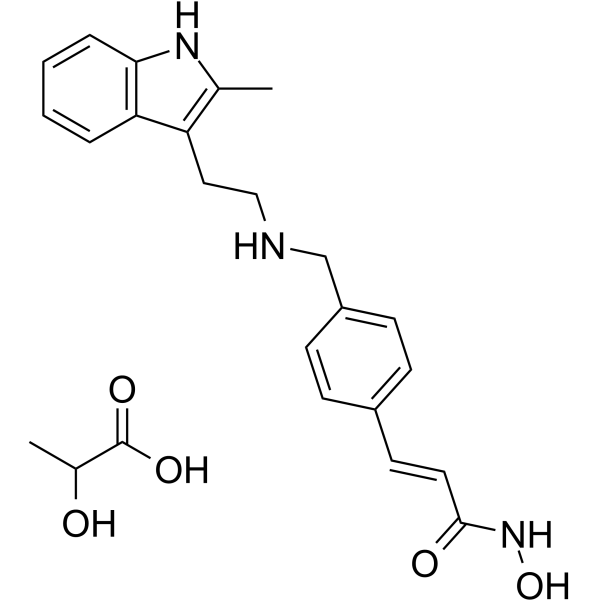
Panobinostat lactate, the lactate salt of Panobinostat (LBH-589; NVP LBH-589; Farydak), which is a broad-spectrum/non-selective/pan-HDAC inhibitor with potential anticancer activity. It inhibits HDAC with IC50 of 5 nM in a cell-free assay. It not only induces apoptosis in multiple myeloma cells via caspase activation and poly(ADP-ribose) polymerase (PARP) cleavage, but also induces potent cell growth inhibition, cell-cycle arrest, and apoptosis in a time- and dose-dependent manner in both Philadelphia chromosome-negative (Ph-) actue lymphoblastic leukemia (ALL) cells lines, which are correlated with induction of histone (H3K9 and H4K8) hyperacetylation, activation of p21 and p27, and suppression of c-Myc. In February 2015, Panobinostat received FDA approval for treating patients with multiple myeloma who had received at least 2 previous treatments, including bortezomib and an immunomodulatory agent.
Physicochemical Properties
| Molecular Formula | C24H29N3O5 |
| Molecular Weight | 439.50416636467 |
| Exact Mass | 439.211 |
| CAS # | 960055-56-5 |
| Related CAS # | Panobinostat;404950-80-7 |
| PubChem CID | 23725423 |
| Appearance | White to off-white solid powder |
| LogP | 4.01 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 32 |
| Complexity | 533 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(/C=C/C1C=CC(=CC=1)CNCCC1=C(C)NC2C=CC=CC1=2)NO.OC(C(=O)O)C |
| InChi Key | XVDWNSFFSMWXJJ-ASTDGNLGSA-N |
| InChi Code | InChI=1S/C21H23N3O2.C3H6O3/c1-15-18(19-4-2-3-5-20(19)23-15)12-13-22-14-17-8-6-16(7-9-17)10-11-21(25)24-26;1-2(4)3(5)6/h2-11,22-23,26H,12-14H2,1H3,(H,24,25);2,4H,1H3,(H,5,6)/b11-10+; |
| Chemical Name | (E)-N-hydroxy-3-[4-[[2-(2-methyl-1H-indol-3-yl)ethylamino]methyl]phenyl]prop-2-enamide;2-hydroxypropanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Panobinostat lactate causes apoptosis in both MOLT-4 and Reh cells in a time and dose-dependent manner. Panobinostat lactate causes histone (H3K9 and H4K8) hyperacetylation and regulates cell-cycle regulatory genes in Reh cells[1]. shows significant antiproliferative action in human NSCLC cell lines, with IC50 values ranging from 5 to 100 nM [2]. | ||
| ln Vivo | In vivo models of tumor growth produced from Meso and NSCLC cells are markedly inhibited by panobinosta lactate (10, 20 mg/kg, ip). The acetylation of histones H3 and H4 in H69 human SCLC cells derived from SCID mice is significantly elevated by panobinosta lactate [2]. In a disseminated multiple myeloma mouse model, panobinostat lactate (5, 10 and 20 mg/kg ip) clearly shows benefits for lower tumor burden, greatly improves TTE, and minimizes bone density loss[3]. | ||
| Animal Protocol |
|
||
| References |
[1]. The novel histone deacetylase inhibitor, LBH589, induces expression of DNA damage response genes and apoptosis in Ph- acute lymphoblastic leukemia cells. Blood. 2008 May 15;111(10):5093-100. [2]. The HDAC inhibitor panobinostat (LBH589) inhibits mesothelioma and lung cancer cells in vitro and in vivo with particular efficacy for small cell lung cancer. Mol Cancer Ther. 2009 Aug;8(8):2221-31. [3]. In vitro and in vivo rationale for the triple combination of panobinostat (LBH589) and dexamethasone with either bortezomib or lenalidomide in multiple myeloma. Haematologica. 2010 May;95(5):794-803. [4]. Vorinostat, a pan-HDAC inhibitor, abrogates productive HPV-18 DNA amplification. Proc Natl Acad Sci U S A. 2018 Nov 20;115(47):E11138-E11147. [5]. Broad activation of latent HIV-1 in vivo. Nat Commun. 2016;7:12731. Published 2016 Sep 8. |
||
| Additional Infomation |
Panobinostat lactate is a lactate salt having panobinostat(1+) as the counterion. A histone deacetylase inhibitor used in combination with bortezomib and dexamethasone for the treatment of multiple myeloma. It has a role as an EC 3.5.1.98 (histone deacetylase) inhibitor, an antineoplastic agent and an angiogenesis modulating agent. It is a lactate salt and an organoammonium salt. It contains a panobinostat(1+). Panobinostat is a drug that was previously approved by the U.S. Food and Drug Administration (FDA) under the brand name Farydak for the treatment of a certain type of cancer.Panobinostatis currently being studied as an investigational drug as part of a strategy to cure HIV infection. As an investigational HIV therapy, panobinostat belongs to a group of drugs called latency-reversing agents. Panobinostat Lactate is the lactate form of panobinostat, a pan histone deacetylase (HDAC) inhibitor, with potential antineoplastic activity. Upon administration, panobinostat selectively targets, binds to and inhibits HDAC, which induces hyperacetylation of core histone proteins. The accumulation of highly acetylated histones leads to chromatin remodeling, an altered pattern of gene expression, inhibition of tumor oncogene transcription and the selective transcription of tumor suppressor genes. This results in the inhibition of tumor cell division and the induction of tumor cell apoptosis. HDAC, upregulated in many tumor cell types, is an enzyme family that deacetylates histone proteins. See also: Panobinostat (has active moiety). Drug Indication Farydak, in combination with bortezomib and dexamethasone, is indicated for the treatment of adult patients with relapsed and/or refractory multiple myeloma who have received at least two prior regimens including bortezomib and an immunomodulatory agent. Farydak, in combination with bortezomib and dexamethasone, is indicated for the treatment of adult patients with relapsed and/or refractory multiple myeloma who have received at least two prior regimens including bortezomib and an immunomodulatory agent. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2753 mL | 11.3766 mL | 22.7531 mL | |
| 5 mM | 0.4551 mL | 2.2753 mL | 4.5506 mL | |
| 10 mM | 0.2275 mL | 1.1377 mL | 2.2753 mL |