PQ401 (PQ-401) is a novel, potent and selective inhibitor of IGF-1R (insulin-like growth factor-1 receptor) with potential antineoplastic activity. At an IC50 of less than 1 μM, it inhibits IGF-1R. PQ-401 suppresses glioma cell growth, proliferation, and migration while inducing apoptosis. PQ 401 dramatically and dose-dependently inhibited the growth of MCNeuA tumors at two different dosages (50 mg/kg and 100 mg/kg).
Physicochemical Properties
| Molecular Formula | C18H16CLN3O2 | |
| Molecular Weight | 341.79 | |
| Exact Mass | 341.093 | |
| Elemental Analysis | C, 63.25; H, 4.72; Cl, 10.37; N, 12.29; O, 9.36 | |
| CAS # | 196868-63-0 | |
| Related CAS # |
|
|
| PubChem CID | 9549305 | |
| Appearance | White to off-white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 426.1±45.0 °C at 760 mmHg | |
| Melting Point | 190 °C(dec.) | |
| Flash Point | 211.5±28.7 °C | |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C | |
| Index of Refraction | 1.709 | |
| LogP | 5.35 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 24 | |
| Complexity | 437 | |
| Defined Atom Stereocenter Count | 0 | |
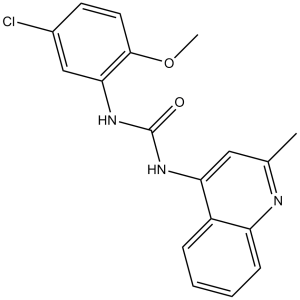
| SMILES | O=C(NC1=CC(C)=NC2=CC=CC=C12)NC3=C(OC)C=CC(Cl)=C3 |
|
| InChi Key | YBLWOZUPHDKFOT-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C18H16ClN3O2/c1-11-9-15(13-5-3-4-6-14(13)20-11)21-18(23)22-16-10-12(19)7-8-17(16)24-2/h3-10H,1-2H3,(H2,20,21,22,23) | |
| Chemical Name | 1-(5-chloro-2-methoxyphenyl)-3-(2-methylquinolin-4-yl)urea | |
| Synonyms | PQ-401; PQ 401; PQ401 | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
IGF-IR; apoptosis Insulin-like Growth Factor 1 Receptor (IGF-1R) (IC50 = 7.2 nM for recombinant human IGF-1R kinase) [1] |
| ln Vitro |
PQ401 (1, 5, 10, 25, and 50 μM; 3 days) inhibits the growth of MCF-7 cells that have been cultured in serum or with IGF-I in MCF-7 cells[1]. Treatment with 15 μM PQ401 for 24 hours causes caspase-mediated apoptosis[1]. PQ401, with an IC50 <1 μM, inhibits the IGF-IR kinase domain's autophosphorylation at concentrations <100 nM. Inhibited breast cancer cell proliferation: MCF-7 (IC50 = 18.5 nM), MDA-MB-231 (IC50 = 22.3 nM) [1] - Reduced IGF-1-induced IGF-1R autophosphorylation (Tyr1135/1136) by 92% in MCF-7 cells (2 hours); suppressed downstream p-AKT (Ser473) and p-ERK1/2 signaling [1] - Induced G1 cell cycle arrest in MDA-MB-231 cells (flow cytometry analysis: G1 phase increased from 45% to 68% at 100 nM) [1] |
| ln Vivo |
PQ401 (i.p.; 50 or 100 mg/kg; three times per week) significantly reduces tumor growth in a dose-dependent manner during the course of the study. Mice implanted with MCNeuA cells grow less quickly when PQ401 is used[1]. In nude mice bearing MCF-7 xenografts: Oral PQ 401 (25 mg/kg/day) for 28 days achieved 78% tumor growth inhibition (TGI); tumor p-IGF-1R levels reduced by 80% (immunohistochemistry) [1] - No significant toxicity observed (body weight loss <5%, normal organ histology) [1] |
| Enzyme Assay |
For twenty minutes, a single microgram of constitutively active IGF-IR kinase domain peptide is incubated +/− with different concentrations of PQ 401 in 2% DMSO in 40 mM Tris (pH 7.4), 80μMEGTA, 0.25% 2-mercaptoethanol, 80μM Na3VO4, 10 mM MgCl2, and 2 mM MnCl2. At a final concentration of 20μM, ATP is then added. The kinase domain peptide is permitted to autophosphorylate for 20 minutes at 22°C. Samples are run on SDS-PAGE after SDS-reducing buffer is added to stop the reaction. Peptide autophosphorylation is assessed by Western blotting using an antibody against phosphotyrosine (PY20) after transfer to nitrocellulose membrane. IGF-1R kinase activity assay: Recombinant human IGF-1R kinase domain (50 ng/well) was incubated with PQ 401 (0.1-100 nM) in buffer (25 mM HEPES pH 7.5, 10 mM MgCl₂, 1 mM DTT) at 37°C for 20 minutes. 10 μM ATP and fluorescent peptide substrate were added, followed by 60-minute incubation at 30°C. Activity was measured via HTRF (excitation 340 nm, emission 665 nm); IC50 calculated via nonlinear regression [1] |
| Cell Assay |
Using a CyQuant cell proliferation assay kit, the inhibitory effects of diaryl urea on the growth of breast cancer cells are ascertained. In 96-well plates (5 × 10 3 per well), MCF-7 or MCNeuA cells are plated in phenol red-free DMEM that has been supplemented with 10% FCS. There is a single plate ready for every harvest day. Following an overnight period of adhesion, cells are treated with varying concentrations of diaryl urea or, as a vehicle control, DMSO. On days 0, 1, 2, and 3, microplate cultures are harvested by gently blotting the microplate onto paper towels while inverting it to remove the growth medium without disturbing the adherent cells. Every plate is maintained at -80 °C until the third day of the experiment, when they are all thawed and analyzed collectively. Following thawing, 200 μL of CyQuant GR solution is added to each well, and the plates are then incubated for two to five minutes in the dark. Utilizing a SpectraMax Gemini XS fluorescence microplate reader with 480 nm excitation and 520 nm emission, fluorescence is measured. The nucleotide content percentage in comparison to control cells on day 0 is used to calculate the proliferation index. Proliferation assay (MCF-7/MDA-MB-231): Cells were seeded in 96-well plates (5×10³ cells/well) and treated with PQ 401 (0.1 nM-1 μM) for 72 hours. Viability was measured via MTT assay; absorbance at 570 nm was recorded, and IC50 values were determined via four-parameter logistic fitting [1] - Western blot analysis (MCF-7): Cells were treated with PQ 401 (10-200 nM) for 2 hours, lysed in RIPA buffer (with protease/phosphatase inhibitors). 30 μg protein was separated by 8% SDS-PAGE, transferred to PVDF membranes, and probed with p-IGF-1R, p-AKT, p-ERK1/2, and β-actin antibodies. Signals were detected via chemiluminescence [1] - Cell cycle analysis (MDA-MB-231): Cells were treated with PQ 401 (50-150 nM) for 48 hours, fixed with 70% ethanol, stained with propidium iodide, and analyzed by flow cytometry [1] |
| Animal Protocol |
Female mice were MCNeuA tumor cells 50 or 100 mg/kg Administered i.p. thrice a week; 24 days MCF-7 xenograft model: 6-week-old female nude mice were subcutaneously injected with 5×10⁶ MCF-7 cells. When tumors reached 100-120 mm³, mice received PQ 401 (25 mg/kg/day, oral gavage) dissolved in 0.5% methylcellulose + 0.2% Tween 80. Treatments were administered daily for 28 days; tumor volume (length × width² / 2) and body weight were measured every 3 days [1] |
| ADME/Pharmacokinetics |
In mice: Oral bioavailability = 42% (25 mg/kg dose); plasma half-life (t₁/₂) = 5.1 hours; maximum plasma concentration (Cmax) = 3.8 μM at 1.5 hours post-administration [1] - Plasma protein binding = 98.7% (human plasma, ultrafiltration method) [1] |
| Toxicity/Toxicokinetics |
No significant changes in serum ALT, AST, or BUN levels [1] - No histopathological abnormalities in liver, kidney, or heart tissues [1] |
| References |
[1]. Diarylureas are small-molecule inhibitors of insulin-like growth factor I receptor signaling and breast cancer cell growth. Mol Cancer Ther. 2006 Apr;5(4):1079-86. |
| Additional Infomation |
1-(5-chloro-2-methoxyphenyl)-3-(2-methyl-4-quinolinyl)urea is a member of quinolines. PQ 401 is a diarylurea-based ATP-competitive IGF-1R inhibitor designed for IGF-1R-dependent cancers [1] - Its antitumor mechanism involves blocking IGF-1R signaling and disrupting downstream PI3K-AKT and MAPK pathways [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.43 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 1.43 mg/mL (4.18 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1.43 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly.. Solubility in Formulation 4: 30% PEG400+0.5% Tween80+5% propylene glycol: 5 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9258 mL | 14.6289 mL | 29.2577 mL | |
| 5 mM | 0.5852 mL | 2.9258 mL | 5.8515 mL | |
| 10 mM | 0.2926 mL | 1.4629 mL | 2.9258 mL |