Virological Response (SVR) rates, higher barriers to resistance, fewer side effects, and a reduced pill burden compared to older agents such as [DB08873], [DB05521], [DB00008], [DB00022], and [DB00811]. By combining multiple antiretroviral medications into fixed dose products, the viral lifecycle can be targeted at multiple stages while simultaneously reducing the risk of developing resistant viral strains. Within Canada and the United States, paritaprevir is currently available in three fixed dose products: Viekira Pak (FDA), Technivie (FDA and Health Canada), and Holkira Pak (Health Canada). More specifically, paritaprevir prevents viral replication by inhibiting the NS3/4A serine protease of Hepatitis C Virus (HCV). Following viral replication of HCV genetic material and translation into a single polypeptide, Nonstructural Protein 3 (NS3) and its activating cofactor Nonstructural Protein 4A (NS4A) are responsible for cleaving genetic material into the following structural and nonstructural proteins required for assembly into mature virus: NS3, NS4A, NS4B, NS5A, and NS5B. By inhibiting viral protease NS3/4A, paritaprevir therefore prevents viral replication and function. In a joint recommendation published in 2016, the American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America (IDSA) recommend Paritaprevir as a first line therapy option when used in combination with other antivirals for genotypes 1a, 1b, and 4. Depending on the genotype, Paritaprevir is often used in combination with other antivirals such as [DB09296], [DB09183], [DB00503], and [DB00811], with the intent to cure, or achieve a sustained virologic response (SVR), after 12 weeks of daily therapy. SVR and eradication of HCV infection is associated with significant long-term health benefits including reduced liver-related damage, improved quality of life, reduced incidence of Hepatocellular Carcinoma, and reduced all-cause mortality. Treatment with direct acting antivirals such as paritaprevir is associated with very minimal side effects, with the most common being headache and fatigue. Lack of significant side effects and short duration of therapy is a considerable advantage over older interferon-based regimens, which were limited by infusion site reactions, reduced blood count, and neuropsychiatric effects. Paritaprevir first came on the market as a fixed-dose combination product with [DB09296], [DB09183], and [DB00503] as the FDA-approved product Viekira Pak. First approved in December 2014, Viekira Pak is indicated for the treatment of HCV genotype 1b without cirrhosis or with compensated cirrhosis, and when combined with [DB00811] for the treatment of HCV genotype 1a without cirrhosis or with compensated cirrhosis. Paritaprevir is also available as a fixed-dose combination product with [DB09296] and [DB00503] as the FDA- and Health Canada-approved product Technivie. First approved in July 2015, Technivie is indicated in combination with [DB00811] for the treatment of patients with genotype 4 chronic hepatitis C virus (HCV) infection without cirrhosis or with compensated cirrhosis. In Canada, paritaprevir is also available as a fixed-dose combination product with [DB09296], [DB09183], and [DB00503] as the Health Canada-approved, commercially available product Holkira Pak. First approved in January 2015, Holkira Pak is indicated for the treatment of HCV genotype 1b with or without cirrhosis, and when combined with [DB00811] for the treatment of HCV genotype 1a with or without cirrhosis. Paritaprevir is a Hepatitis C Virus NS3/4A Protease Inhibitor. The mechanism of action of paritaprevir is as a HCV NS3/4A Protease Inhibitor, and Organic Anion Transporting Polypeptide 1B1 Inhibitor, and Organic Anion Transporting Polypeptide 1B3 Inhibitor, and Breast Cancer Resistance Protein Inhibitor, and UGT1A1 Inhibitor, and P-Glycoprotein Inhibitor. Paritaprevir is an orally bioavailable, synthetic acylsulfonamide inhibitor of the hepatitis C virus (HCV) protease complex comprised of non-structural protein 3 and 4A (NS3/NS4A), with potential activity against HCV genotype 1. Upon administration, paritaprevir reversibly binds to the active center and binding site of the HCV NS3/NS4A protease and prevents NS3/NS4A protease-mediated polyprotein maturation. This disrupts both the processing of viral proteins and the formation of the viral replication complex, which inhibits viral replication in HCV genotype 1-infected host cells. NS3, a serine protease, is essential for the proteolytic cleavage of multiple sites within the HCV polyprotein and plays a key role during HCV ribonucleic acid (RNA) replication. NS4A is an activating factor for NS3. HCV is a small, enveloped, single-stranded RNA virus belonging to the Flaviviridae family, and infection is associated with the development of hepatocellular carcinoma (HCC). See also: Ombitasvir; Paritaprevir; Ritonavir (component of) ... View More ... Drug Indication When used within the fixed-dose combination product with [DB09296], [DB09183], and [DB00503] as the FDA-approved product Viekira Pak, paritaprevir is indicated for the treatment of HCV genotype 1b without cirrhosis or with compensated cirrhosis, and when combined with [DB00811] for the treatment of HCV genotype 1a without cirrhosis or with compensated cirrhosis. When used within the fixed-dose combination product with [DB09296] and [DB00503] as the FDA- and Health Canada-approved product Technivie, paritaprevir is indicated in combination with [DB00811] for the treatment of patients with genotype 4 chronic hepatitis C virus (HCV) infection without cirrhosis or with compensated cirrhosis. When used within the fixed-dose combination product with [DB09296], [DB09183], and [DB00503] as the Health Canada-approved, commercially available product Holkira Pak, paritaprevir is indicated for the treatment of HCV genotype 1b with or without cirrhosis, and when combined with [DB00811] for the treatment of HCV genotype 1a with or without cirrhosis. FDA Label Mechanism of Action Paritaprevir is a potent inhibitor of the NS3/4A serine protease of Hepatitis C Virus (HCV). Following viral replication of HCV genetic material and translation into a single polypeptide, Nonstructural Protein 3 (NS3) and its activating cofactor Nonstructural Protein 4A (NS4A) are responsible for cleaving it into the following structural and nonstructural proteins required for assembly into mature virus: NS3, NS4A, NS4B, NS5A, and NS5B. By inhibiting viral protease NS3/4A, paritaprevir therefore prevents viral replication and function. 1. Paritaprevir (ABT-450) is an NS3/4A protease inhibitor (direct-acting antiviral agent, DAA) for the treatment of HCV infection; rapid emergence of drug resistance is a major challenge for DAAs, leading to potential cross-resistance within the same drug class [2] 2. Paritaprevir is formulated as a fixed-dose tablet with ombitasvir (NS5A replication complex inhibitor) and ritonavir (CYP3A4 inhibitor), and is used in combination with dasabuvir (NS5B polymerase inhibitor) for chronic HCV genotype 1 infection; the regimen is interferon-free and approved in the USA (Viekira Pak™) and EU (Viekirax® + Exviera®) [1] 3. The 3-day monotherapy study of Paritaprevir (ABT-450) provided insights for subsequent clinical trials of combination therapy with other DAAs in HCV-infected patients [2] 4. Paritaprevir-containing combination therapy is effective in a broad range of HCV genotype 1-infected populations, including those with compensated cirrhosis, liver transplants, or HIV-1 co-infection [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (2.72 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (2.72 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3057 mL | 6.5284 mL | 13.0569 mL | |
| 5 mM | 0.2611 mL | 1.3057 mL | 2.6114 mL | |
| 10 mM | 0.1306 mL | 0.6528 mL | 1.3057 mL |