Pantoprazole Sodium hydrate (BY1023; SKF96022; Protonix) is an anti-ulcer drug acting as a proton pump inhibitor (PPI) and is used for short-term treatment of erosion and ulceration of the esophagus caused by gastroesophageal reflux disease. Pantoprazole inhibits the activity of H+/K+-ATPase proton pumb in the parietal cells of gastric mucosa. This inhibition affects the acid secretion and thus, pantoprazole are used as drugs for the treatment of various acid-related disorders. Pantoprazole is activated slowly. The activated sulfonamide of pantoprazole binds to Cys813 and Cys822 of the pumb and inhibits acid secretion selectively.
Physicochemical Properties
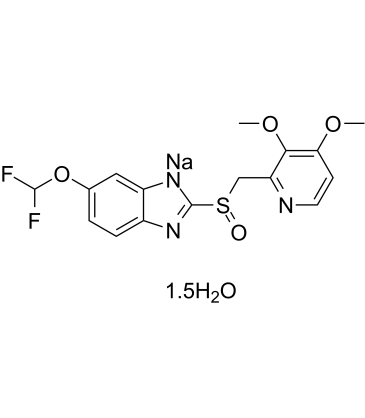
| Molecular Formula | 2(C16H14F2N3NAO4S).3(H2O) |
| Molecular Weight | 864.75 |
| Exact Mass | 864.145 |
| CAS # | 164579-32-2 |
| Related CAS # | Pantoprazole;102625-70-7;Pantoprazole sodium;138786-67-1;S-Pantoprazole sodium trihydrate;1416988-58-3 |
| PubChem CID | 4679 |
| Appearance | Off-white solid |
| Melting Point |
149-150 139-140 °C, decomposes Mol wt: 405.36. White to off-white solid; mp: >130 °C (dec); UV max (methanol): 289 (E=1.64X10+4) /Sodium salt/ |
| LogP | 2.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 26 |
| Complexity | 490 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | VNKNFEINTHUQGZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/2C16H14F2N3O4S.2Na.3H2O/c2*1-23-13-5-6-19-12(14(13)24-2)8-26(22)16-20-10-4-3-9(25-15(17)18)7-11(10)21-16;;;;;/h2*3-7,15H,8H2,1-2H3;;;3*1H2/q2*-1;2*+1;;; |
| Chemical Name | disodium;5-(difluoromethoxy)-2-[(3,4-dimethoxypyridin-2-yl)methylsulfinyl]benzimidazol-1-ide;trihydrate |
| Synonyms | BY1023 sodium hydrate; SKF96022 sodium hydrate; BY1023; SKF96022; Protonix; BY 1023; BY-1023; SKF 96022; Pantoprazole sodium sesquihydrate; 164579-32-2; Protonix; Pantoprazole Sodium [USAN]; Somac Control; disodium;5-(difluoromethoxy)-2-[(3,4-dimethoxypyridin-2-yl)methylsulfinyl]benzimidazol-1-ide;trihydrate; Pantoloc Control; SKF-96022 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Proton pump In EMT-6 and MCF7 cells, pantoprazole sodium hydrate (BY1023 sodium hydrate; 1–10,000 μM) raises endosomal pH in a concentration-dependent manner [1]. Pantoprazole sodium hydrate prevents exosome release. Pantoprazole sodium hydrate prevents tumor cells (melanoma, adenocarcinoma, and lymphoma cell lines) from acidifying the extracellular medium by inhibiting V-H+-ATPase activity [2]. |
| ln Vitro |
In EMT-6 and MCF7 cells, pantoprazole sodium hydrate (BY1023 sodium hydrate; 1–10,000 μM) raises endosomal pH in a concentration-dependent manner [1]. Pantoprazole sodium hydrate prevents exosome release. Pantoprazole sodium hydrate prevents tumor cells (melanoma, adenocarcinoma, and lymphoma cell lines) from acidifying the extracellular medium by inhibiting V-H+-ATPase activity [2]. Pantoprazole, at concentrations above 200 µmol/L, increased endosomal pH in both murine EMT-6 and human MCF-7 cancer cells. The effect was concentration-dependent and was enhanced in the presence of doxorubicin. [1] Pretreatment with pantoprazole (1 mmol/L) altered the intracellular distribution of doxorubicin in MCF-7 and EMT-6 cells, reducing doxorubicin fluorescence in punctuate cytoplasmic compartments (indicative of endosomal sequestration) while retaining it in the nucleus. [1] Flow cytometry analysis showed that pretreatment with pantoprazole (1 mmol/L) significantly increased the net cellular uptake of doxorubicin in EMT-6 cells but decreased it by 24% in MCF-7 cells and 36% in A431 cells. At a lower concentration (100 µmol/L), similar trends were observed but were not statistically significant. [1] Pretreatment of multilayered cell cultures (MCCs) derived from EMT-6 and MCF-7 cells with pantoprazole (1 mmol/L) significantly increased the penetration of doxorubicin through the tissue. The increase was more than 2-fold in EMT-6 MCCs and approximately 1.3-fold in MCF-7 MCCs. Fluorescence microscopy confirmed increased doxorubicin signal in cells distal from the drug source in pretreated MCCs. [1] |
| ln Vivo |
In MCF-7 xenografts, the combination of pantoprazole sodium hydrate (BY1023 sodium hydrate; 200 mg/kg; IP; once weekly for 3 weeks) and doxorubicin significantly prolonged the tumor growth delay [1]. Oral pantoprazole sodium hydrate (0.3–3 mg/kg) decreases mepizole-induced stimulated acid secretion in acute fistula rats and basal acid secretion in pyloric ligation rats in a dose-dependent manner [4]. In mice bearing MCF-7 xenograft tumors, pretreatment with pantoprazole (200 mg/kg, i.p.) 2 hours before doxorubicin administration led to a significant increase in doxorubicin fluorescence within the tumor tissue compared to doxorubicin alone, indicating improved drug distribution. Quantitative analysis showed a shallower gradient of decreasing doxorubicin intensity with distance from functional blood vessels in the pantoprazole-pretreated group. This effect was not observed in EMT-6 tumors. [1] Pretreatment with a single dose of pantoprazole (200 mg/kg, i.p.) before a single dose of doxorubicin (8 mg/kg, i.v.) significantly increased tumor growth delay in MCF-7 xenografts compared to doxorubicin alone or control groups. Pantoprazole alone had no effect on tumor growth. [1] In A431 xenograft tumors (relatively resistant to doxorubicin), pretreatment with pantoprazole before a single dose of doxorubicin resulted in a small, marginally significant increase in growth delay. However, when administered weekly for three weeks in combination with doxorubicin, no significant growth delay was observed in this model. [1] Weekly administration of pantoprazole (200 mg/kg, i.p.) before doxorubicin (6 mg/kg, i.v.) for three weeks led to an even greater growth delay in MCF-7 xenografts compared to the single-dose combination regimen. [1] |
| Enzyme Assay | The action of the H+/K(+)-ATPase inhibitors pantoprazole and omeprazole was compared in different in vitro test systems. In gastric membrane vesicles under conditions shown to result in acidification of the vesicle interior, pantoprazole and omeprazole inhibited H+/K(+)-ATPase activity with IC50 values of 6.8 and 2.4 microM, respectively. When intravesicular acidification was reduced by inclusion of imidazole (5 mM), a membrane permeable weak base, the inhibitory action of omeprazole was partially lost (IC50 30 microM) and that of pantoprazole almost completely lost. After incubation for 40 min with pumping membrane vesicles, a half-maximal reduction in intravesicular H+ concentration occurred at pantoprazole and omeprazole concentrations of 1.1 and 0.6 microM, respectively. Again, when the intravesicular H+ concentration was reduced by inclusion of imidazole (2.5 mM), pantoprazole (20 and 60 microM) did not reduce the remaining intravesicular proton concentration, whereas omeprazole (10 and 30 microM) did. Both drugs inhibited, with similar potency, papain activity at pH 3.0 and inactivated the enzyme in a similar time-dependent manner; at pH 5.0 omeprazole (IC50 17 microM) was more potent than pantoprazole (IC50 37 microM) and enzyme inhibition was faster than with pantoprazole. These results indicate that pantoprazole is a potent inhibitor of H+/K(+)-ATPase under highly acidic conditions and that it is more stable than omeprazole at a slightly acidic pH such as pH 5.0[3]. |
| Cell Assay |
Murine EMT-6 and human MCF-7 cells were treated with pantoprazole to evaluate changes in endosomal pH using fluorescence spectroscopy, and uptake of doxorubicin using flow cytometry. Effects of pantoprazole on tissue penetration of doxorubicin were evaluated in multilayered cell cultures (MCC). Pantoprazole (>200 μmol/L) increased endosomal pH in cells, and also increased nuclear uptake of doxorubicin. Pretreatment with pantoprazole increased tissue penetration of doxorubicin in MCCs [1]. To measure endosomal pH, EMT-6 or MCF-7 cells were treated with varying concentrations of pantoprazole (and other agents for comparison) for 3 hours in the presence of a fluorescent pH-sensitive dextran conjugate (FITC/TMR-dextran) that is taken up into endosomes. After a 2-hour incubation in media, fluorescence was measured using flow cytometry. The ratio of pH-dependent (FITC) to pH-independent (TMR) fluorescence was calibrated using the ionophore nigericin in buffers of known pH to determine endosomal pH. [1] To evaluate doxorubicin distribution within cells, cells grown on chambered cover glass were pretreated with pantoprazole (1 mmol/L) for 2 hours, then incubated with media containing doxorubicin (2 µg/mL) for 1 hour. After washing, intracellular doxorubicin fluorescence was visualized using fluorescence microscopy with specific excitation/emission filters. To visualize endosomes, cells were co-stained with the pH-sensitive dye LysoSensor Yellow/Blue. [1] To measure net cellular uptake of doxorubicin by flow cytometry, cells were treated with saline or pantoprazole (1 mmol/L) for 2 hours, followed by incubation with doxorubicin (1.8 µmol/L) for 1 hour. Cells were washed, and mean doxorubicin fluorescence was measured using a flow cytometer equipped with a 530 nm emission filter. [1] To assess drug penetration in an in vitro tumor-like environment, multilayered cell cultures (MCCs) were established by growing EMT-6 or MCF-7 cells on collagen-coated membranes for 6-8 days. MCCs were pretreated with or without pantoprazole (1 mmol/L) for 2 hours. The penetration of radiolabeled doxorubicin (10 µmol/L, mixed with agar to prevent convection) through the MCC into a receiving compartment was monitored over time by liquid scintillation counting. The penetration of a non-cell-penetrating tracer (³H-sucrose) was used as an internal standard to control for MCC thickness variations. Parallel MCCs were exposed to doxorubicin and frozen sections were imaged by fluorescence microscopy to visualize drug distribution. [1] |
| Animal Protocol |
Animal/Disease Models: Mice bearing MCF-7 or A431 xenografts [1] Doses: 200 mg/kg Route of Administration: IP; once weekly for 3 weeks; alone or in combination with doxorubicin (6 mg/kg iv) First 2 hour Experimental Results: The growth delay of MCF-7 xenografts with doxorubicin was even greater compared to the single dose combination. A single dose of doxorubicin Dramatically increased tumor growth delay. alone had no effect on growth delay. For tumor growth delay studies, female athymic nude mice bearing subcutaneous MCF-7 or A431 xenografts (or syngeneic Balb/C mice bearing EMT-6 tumors) were used. When tumors reached 5-8 mm in diameter, mice were randomized into treatment groups. Pantoprazole was administered intraperitoneally (i.p.) at a dose of 200 mg/kg, dissolved in 0.9% saline. Doxorubicin was administered intravenously (i.v.) at a dose of 8 mg/kg (single-dose study) or 6 mg/kg (multiple-dose study). For combination therapy, pantoprazole was given i.p. 2 hours before doxorubicin injection. Tumor volumes and body weights were measured regularly until endpoints were reached. [1] For studying doxorubicin distribution in tumors, mice bearing EMT-6 or MCF-7 tumors (8-12 mm mean diameter) were treated. They received pantoprazole (200 mg/kg, i.p.) or vehicle 2 hours before a high dose of doxorubicin (25 mg/kg, i.v.) to facilitate fluorescence detection. To mark functional vasculature and hypoxia, mice also received intravenous DIOC7 (1 mg/kg) and intraperitoneal EF5 respectively, shortly before sacrifice. Mice were sacrificed 10 minutes after doxorubicin injection, tumors were excised, frozen, sectioned, and analyzed by fluorescence microscopy and immunohistochemistry for drug distribution relative to blood vessels. [1] For toxicity studies, mice were treated with pantoprazole at doses of 100, 150, 200, 250, or 300 mg/kg (i.p.), alone or 2 hours before doxorubicin (8 mg/kg, i.v.). Body weight was monitored every other day. [1] For pharmacokinetic analysis, Balb/C mice were treated with a single i.p. dose of pantoprazole (200 mg/kg). Blood was collected via cardiac puncture at various time points, plasma was isolated, and pantoprazole concentration was determined using a validated HPLC-MS/MS method. [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Pantoprazole is absorbed after oral administration as an enteric-coated tablet with maximum plasma concentrations attained within 2 – 3 hours and a bioavailability of 77% that does not change with multiple dosing. Following an oral dose of 40mg, the Cmax is approximately 2.5 μg/mL with a tmax of 2 to 3 hours. The AUC is approximately 5 μg.h/mL. There is no food effect on AUC (bioavailability) and Cmax. Delayed-release tablets are prepared as enteric-coated tablets so that absorption of pantoprazole begins only after the tablet leaves the stomach. After a single oral or intravenous (IV) dose of 14C-labeled pantoprazole to healthy, normal metabolizing subjects, about 71% of the dose was excreted in the urine, with 18% excreted in the feces by biliary excretion. There was no kidney excretion of unchanged pantoprazole. The apparent volume of distribution of pantoprazole is approximately 11.0-23.6 L, distributing mainly in the extracellular fluid. **Adults**: With intravenous administration of pantoprazole to extensive metabolizers, total clearance is 7.6-14.0 L/h. In a population pharmacokinetic analysis, the total clearance increased with increasing body weight in a non-linear fashion. **Children**: clearance values in the children 1 to 5 years old with endoscopically proven GERD had a median value of 2.4 L/h. Time to peak concentration: Following an oral dose of 40 mg in extensive metabolizers with normal hepatic function: 2.4 hours. When pantoprazole is taken with food, the time to peak concentration is variable and may be significantly increased. /Pantoprazole sodium/ Peak serum concentration: Following an oral dose of 40 mg in extensive metabolizers with normal hepatic function: 2.4 ug/mL. Following an intravenous dose of 40 mg administered over 15 minutes to extensive metabolizers with normal hepatic function: 5.51 ug/mL. /Pantoprazole sodium/ Elimination: Renal: 71%. Fecal: 18% (biliary excretion). Dialysis removes insignificant amounts of pantoprazole. /Pantoprazole sodium/ Rapidly absorbed. However, absorption maybe delayed up to 2 hours or more if pantoprazole is taken with food. Bioavailability (oral): 77%. /Pantoprazole sodium/ For more Absorption, Distribution and Excretion (Complete) data for PANTOPRAZOLE (6 total), please visit the HSDB record page. Metabolism / Metabolites Pantoprazole is heavily metabolized in the liver by the cytochrome P450 (CYP) system. Pantoprazole metabolism is independent of the route of administration (intravenous or oral). The main metabolic pathway is _demethylation_, by _CYP2C19_ hepatic cytochrome enzyme, followed by sulfation; other metabolic pathways include oxidation by CYP3A4. There is no evidence that any of the pantoprazole metabolites are pharmacologically active. After hepatic metabolism, almost 80% of an oral or intravenous dose is excreted as metabolites in urine; the remainder is found in feces and originates from biliary secretion. Pantoprazole is extensively metabolized in the liver through the cytochrome P450 (CYP) system. Pantoprazole metabolism is independant of route of administration (intravenous or oral). The main metabolic pathway is demethylation,by CYP2C19, with subsequent sulfation; other metabolic pathways include oxidation by CYP3A4. ... CYP2C19 displays a known genetic polymorphism due to its deficiency in some sub-populations (eg 3% of Caucasians and African-Americans and 17 to 23% of Asians). /Pantoprazole sodium/ Biological Half-Life About 1 hour Elimination: Following oral or intravenous administration: 1 hour. The half-life of pantoprazole is prolonged (7 to 9 hours) in patients with cirrhosis of the liver and in genetically determined slow metabolizers (3.5 to 10 hours). /Pantoprazole sodium/ Following a single intraperitoneal injection of pantoprazole (200 mg/kg) in mice, the peak plasma concentration was approximately 300 µmol/L, occurring within the first hour. [1] The plasma concentration declined to about 150 µmol/L at 2 hours post-injection. [1] By 5 hours post-injection, the plasma concentration had decreased to less than 1% of the peak concentration. [1] At 24 hours post-injection, less than 0.01 µmol/L of pantoprazole was detectable in plasma. [1] |
| Toxicity/Toxicokinetics |
Hepatotoxicity Despite its wide use, pantoprazole has only rarely been associated with hepatic injury. In large scale, long term trials of pantoprazole, serum ALT elevations have occurred in less than 1% of patients and at rates similar to those that occur with placebo or comparator drugs. Only a small number of cases of clinically apparent liver disease attributed to pantoprazole have been published, but the clinical pattern of injury has resembled acute hepatic necrosis which has been described with other proton pump inhibitors. Clinically apparent liver injury due to proton pump inhibitors generally arises within the first 4 weeks of therapy and is characterized by an acute hepatocellular pattern of injury with rapid recovery upon withdrawal. Rash, fever and eosinophilia are rare, as is autoantibody formation. In large case series of drug induced liver injury, pantoprazole has accounted for few instances of symptomatic acute liver injury. Likelihood score: C (probable rare cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Maternal pantoprazole doses of 40 mg daily produce low levels in milk and would not be expected to cause any adverse effects in breastfed infants. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk A retrospective claims database study in the United States found that users of proton pump inhibitors had an increased risk of gynecomastia. A review article reported that a search of database from the European Pharmacovigilance Centre found 48 cases of gynecomastia, 3 cases of galactorrhea, 14 cases of breast pain and 4 cases of breast enlargement associated with pantoprazole. A search of the WHO global pharmacovigilance database found 97 cases of gynecomastia, 13 cases of galactorrhea, 35 cases of breast pain and 16 cases of breast enlargement associated with pantoprazole. Protein Binding Approximately 98% Interactions Pantoprazole causes prolonged inhibition of gastric acid secretion, and thereby may interfere with the absorption of these medications /ampicillin or iron salts or ketoconazole/ and others for which bioavailability is determined by gastric pH. /Pantoprazole sodium/ Pantoprazole, although metabolized by hepatic cytochrome p450 systems, does not appear to either inhibit or induce cytochrome p450 enzyme activity. To date, no clinically significant interactions have been noted from such commonly used drugs as diazepam, phenytoin, nifedipine, theophylline, digoxin, warfarin, or oral contraceptives. /Pantoprazole sodium/ Pantoprazole, by increasing gastric pH, has the potential to affect the bioavailability of any medication for which absorption is pH-dependent. Also, pantoprazole may prevent the degradation of acid-labile drugs. /Pantoprazole sodium/ In other in vivo studies, ethanol, glyburide, caffeine, antipyrine, metronidazole, and amoxicillin, had no clinically relevant interactions with pantoprazole. /Pantoprazole sodium/ In mice, the maximum tolerated dose of pantoprazole when combined with doxorubicin (8 mg/kg) was determined to be 200 mg/kg. At this dose, combined treatment caused a temporary decrease in body weight (approximately 15%) within the first 5-8 days, followed by recovery. [1] At higher doses of pantoprazole (250 or 300 mg/kg) combined with doxorubicin, mice showed continual loss of body weight beyond 20 days. [1] Treatment with pantoprazole alone (up to 300 mg/kg) or doxorubicin alone (8 mg/kg) resulted in minimal increase in body weight, similar to saline controls. [1] |
| References |
[1]. Use of the proton pump inhibitor pantoprazole to modify the distribution and activity of doxorubicin: a potential strategy to improve the therapy of solid tumors. Clin Cancer Res. 2013 Dec 15;19(24):6766-76. [2]. Advances in the discovery of exosome inhibitors in cancer. J Enzyme Inhib Med Chem. 2020 Dec;35(1):1322-1330. [3]. Pantoprazole: a novel H+/K(+)-ATPase inhibitor with an improved pH stability. Eur J Pharmacol. 1992 Aug 6;218(2-3):265-71. [4]. Effects of pantoprazole, a novel H+/K+-ATPase inhibitor, on duodenal ulcerogenic and healing responses in rats: a comparative study with omeprazole and lansoprazole. J Gastroenterol Hepatol. 1999 Mar;14(3):251-7. |
| Additional Infomation |
Therapeutic Uses Pantoprazole delayed-release tablets are indicated for the short-term (up to 8 weeks) treatment of heartburn and other symptoms associated with gastroesophageal reflux disease (GERD). Pantoprazole for injection is indicated for the short-term (7 to 10 days) treatment of GERD in patients who are unable to continue taking pantoprazole delayed-release tablets. Pantoprazole for injection is not indicated for initial treatment of GERD. /Included in US product labeling/ /Pantoprazole sodium/ Pantoprazole is indicated for the prevention of relapse in patients with reflux esophagitis. /NOT included in US product labeling/ /Pantoprazole sodium/ Pantoprazole is indicated for short-term (up to 4 weeks) treatment for symptom relief and healing in patients with active duodenal ulcer. /NOT included in US product labeling/ /Pantoprazole sodium/ Pantoprazole, in combination with clarithromycin and either amoxicillin or metronidazole, is indicated for treatment of patients with an active duodenal ulcer who are Helicobacter pylori positive. /NOT included in US product labeling/ /Pantoprazole sodium/ For more Therapeutic Uses (Complete) data for PANTOPRAZOLE (6 total), please visit the HSDB record page. Drug Warnings Anaphylaxis has been reported with the use of IV pantoprazole sodium. Immediate medical intervention and drug discontinuance are required if anaphylaxis or other severe hypersensitivity reactions occurs. /Pantoprazole sodium/ Adverse effects occurring in more than 1% of patients receiving oral pantoprazole for up to 8 weeks and more frequently than in those receiving placebo include diarrhea and hyperglycemia. Adverse effects occurring in 1% or more of patients receiving oral pantoprazole for up to 12 months and more frequently than in those receiving ranitidine include headache, abdominal pain, and abnormal liver function test results. Adverse effects occurring in 4% or more of patients receiving IV pantoprazole and that were possibly, probably, or definitely related to treatment include abdominal pain, chest pain, rash, and pruritus. Adverse effects occurring in more than 1% of patients receiving IV pantoprazole and that generally had an unclear relationship to the drug include headache, injection site reaction, dyspepsia, diarrhea, vomiting, dizziness, and rhinitis. /Pantoprazole sodium/ There have been spontaneous reports of adverse events; angioedema (Quincke's edema); anterior ischemic optic neuropathy; severe dermatologic reactions, including erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis (TEN, some fatal); hepatocellular damage leading to jantice and and hepatic failure; pancreatitis; pancytopenia; and rhabdomyolysis. In addition, also observed have been confusion, hypokinesia, speech disorder, increased salivation, vertigo, nausea, tinnitus, and blurred vision. /Pantoprazole sodium/ FDA Pregnancy Risk Category: B /NO EVIDENCE OF RISK IN HUMANS. Adequate, well controlled studies in pregnant women have not shown increased risk of fetal abnormalities despite adverse findings in animals, or, in the absence of adequate human studies, animal studies show no fetal risk. The chance of fetal harm is remote but remains a possibility./ /Pantoprazole sodium/ For more Drug Warnings (Complete) data for PANTOPRAZOLE (8 total), please visit the HSDB record page. Pharmacodynamics This drug acts to decrease gastric acid secretion, which reduces stomach acidity. Pantoprazole administration leads to long-lasting inhibition of gastric acid secretion. **General Effects** Pantoprazole has been shown to reduce acid reflux-related symptoms, heal inflammation of the esophagus, and improve patient quality of life more effectively than histamine-2 receptor antagonists (H2 blockers). This drug has an excellent safety profile and a low incidence of drug interactions. It can be used safely in various high-risk patient populations, including the elderly and those with renal failure or moderate hepatic dysfunction. Due to their good safety profile and as several PPIs are available over the counter without a prescription, their current use in North America is widespread. Long term use of PPIs such as pantoprazole have been associated with possible adverse effects, however, including increased susceptibility to bacterial infections (including gastrointestinal _C. difficile_), reduced absorption of micronutrients including iron and B12, and an increased risk of developing hypomagnesemia and hypocalcemia which may contribute to osteoporosis and bone fractures later in life. PPIs such as pantoprazole have also been shown to inhibit the activity of dimethylarginine dimethylaminohydrolase (DDAH), an enzyme necessary for cardiovascular health. DDAH inhibition causes a consequent accumulation of the nitric oxide synthase inhibitor asymmetric dimethylarginie (ADMA), which is thought to cause the association of PPIs with increased risk of cardiovascular events in patients with unstable coronary syndromes. **A note on laboratory testing abnormalities** During treatment with antisecretory medicinal products such as pantoprazole, serum gastrin (a peptide hormone that stimulates secretion of gastric acid) increases in response to the decreased acid secretion caused by proton pump inhibition. The increased gastrin level may interfere with investigations for neuroendocrine tumors. Published evidence suggests that proton pump inhibitors should be stopped 14 days before chromogranin A (CgA) measurements. This permits chromogranin A levels, that might be falsely elevated after proton pump inhibitor treatment, to return to the normal reference range. Reports have been made of false-positive results in urine screening tests for tetrahydrocannabinol (THC) in patients receiving the majority of proton pump inhibitors, including pantoprazole. A confirmatory method should be used. Pantoprazole is a proton pump inhibitor (PPI) that inhibits vacuolar H⁺-ATPase (V-H⁺-ATPase). In the context of this cancer research, it is investigated not for its anti-acid effects but as a modulator of the tumor microenvironment. By increasing the pH of acidic intracellular compartments (like endosomes and lysosomes), it can inhibit the sequestration of weakly basic chemotherapeutic drugs such as doxorubicin. This mechanism is proposed to increase the availability of the drug for its nuclear target and to enhance its distribution from blood vessels into deeper tumor tissue, potentially overcoming a mechanism of drug resistance related to poor drug penetration. [1] The study concluded that pretreatment with pantoprazole could improve the therapeutic index of doxorubicin in some solid tumors by enhancing its distribution and cytotoxicity. The preclinical findings contributed to the initiation of clinical trials evaluating high-dose pantoprazole in combination with chemotherapy. [1] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~250 mg/mL (~578.21 mM) DMSO : ~100 mg/mL (~231.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.78 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1564 mL | 5.7820 mL | 11.5640 mL | |
| 5 mM | 0.2313 mL | 1.1564 mL | 2.3128 mL | |
| 10 mM | 0.1156 mL | 0.5782 mL | 1.1564 mL |