PT2385 (PT-2385) is a nove, potent, selective and orally bioactive inhibitor of hypoxia inducible factor (HIF-2α) with anticancer and anti-hepatic steatosis activity. It inhibits HIF-2α with luciferase EC50 of 27 nM and no significant off-target activity. PT2385 can allosterically binds to HIF-2α, thereby preventing HIF-2α heterodimerization and its subsequent binding to DNA. Human-intestine biopsies from individuals with or without obesity revealed that intestinal HIF-2α signaling was positively correlated with body-mass index and hepatic toxicity. The causality of this correlation was verified in mice with an intestine-specific disruption of Hif2a, in which high-fat-diet-induced hepatic steatosis and obesity were substantially lower as compared to control mice. PT2385 had preventive and therapeutic effects on metabolic disorders that were dependent on intestine HIF-2α. Intestine HIF-2α inhibition markedly reduced intestine and serum ceramide levels. Mechanistically, intestine HIF-2α regulates ceramide metabolism mainly from the salvage pathway, by positively regulating the expression of Neu3, the gene encoding neuraminidase 3. These results suggest that intestinal HIF-2α could be a viable target for hepatic steatosis therapy.
Physicochemical Properties
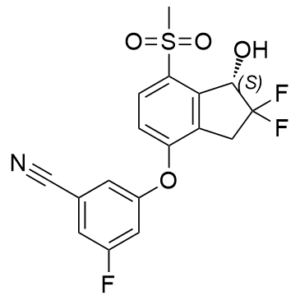
| Molecular Formula | C17H12F3NO4S | |
| Molecular Weight | 383.34 | |
| Exact Mass | 383.043 | |
| Elemental Analysis | C, 53.27; H, 3.16; F, 14.87; N, 3.65; O, 16.69; S, 8.36 | |
| CAS # | 1672665-49-4 | |
| Related CAS # |
|
|
| PubChem CID | 91754484 | |
| Appearance | White to off-white solid powder | |
| Density | 1.6±0.1 g/cm3 | |
| Boiling Point | 524.7±50.0 °C at 760 mmHg | |
| Flash Point | 271.1±30.1 °C | |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C | |
| Index of Refraction | 1.613 | |
| LogP | 1.8 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 26 | |
| Complexity | 686 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | S(C([H])([H])[H])(C1C([H])=C([H])C(=C2C([H])([H])C([C@]([H])(C2=1)O[H])(F)F)OC1=C([H])C(=C([H])C(C#N)=C1[H])F)(=O)=O |
|
| InChi Key | ONBSHRSJOPSEGS-INIZCTEOSA-N | |
| InChi Code | InChI=1S/C17H12F3NO4S/c1-26(23,24)14-3-2-13(12-7-17(19,20)16(22)15(12)14)25-11-5-9(8-21)4-10(18)6-11/h2-6,16,22H,7H2,1H3/t16-/m0/s1 | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
HIF-2α (Kd<50 nM)[1] HIF-2 is selectively antagonistic to PT-2385 (PT2385), but not HIF-1. HIF-1α is not inhibited by PT-2385[1]. |
| ln Vitro |
HIF-2 is selectively antagonistic to PT-2385 (PT2385), but not HIF-1. HIF-1α is not inhibited by PT-2385[1]. In the intestinal epithelial cell line HCT116, treatment with PT-2385 (10 μM) completely abolished the hypoxia-mimetic agent (CoCl₂)-mediated induction of NEU3 mRNA expression, a key enzyme in the ceramide salvage pathway and a direct target gene of HIF-2α. This was accompanied by decreased expression of the HIF-2α target genes DMT1 and DCYTB [2] Treatment of HCT116 cells with PT-2385 (10 μM) significantly blunted the hypoxia (CoCl₂)-mediated increase in cellular ceramide levels [2] |
| ln Vivo |
Tumor regression is accelerated and dose-dependent when PT-2385 (30 or 100 mg/kg; oral gavage; twice daily) is administered[3]. In vivo, PT-2385 (PT2385) exhibits dose-dependent inhibition of HIF-2α-regulated gene expression. In 786-O xenografts, tumor regression was achieved with PT-2385 (3 and 10 mg/kg, oral, twice daily dosages). Moreover, PT-2385 (1, 3 and 10 mg/kg) reduced the amounts of tumor-derived VEGFA protein. Treatment with 10 mg/kg of PT-2385 decreases angiogenesis (CD-31) and proliferation (Ki67) [1]. Preventive treatment: When administered orally (20 mg/kg, once daily) to C57BL/6N mice simultaneously with a high-fat diet (HFD) for 12 weeks, PT-2385 substantially prevented HFD-induced body weight gain and improved insulin sensitivity [2] Preventive treatment: Oral administration of PT-2385 (20 mg/kg, once daily) for 12 weeks to HFD-fed control mice (Hif2afl/fl) eliminated hepatic lipid accumulation, lowered liver weight and hepatic triglyceride/cholesterol content, and reduced serum alanine aminotransferase (ALT) levels. These beneficial effects were absent in mice with intestine-specific HIF-2α knockout (Hif2aΔIE), indicating that the action of PT-2385 was dependent on intestinal HIF-2α [2] Preventive treatment: In HFD-fed control mice (Hif2afl/fl), PT-2385 treatment markedly reduced ceramide levels in both the small intestine and serum, and suppressed the intestinal mRNA expression of genes involved in ceramide synthesis pathways (e.g., Degs2, Smpd1, Smpd3, Smpd4, Enpp7, Neu3, Glb1, Gba2). These effects were not observed in HFD-fed Hif2aΔIE mice [2] Therapeutic treatment: Oral administration of PT-2385 (20 mg/kg, once daily) for 4 weeks to C57BL/6N mice with established obesity and hepatic steatosis (after 8 weeks of HFD) reversed metabolic disorders. Treatment reduced body weight, improved insulin sensitivity, decreased hepatic lipid droplets (as shown by H&E and Oil Red O staining), lowered liver weight, hepatic triglyceride/cholesterol content, and serum ALT levels [2] Therapeutic treatment: In the therapeutic model, PT2385 treatment inhibited intestinal HIF-2α signaling (evidenced by decreased Dmt1 and Dcytb mRNA), reduced intestinal and serum ceramide levels, suppressed intestinal expression of ceramide synthesis-related genes (including Neu3), and decreased hepatic expression of genes involved in fatty acid synthesis, transport, and inflammation (e.g., Srebp1c, Cidea, Cd36, Fasn, Tnfa) [2] Western blot analysis confirmed that PT2385 treatment reduced NEU3 protein expression in the small intestine of HFD-fed mice [2] |
| Enzyme Assay |
The potent, selective, and orally active small-molecule inhibitor PT2385 as a specific antagonist of HIF2α that allosterically blocks its dimerization with the HIF1α/2α transcriptional dimerization partner ARNT/HIF1β. PT2385 inhibited the expression of HIF2α-dependent genes, including VEGF-A, PAI-1, and cyclin D1 in ccRCC cell lines[3]. Isothermal titration calorimetry: Human HIF2α-B was expressed and purified as described earlier. Rat HIF2α differs from human HIF2α-B by only three amino acids. So, the expression vector for human HIF2α-B was mutated at these residues (T262L, I 265V, and I326V) to make rat HIF2α-B. Pas-B domain of HIF1α was unstable, and the complex of HIF1α-B*:ARNT-B* was used for isothermal titration calorimetry (ITC). The complex was coexpressed in Escherichia coli harboring pET28-HIF1α-B* and pGB1-ARNT-B* and copurified in a single step of Ni-affinity chromatography. The binding affinity between PT2385 and PAS-B domains was determined using ITC on an iTC200 system. PAS-B at 0.4 mmol/L was titrated into 40 μmol/L of PT2385 in the cell in buffer consisting of 20 mmol/L Tris-HCl, pH 8.0, 150 mmol/L KCl, and 1% DMSO[3]. |
| Cell Assay |
786-O, A498, Hep3B, and Caki-1 cell lines were purchased from ATCC. All were acquired in 2012 and were maintained in culture for no more than 30 continuous passages. The cells were cultured in DMEM supplemented with 10% FBS, 100 units penicillin, and 100 μg/mL streptomycin.[3] For compound treatment, 5 × 105 cells were plated into 6-well cell culture plates in 2 mL of medium. Compound dissolved in DMSO was added as the cultures reached confluence with the final concentration of DMSO at 0.1%. For hypoxia-treated cells, cell cultures were placed in the chamber supplied with 1% oxygen and 5% CO2 for more then 4 hours before compound addition and maintained under the hypoxic condition for the duration of compound treatment.[3] All relevant human cell lines used in experiments were obtained from ATCC. ATCC authenticated cell lines with short tandem repeat profiling.[3] |
| Animal Protocol |
Animal/Disease Models: SCID/beige mice with 786-O and A498 RCC cell lines [3] Doses: 30 or 100 mg/kg Route of Administration: po (oral gavage); twice (two times) daily Experimental Results: Caused rapid, dose-dependent tumor regression . For both preventive and therapeutic efficacy studies in mice with diet-induced obesity/steatosis, PT-2385 was suspended in a vehicle containing saline with 0.5% sodium carboxymethyl cellulose, 2.5% Tween 80, and 2.5% dimethyl sulfoxide [2] The suspension was administered to mice by oral gavage once daily [2] In the main therapeutic study, C57BL/6N mice were first fed a high-fat diet (HFD, 60% kcal from fat) for 8 weeks to induce obesity and steatosis. Subsequently, these mice were treated with either vehicle or PT-2385 (20 mg/kg) by oral gavage once daily for an additional 4 weeks while continuing the HFD [2] In the preventive and mechanism-dependency study, control (Hif2afl/fl) and intestine-specific HIF-2α knockout (Hif2aΔIE) littermate mice were fed an HFD and simultaneously treated with vehicle or PT-2385 (20 mg/kg, once daily by oral gavage) for 12 weeks [2] In a short-term experiment to assess molecular effects, chow diet-fed Vhl/Hif1afl/fl and Vhl/Hif1aΔIE mice were treated with vehicle or PT-2385 (20 mg/kg, once daily by oral gavage) for 3 days [2] |
| References |
[1]. Eli Wallace, Ph.D. PT2385: HIF-2α Antagonist for the Treatment of VHL Mutant ccRCC. 12th International VHL Medical Symposium April 8, 2016. [2]. Activation of intestinal hypoxia-inducible factor 2α during obesity contributes to hepatic steatosis. Nat Med. 2017 Nov;23(11):1298-1308. [3]. A Small-Molecule Antagonist of HIF2α Is Efficacious in Preclinical Models of Renal Cell Carcinoma. Cancer Res. 2016 Sep 15;76(18):5491-500. |
| Additional Infomation |
PT-2385 is under investigation in clinical trial NCT03108066 (PT2385 for the Treatment of Von Hippel-lindau Disease-associated Clear Cell Renal Cell Carcinoma). HIF-2alpha Inhibitor PT2385 is an orally active, small molecule inhibitor of hypoxia inducible factor (HIF)-2alpha, with potential antineoplastic activity. Upon oral administration, HIF-2alpha inhibitor PT2385 allosterically binds to HIF-2alpha, thereby preventing HIF-2alpha heterodimerization and its subsequent binding to DNA. This results in decreased transcription and expression of HIF-2alpha downstream target genes, many of which regulate tumor cell growth and survival. Blocking HIF-2alpha reduces the proliferation of HIF-2alpha-expressing tumor cells. HIF-2alpha, a heterodimeric transcription factor overexpressed in many cancers, promotes tumorigenesis. PT-2385 is an orally bioavailable, specific HIF-2α antagonist. It inhibits HIF-2α transcriptional activity by allosterically blocking the heterodimerization between HIF-2α and its constitutive partner HIF-1β (ARNT), while having no effect on HIF-1α [2] The study identifies intestinal HIF-2α as a novel regulator of ceramide metabolism (via direct transcriptional activation of Neu3) and a contributor to obesity-related hepatic steatosis [2] PT-2385, which was in clinical trials for the treatment of renal cell carcinoma at the time of publication, is shown in this study to have preventive and therapeutic effects on metabolic disorders (obesity, insulin resistance, non-alcoholic fatty liver disease) in preclinical mouse models, primarily through inhibition of intestinal HIF-2α [2] The beneficial metabolic effects of PT-2385 are dependent on the presence of intestinal HIF-2α, as they are absent in mice with intestine-specific deletion of Hif2a [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.87 mg/mL (7.49 mM) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.52 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.52 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 4: ≥ 2.5 mg/mL (6.52 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6087 mL | 13.0433 mL | 26.0865 mL | |
| 5 mM | 0.5217 mL | 2.6087 mL | 5.2173 mL | |
| 10 mM | 0.2609 mL | 1.3043 mL | 2.6087 mL |