Physicochemical Properties
| Molecular Formula | C23H27NO4 |
| Molecular Weight | 381.46 |
| Exact Mass | 381.194 |
| CAS # | 2089415-51-8 |
| PubChem CID | 137643434 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 4.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 28 |
| Complexity | 495 |
| Defined Atom Stereocenter Count | 0 |
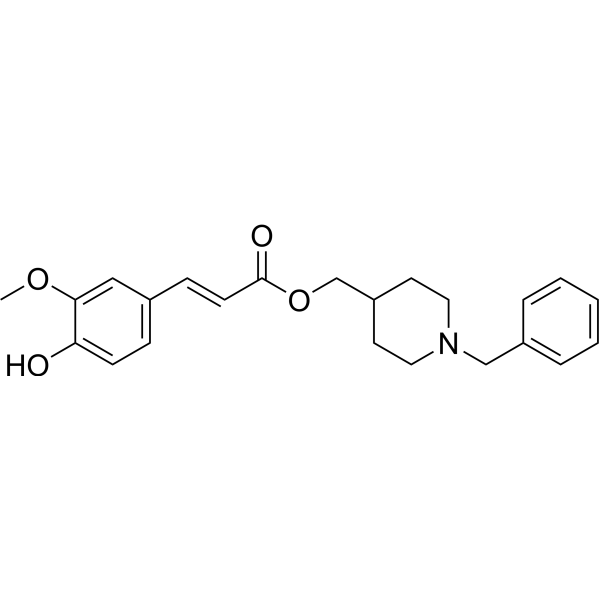
| SMILES | C(OCC1CCN(CC2=CC=CC=C2)CC1)(=O)/C=C/C1=CC=C(O)C(OC)=C1 |
| InChi Key | QJDRBHVMOOPIGE-CSKARUKUSA-N |
| InChi Code | InChI=1S/C23H27NO4/c1-27-22-15-18(7-9-21(22)25)8-10-23(26)28-17-20-11-13-24(14-12-20)16-19-5-3-2-4-6-19/h2-10,15,20,25H,11-14,16-17H2,1H3/b10-8+ |
| Chemical Name | (1-benzylpiperidin-4-yl)methyl (E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The target of PQM130 (a feruloyl-donepezil hybrid compound) is Acetylcholinesterase (AChE), with key activity data: - AChE (human recombinant): IC₅₀ = 0.058 μM [1] - Butyrylcholinesterase (BuChE, human recombinant): IC₅₀ = 3.2 μM (selectivity index > 55 vs. AChE) [1] |
| ln Vitro |
1. AChE/BuChE inhibitory activity: PQM130 potently inhibited AChE with an IC₅₀ of 0.058 μM, and showed weak inhibition of BuChE (IC₅₀ = 3.2 μM), exhibiting high selectivity for AChE (selectivity index > 55) [1] 2. Antioxidant activity: - DPPH radical scavenging assay: PQM130 scavenged DPPH radicals with an IC₅₀ of 12.8 μM [1] - ABTS radical cation scavenging assay: IC₅₀ = 8.5 μM [1] - Oxygen Radical Absorbance Capacity (ORAC) assay: Trolox equivalent antioxidant capacity (TEAC) = 2.8 mmol/g [1] 3. Neuroprotective effect against oxidative stress: - Human neuroblastoma SH-SY5Y cells were pretreated with PQM130 (1–20 μM) for 2 hours, then exposed to H₂O₂ (200 μM) for 24 hours. The compound dose-dependently increased cell viability (MTT assay): 20 μM PQM130 restored cell viability from 52% (H₂O₂ alone) to 89% [1] - Intracellular ROS levels (DCFH-DA staining) were reduced by 68% at 20 μM, and MDA (lipid peroxidation product) levels were decreased by 56% [1] - Western blot showed increased expression of antioxidant enzymes (SOD1, GSH-Px) and reduced expression of apoptotic proteins (Bax, cleaved caspase-3), with elevated Bcl-2 levels [1] 4. Inhibition of Aβ aggregation: Thioflavin T (ThT) fluorescence assay showed that PQM130 (5–40 μM) dose-dependently inhibited Aβ₁₋₄₂ aggregation. At 40 μM, aggregation was inhibited by 72% compared to the vehicle control [1] |
| ln Vivo |
Through modulation of GSK3β and extracellular signal-regulated kinase (ERK1/2) Synthesis, Icv AβO injection, followed by PQM130 (0.5-1 mg/kg, i.p., daily for 10 days) treatment, reduced oxidative damage and neuroinflammation and promoted cell survival and protein[1]. 1. Amelioration of cognitive impairments in APP/PS1 transgenic mice: - Animal model: 6-month-old male APP/PS1 double-transgenic mice (Alzheimer's disease model) were randomly divided into 4 groups (n=10/group): vehicle control (0.5% carboxymethylcellulose), PQM130 10 mg/kg, 20 mg/kg, 40 mg/kg, and wild-type (WT) control [1] - Morris water maze test: After 8 weeks of treatment, PQM130 dose-dependently reduced escape latency (40 mg/kg group: 28.5 ± 3.2 s vs. vehicle: 52.3 ± 4.1 s) and increased time spent in the target quadrant (40 mg/kg group: 38.2 ± 2.8% vs. vehicle: 22.5 ± 2.1%) [1] - Novel object recognition test: The discrimination index (DI) in the 40 mg/kg group was 0.58 ± 0.06, significantly higher than the vehicle group (0.23 ± 0.04), indicating improved recognition memory [1] 2. Attenuation of AD-related pathology: - Aβ deposition: Immunohistochemical staining of hippocampal and cortical tissues showed that 40 mg/kg PQM130 reduced Aβ₁₋₄₂ positive plaques by 65% (hippocampus) and 58% (cortex) [1] - Tau phosphorylation: Western blot of brain tissues showed dose-dependent reduction of p-Tau (Ser396/Ser404) levels; 40 mg/kg group reduced p-Tau/Total Tau ratio by 52% [1] - Neuroinflammation: RT-PCR and ELISA showed reduced mRNA and protein levels of pro-inflammatory cytokines (TNF-α, IL-6) in the hippocampus (40 mg/kg group: TNF-α reduced by 48%, IL-6 reduced by 53%) [1] 3. Improvement of oxidative stress in the brain: - Brain tissue analysis showed that PQM130 (40 mg/kg) increased SOD activity by 42% and GSH-Px activity by 38%, while decreasing MDA levels by 45% compared to the vehicle group [1] |
| Enzyme Assay |
1. AChE inhibitory activity assay (Ellman method): Recombinant human AChE was mixed with assay buffer, 5,5'-dithiobis-(2-nitrobenzoic acid) (DTNB), and acetylthiocholine iodide (substrate). Serial dilutions of PQM130 were added, and the mixture was incubated at 37°C for 30 minutes. The absorbance was measured at 412 nm, and inhibition rates were calculated relative to the vehicle control. Nonlinear regression was used to derive the IC₅₀ value (0.058 μM). The same protocol was used for BuChE inhibitory activity assay with butyrylthiocholine iodide as the substrate [1] 2. Aβ aggregation inhibition assay (ThT fluorescence): Aβ₁₋₄₂ peptide was dissolved in hexafluoroisopropanol, lyophilized, and reconstituted in PBS to form aggregated Aβ. Serial concentrations of PQM130 were mixed with aggregated Aβ, and ThT reagent was added. The mixture was incubated at 37°C for 24 hours, and fluorescence intensity (excitation 440 nm, emission 485 nm) was measured. Inhibition rate was calculated relative to Aβ alone group [1] |
| Cell Assay |
1. Neuroprotective assay against H₂O₂-induced damage: - SH-SY5Y cells were seeded in 96-well plates (5×10³ cells/well) and cultured overnight. Cells were pretreated with PQM130 (1–20 μM) for 2 hours, then exposed to 200 μM H₂O₂ for 24 hours [1] - Cell viability was measured by MTT assay: MTT reagent was added, incubated for 4 hours, formazan crystals were dissolved, and absorbance was measured at 570 nm [1] - ROS detection: Cells were loaded with DCFH-DA dye for 30 minutes before H₂O₂ exposure, and fluorescence intensity was measured by microplate reader (excitation 488 nm, emission 525 nm) [1] - Western blot: Cells were lysed, 30 μg protein was separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against SOD1, GSH-Px, Bax, Bcl-2, cleaved caspase-3, and β-actin (loading control) [1] 2. Antioxidant activity assays (DPPH/ABTS/ORAC): - DPPH assay: Serial dilutions of PQM130 were mixed with DPPH solution, incubated at room temperature for 30 minutes, and absorbance was measured at 517 nm [1] - ABTS assay: ABTS radical cation was generated by reacting ABTS with potassium persulfate. PQM130 was added, incubated for 15 minutes, and absorbance was measured at 734 nm [1] - ORAC assay: Fluorescein was used as a fluorescent probe, 2,2'-azobis(2-amidinopropane) dihydrochloride as a free radical generator. PQM130 was mixed with fluorescein and radical generator, incubated at 37°C, and fluorescence intensity was recorded every 5 minutes for 120 minutes. TEAC was calculated using Trolox as standard [1] |
| Animal Protocol |
Animal/Disease Models: Adult male C57Bl/6 mice (9 weeks old, weight 25-30 grams) [1]. Doses: 0.5 and 1.0 mg/kg. Route of Administration: Daily IP injections for 10 days. Experimental Results: Aβ1-42O-induced cognitive deficits in mice were improved. Preventing Aβ1-42O-induced neuronal death in mice. Antagonizes Aβ1-42O-induced oxidative stress in mice. Regulates the expression of GSK3β and ERK1/2 proteins in mice. Reduce Aβ1-42O-induced astrocyte activation in mice. 1. Cognitive and pathological evaluation in APP/PS1 mice: - 6-month-old male APP/PS1 double-transgenic mice and WT mice (C57BL/6 background) were used. Mice were acclimated for 1 week before treatment [1] - Drug formulation: PQM130 was dissolved in 0.5% carboxymethylcellulose to prepare suspensions of 10, 20, 40 mg/kg [1] - Administration: Oral gavage once daily for 8 weeks. Vehicle control group received 0.5% carboxymethylcellulose alone [1] - Behavioral tests: Morris water maze (5 days of training, 1 day of probe trial) and novel object recognition test were performed after 8 weeks of treatment [1] - Tissue collection: Mice were euthanized, brains were dissected, hippocampus and cortex were separated for immunohistochemistry, Western blot, RT-PCR, and oxidative stress parameter detection (SOD, GSH-Px, MDA) [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity: - ICR mice (n=10/group) were given a single oral dose of PQM130 (2000 mg/kg). Mice were monitored for 14 days for mortality, behavior, and body weight [1] - No mortality or abnormal behavior was observed. Body weight increased normally, and gross pathological examination of major organs (liver, kidney, brain, heart) showed no abnormalities [1] 2. Subchronic toxicity (8-week treatment in APP/PS1 mice): - Serum biochemical analysis showed no significant changes in ALT, AST, BUN, or creatinine levels in PQM130-treated groups (10–40 mg/kg) compared to vehicle control [1] - Histopathological examination of liver, kidney, and brain tissues showed no obvious lesions [1] |
| References |
[1]. PQM130, a Novel Feruloyl-Donepezil Hybrid Compound, Effectively Ameliorates the Cognitive Impairments and Pathology in a Mouse Model of Alzheimer's Disease. Front Pharmacol. 2019 Jun 12;10:658. |
| Additional Infomation |
1. Structural and background: PQM130 is a hybrid compound synthesized by conjugating ferulic acid (with antioxidant/anti-inflammatory activity) and donepezil (a clinically used AChE inhibitor). The hybrid design combines the dual effects of AChE inhibition and antioxidant/anti-inflammatory activity to target multiple AD pathologies [1] 2. Mechanism of action: - Inhibits AChE to increase acetylcholine levels in the brain, improving cholinergic neurotransmission [1] - Scavenges free radicals and enhances antioxidant enzyme activity, reducing oxidative stress and lipid peroxidation [1] - Inhibits Aβ₁₋₄₂ aggregation and reduces Aβ deposition [1] - Suppresses tau hyperphosphorylation and neuroinflammation (TNF-α, IL-6), attenuating neuronal damage [1] 3. Therapeutic potential: PQM130 effectively improves cognitive function and alleviates AD-related pathology (Aβ deposition, tau phosphorylation, oxidative stress, neuroinflammation) in APP/PS1 mice with low toxicity. It is a promising multi-target candidate for the treatment of Alzheimer's disease [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~1.82 mg/mL (~4.77 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6215 mL | 13.1075 mL | 26.2151 mL | |
| 5 mM | 0.5243 mL | 2.6215 mL | 5.2430 mL | |
| 10 mM | 0.2622 mL | 1.3108 mL | 2.6215 mL |