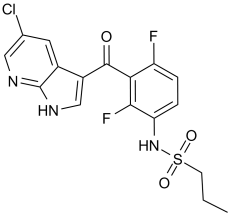
PLX4720 (PLX-4720; PLX 4720), a 7-azaindole/pyrrolopyridine-based vemurafenib derivative discovered by a structure-guided discovery approach, is a novel, potent and selective inhibitor of B-RafV600E mutant with potential antitumor activity. In a cell-free assay, it inhibits B-RafV600E with an IC50 of 13 nM and is 10 times more selective for B-RafV600E than for wild-type B-Raf. Oral administration of PLX-4720 causes tumor growth delays in B-RafV600E-dependent tumor xenograft models, induces cell cycle arrest and apoptosis in B-RafV600E-positive melanoma cells, and inhibits ERK phosphorylation in tumor cell lines containing B-RafV600E.
Physicochemical Properties
| Molecular Formula | C17H14CLF2N3O3S |
| Molecular Weight | 413.83 |
| Exact Mass | 413.041 |
| Elemental Analysis | C, 49.34; H, 3.41; Cl, 8.57; F, 9.18; N, 10.15; O, 11.60; S, 7.75 |
| CAS # | 918505-84-7 |
| Related CAS # | PLX-4720-d7;1304096-50-1 |
| PubChem CID | 24180719 |
| Appearance | white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 621.4±65.0 °C at 760 mmHg |
| Flash Point | 329.6±34.3 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.646 |
| LogP | 3.14 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 27 |
| Complexity | 648 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(C1C(F)=C(NS(CCC)(=O)=O)C=CC=1F)C1C2C(=NC=C(C=2)Cl)NC=1 |
| InChi Key | YZDJQTHVDDOVHR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H14ClF2N3O3S/c1-2-5-27(25,26)23-13-4-3-12(19)14(15(13)20)16(24)11-8-22-17-10(11)6-9(18)7-21-17/h3-4,6-8,23H,2,5H2,1H3,(H,21,22) |
| Chemical Name | N-[3-(5-chloro-1H-pyrrolo[2,3-b]pyridine-3-carbonyl)-2,4-difluorophenyl]propane-1-sulfonamide |
| Synonyms | PLX 4720; PLX4720; 918505-84-7; PLX-4720; PLX4720; N-(3-(5-chloro-1H-pyrrolo[2,3-b]pyridine-3-carbonyl)-2,4-difluorophenyl)propane-1-sulfonamide; PLX 4720; N-[3-[(5-Chloro-1H-pyrrolo[2,3-b]pyridin-3-yl)carbonyl]-2,4-difluorophenyl]-1-propanesulfonamide; N-[3-(5-chloro-1H-pyrrolo[2,3-b]pyridine-3-carbonyl)-2,4-difluorophenyl]propane-1-sulfonamide; MFCD14635203; PLX-4720 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
B-Raf (V600E) (IC50 = 13 nM); B-Raf (IC50 = 160 nM); BRK (IC50 = 130 nM); ; FRK (IC50 = 1300 nM); Csk (IC50 = 1500 nM); Src (IC50 = 1700 nM); FAK (IC50 = 1700 nM); FGFR (IC50 = 1900 nM); KDR (IC50 = 2300 nM); HGK (IC50 = 2800 nM); CSF1R (IC50 = 3300 nM); Aurora A (IC50 = 3400 nM) PLX-4720 is a highly selective inhibitor of the oncogenic mutant BRAF kinase (BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ). In recombinant human BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ kinase assays, it exhibits an IC₅₀ of 13 nM; it has minimal activity against wild-type BRAF (IC₅₀ > 10 μM) and other RAF family members (e.g., CRAF, IC₅₀ = 3.2 μM) [1] - PLX-4720 shows no significant inhibition of non-RAF kinases, including EGFR (IC₅₀ > 50 μM), MEK1 (IC₅₀ > 10 μM), and AKT (IC₅₀ > 20 μM), confirming its specificity for BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ [1] |
| ln Vitro |
PLX-4720 exhibits >10 times selectivity against wild-type B-Raf and >100 times selectivity over other kinases, including Frk, Src, Fak, FGFR, and Aurora A, with an IC50 of 1.3-3.4 μM. PLX-4720 significantly reduces the ERK phosphorylation in cell lines expressing B-RafV600E, but not in cells expressing wild-type B-Raf (IC50 = 14–46 nM). PLX-4720 significantly slows the expansion of tumor cell lines that carry the B-RafV600E oncogene, including COLO205, A375, WM2664, and COLO829, with GI50 values of 0.31 μM, 0.50 μM, 1.5 μM, and 1.7 μM, respectively. Additionally, treatment with PLX-4720 at 1 μM only causes cell cycle arrest and apoptosis in B-RafV600E-positive 1205Lu cells while having no effect on B-Raf wild-type C8161 cells[1]. In comparison to PTEN-cell lines (4-fold), PLX-4720 treatment (10 μM) significantly increases the expression of BIM in PTEN+ cells by > 14 times, which provides an explanation for PTEN-cells' resistance to PLX-4720's ability to induce apoptosis[2]. BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ Melanoma Cell Proliferation Inhibition: In human BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ-positive melanoma cell lines (A375, SK-MEL-28), PLX-4720 (0.001–10 μM) concentration-dependently inhibits cell proliferation: 0.021 μM reduces A375 cell viability by 50% (IC₅₀ = 21 nM), and 1 μM achieves 95% inhibition. This effect is accompanied by a 90% reduction in phosphorylated ERK1/2 (p-ERK, downstream MAPK marker) at 0.1 μM (Western blot analysis) [1] - PTEN-Deficient Melanoma Cell Resistance: In PTEN-knockout A375 cells (PTEN⁻/⁻), PLX-4720 (0.01–5 μM) shows reduced antiproliferative activity: the IC₅₀ increases from 21 nM (PTEN⁺/⁺) to 150 nM (PTEN⁻/⁻). This resistance is mediated by suppressed BIM (pro-apoptotic protein) expression—PTEN⁻/⁻ cells show 60% lower BIM levels vs. PTEN⁺/⁺ cells, and BIM overexpression restores sensitivity (IC₅₀ = 30 nM) [2] - BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ Thyroid Cancer Cell Inhibition: In human BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ-positive papillary thyroid cancer cells (BCPAP), PLX-4720 (0.05–5 μM) inhibits proliferation (IC₅₀ = 45 nM) and reduces thrombospondin-1 (TSP-1, pro-angiogenic protein) secretion by 70% at 0.5 μM (ELISA detection). It also blocks TSP-1-induced endothelial cell migration (by 80% at 0.5 μM) [3] - NRP1-Mediated Adaptive Resistance: In NRP1-overexpressing SK-MEL-28 cells, PLX-4720 (0.01–10 μM) shows decreased efficacy: 1 μM reduces viability by only 40% (vs. 95% in parental cells). NRP1 knockdown (via siRNA) restores sensitivity, with viability reduced by 85% at 1 μM [4] |
| ln Vivo |
In B-RafV600E-dependent COLO205 tumor xenografts, oral administration of PLX-4720 at 20 mg/kg/day results in significant tumor growth delays and regressions, with no overtly harmful side effects in mice, even at doses as high as 1 g/kg. While having no effect on the C8161 xenografts containing wild-type B-Raf, PLX-4720 at 100 mg/kg twice daily almost completely eradicates the 1205Lu xenografts bearing B-RafV600E. When PLX-4720 is applied to cells carrying the V600E mutation, the anti-tumor effects are correlated with the blockade of the MAPK pathway[1]. Treatment with PLX-4720 at 30 mg/kg/day significantly inhibits the growth of 8505c xenograft tumors by >90% and significantly lowers distant lung metastases[3]. Melanoma Xenograft Model: In nude mice bearing A375 BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ melanoma xenografts, oral administration of PLX-4720 (25, 50, 100 mg/kg/day, once daily) dose-dependently inhibits tumor growth: 100 mg/kg reduces tumor volume by 90% at day 28 vs. vehicle, and induces complete tumor regression in 40% of mice. Tumor p-ERK levels are reduced by 95% (immunohistochemistry), with no significant weight loss (<5%) [1] - PTEN-Deficient Melanoma Xenograft: In nude mice bearing PTEN⁻/⁻ A375 xenografts, oral PLX-4720 (100 mg/kg/day) shows reduced efficacy: tumor volume is reduced by only 35% at day 28 (vs. 90% in PTEN⁺/⁺ xenografts). Co-administration of a BIM agonist (10 mg/kg, i.p.) restores inhibition to 75% [2] - Thyroid Cancer Xenograft Model: In SCID mice bearing BCPAP thyroid cancer xenografts, oral PLX-4720 (75 mg/kg/day) reduces tumor volume by 65% at day 35 and decreases intratumoral microvessel density (by 50%, CD31 staining) via TSP-1 inhibition. No significant hepatic or renal toxicity is observed (serum ALT/AST and creatinine within normal range) [3] |
| Enzyme Assay |
In 20 mM Hepes (pH 7.0), 10 mM MgCl2, 1 mM DTT, 0.01% Tween-20, 100 nM biotin-MEK protein, varying ATP concentrations, and increasing concentrations of PLX-4720, 20-μL reactions are carried out for each enzyme (0.1 ng). At 2, 5, 8, 10, 20, and 30 minutes, reactions are stopped using 5 μL of a solution containing 20 mM hepes (pH 7.0), 200 mM sodium chloride, 80 mM EDTA, and 0.3% BSA. The AlphaScreen Protein A Detection Kit's phospho-MEK Antibody, Streptavidin-coated Donor beads, and Protein A Acceptor beads are also included in the stop solution. For 30 minutes, the antibody and beads are preincubated in stop solution at room temperature in the dark. The final antibody dilution is 1/2000, and the final bead concentration is 10 g/mL. The assay plates are read on a PerkinElmer AlphaQuest reader after an hour of room temperature incubation. Recombinant BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ Kinase Assay: Recombinant human BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ or wild-type BRAF protein (40 ng/well) was incubated in kinase buffer (25 mM Tris-HCl pH 7.5, 5 mM MgCl₂, 1 mM EGTA, 2 mM DTT, 10 μM ATP) with a biotinylated MEK1-derived peptide (substrate, 2 μM) and various concentrations of PLX-4720 (0.001–50 μM) at 30°C for 45 min. Phosphorylated substrate was detected using a homogeneous time-resolved fluorescence (HTRF) assay (Eu-labeled anti-phospho-MEK antibody + streptavidin-APC). Kinase activity was normalized to vehicle control, and IC₅₀ values were calculated via nonlinear regression [1] |
| Cell Assay |
PLX-4720 is applied to cells at different concentrations for 24, 48, and 72 hours. The MTT assay or CellTiter-Glo Luminescent Cell Viability Assay are used to measure cell proliferation. Supernatant and cells are collected, pelleted, and fixed with 70% ethanol for cell cycle analysis. Cells are incubated for 1 hour at 37°C in 0.5 mg/mL RNase I to remove any remaining RNA contamination before staining with propidium iodide (10 μg/mL). Following that, samples are examined using the EPICS XL device. Media and cells are collected, pelleted, and stained with annexin-FITC and propidium iodide to determine the level of apoptosis. The EPICS XL instrument is then used to analyze the samples. Melanoma Cell Proliferation Assay: A375/SK-MEL-28 cells were seeded in 96-well plates (4×10³ cells/well) in DMEM + 10% FBS. After 24 h adhesion, PLX-4720 (0.001–10 μM) was added, and cells were incubated for 72 h. Cell viability was measured via MTT assay (absorbance at 570 nm). For Western blot, cells were treated with drug for 24 h, lysed in RIPA buffer, and probed with anti-p-ERK, anti-total ERK, and anti-GAPDH antibodies [1] - PTEN-Deficient Cell BIM Expression Assay: PTEN⁺/⁺ and PTEN⁻/⁻ A375 cells were seeded in 6-well plates (2×10⁵ cells/well) and treated with PLX-4720 (0.1 μM) for 48 h. Cells were lysed, and BIM expression was analyzed via Western blot (anti-BIM antibody). For BIM overexpression, cells were transfected with a BIM-expressing plasmid (2 μg/well) 24 h before drug treatment, and viability was measured via CCK-8 assay [2] - Thyroid Cancer TSP-1 Secretion Assay: BCPAP cells were seeded in 24-well plates (1×10⁵ cells/well) and treated with PLX-4720 (0.05–5 μM) for 72 h. Supernatants were collected, and TSP-1 concentration was measured via sandwich ELISA (detection at 450 nm). Endothelial cell migration was assessed using a transwell assay: HUVECs were seeded in upper chambers with BCPAP supernatant (from drug-treated cells), and migrated cells were counted after 6 h [3] - NRP1-Mediated Resistance Assay: SK-MEL-28 cells were transfected with NRP1-overexpressing plasmid or siRNA (2 μg/well) for 24 h, then seeded in 96-well plates. PLX-4720 (0.01–10 μM) was added, and viability was measured via MTT assay after 72 h. NRP1 expression was confirmed via Western blot (anti-NRP1 antibody) [4] |
| Animal Protocol |
SCID mice have their flanks injected subcutaneously with 2×106 metastatic melanoma cells, which are given approximately two weeks to reach a volume of 0.125 mm3. The animals then either receive 100 mg/kg PLX4720 (oral gavage) or vehicle control twice daily for 15 days. The tumor volume is measured every 72 hours. Normalized to the tumor volume on the first day of treatment, the average tumor size for each respective group. Animals are put to death 15 days into the treatment process, and tumors are removed, formalin-fixed, paraffin-embedded, and immunohistochemically examined. A375 Melanoma Xenograft Protocol: Female nude mice (6–7 weeks old) were subcutaneously injected with A375 cells (6×10⁶ cells/mouse) into the right flank. When tumors reached 80–100 mm³, mice were randomized into 4 groups (n=7/group): Vehicle (0.5% methylcellulose + 0.1% Tween 80, p.o.), PLX-4720 25 mg/kg (p.o., q.d.), 50 mg/kg (p.o., q.d.), 100 mg/kg (p.o., q.d.). Drugs were administered daily for 28 days. Tumor volume (V = π×L×W²/6) and body weight were measured every 3 days. At study end, tumors were excised for p-ERK immunohistochemistry [1] - PTEN⁻/⁻ A375 Xenograft Protocol: Female nude mice were implanted with PTEN⁻/⁻ A375 cells (6×10⁶ cells/mouse) subcutaneously. When tumors reached 100 mm³, mice were divided into 3 groups (n=6/group): Vehicle, PLX-4720 100 mg/kg (p.o., q.d.), PLX-4720 100 mg/kg + BIM agonist 10 mg/kg (i.p., q.d.). Treatment lasted 28 days, with tumor volume measured every 3 days [2] - BCPAP Thyroid Cancer Xenograft Protocol: Male SCID mice (8 weeks old) were injected with BCPAP cells (8×10⁶ cells/mouse) subcutaneously. When tumors reached 120 mm³, mice were randomized into 2 groups (n=5/group): Vehicle, PLX-4720 75 mg/kg (p.o., q.d.). Treatment continued for 35 days. Tumor volume was measured every 4 days; at study end, tumors were collected for CD31 staining (microvessel density) and serum was analyzed for liver/kidney function markers [3] |
| Toxicity/Toxicokinetics |
Acute Toxicity: In nude mice treated with oral PLX-4720 (up to 200 mg/kg/day for 14 days), no mortality or severe toxicity (e.g., convulsions, organ damage) is observed. Mild transient diarrhea (<10% incidence) is reported at 150–200 mg/kg, with no significant weight loss (<5%) [1] - Chronic Toxicity: In SCID mice treated with PLX-4720 (75 mg/kg/day for 35 days), serum ALT/AST levels (liver markers) and creatinine (renal marker) remain within normal ranges. No histopathological changes are observed in liver, kidney, or heart tissues [3] - Plasma Protein Binding: In mouse plasma (measured via ultrafiltration), PLX-4720 has a protein binding rate of ~98% at concentrations of 0.01–10 μM, with no concentration dependence [1] |
| References |
[1]. Discovery of a selective inhibitor of oncogenic B-Raf kinase with potent antimelanoma activity. Proc Natl Acad Sci U S A, 2008, 105(8), 3041-3046. [2]. PTEN loss confers BRAF inhibitor resistance to melanoma cells through the suppression of BIM expression. Cancer Res, 2011, 71(7), 2750-2760. [3]. B-Raf(V600E) and thrombospondin-1 promote thyroid cancer progression. Proc Natl Acad Sci U S A, 2010, 107(23), 10649-10654. [4]. Neuropilin-1 upregulation elicits adaptive resistance to oncogene-targeted therapies. J Clin Invest. 2018 Aug 31;128(9):3976-3990. |
| Additional Infomation |
PLX-4720 is a pyrrolopyridine that is vemurafenib in which the p-chlorophenyl group has been replaced by chlorine. It is a potent and selective inhibitor of the Raf kinase B-Raf(V600E). It has a role as a B-Raf inhibitor and an antineoplastic agent. It is a pyrrolopyridine, a sulfonamide, a difluorobenzene, an organochlorine compound and an aromatic ketone. BRAF(V600E) is the most frequent oncogenic protein kinase mutation known. Furthermore, inhibitors targeting "active" protein kinases have demonstrated significant utility in the therapeutic repertoire against cancer. Therefore, we pursued the development of specific kinase inhibitors targeting B-Raf, and the V600E allele in particular. By using a structure-guided discovery approach, a potent and selective inhibitor of active B-Raf has been discovered. PLX4720, a 7-azaindole derivative that inhibits B-Raf(V600E) with an IC(50) of 13 nM, defines a class of kinase inhibitor with marked selectivity in both biochemical and cellular assays. PLX4720 preferentially inhibits the active B-Raf(V600E) kinase compared with a broad spectrum of other kinases, and potent cytotoxic effects are also exclusive to cells bearing the V600E allele. Consistent with the high degree of selectivity, ERK phosphorylation is potently inhibited by PLX4720 in B-Raf(V600E)-bearing tumor cell lines but not in cells lacking oncogenic B-Raf. In melanoma models, PLX4720 induces cell cycle arrest and apoptosis exclusively in B-Raf(V600E)-positive cells. In B-Raf(V600E)-dependent tumor xenograft models, orally dosed PLX4720 causes significant tumor growth delays, including tumor regressions, without evidence of toxicity. The work described here represents the entire discovery process, from initial identification through structural and biological studies in animal models to a promising therapeutic for testing in cancer patients bearing B-Raf(V600E)-driven tumors.[1] This study addresses the role of PTEN loss in intrinsic resistance to the BRAF inhibitor PLX4720. Immunohistochemical staining of a tissue array covering all stages of melanocytic neoplasia (n = 192) revealed PTEN expression to be lost in >10% of all melanoma cases. Although PTEN expression status did not predict for sensitivity to the growth inhibitory effects of PLX4720, it was predictive for apoptosis, with only limited cell death observed in melanomas lacking PTEN expression (PTEN-). Mechanistically, PLX4720 was found to stimulate AKT signaling in the PTEN- but not the PTEN+ cell lines. Liquid chromatography multiple reaction monitoring mass spectrometry (LC-MRM) was performed to identify differences in apoptosis signaling between the two cell line groups. PLX4720 treatment significantly increased BIM expression in the PTEN+ (>14-fold) compared with the PTEN- cell lines (four-fold). A role for PTEN in the regulation of PLX4720-mediated BIM expression was confirmed by siRNA knockdown of PTEN and through reintroduction of PTEN into cells that were PTEN-. Further studies showed that siRNA knockdown of BIM significantly blunted the apoptotic response in PTEN+ melanoma cells. Dual treatment of PTEN- cells with PLX4720 and a PI3K inhibitor enhanced BIM expression at both the mRNA and protein level and increased the level of apoptosis through a mechanism involving AKT3 and the activation of FOXO3a. In conclusion, we have shown for the first time that loss of PTEN contributes to intrinsic BRAF inhibitor resistance via the suppression of BIM-mediated apoptosis.[2] PLX-4720 is a research-grade selective inhibitor of BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ, developed as a先导 compound for clinical BRAF inhibitors (e.g., vemurafenib). It is not approved for clinical use [1] - Mechanism of Action: Its antitumor effect is mediated by specific inhibition of BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ kinase activity, blocking the constitutively activated MAPK (RAS-RAF-MEK-ERK) signaling pathway, which drives proliferation and survival of BRAF-mutant cancers [1,3] - Resistance Mechanisms: Preclinical resistance to PLX-4720 occurs via PTEN loss (suppressed BIM expression, [2]) and NRP1 overexpression (enhanced survival signaling, [4]). Combination strategies (BIM agonists, NRP1 inhibitors) restore sensitivity [2,4] - Research Applications: PLX-4720 is widely used in preclinical studies to investigate BRAFⁿᵉᵗ/ᵛ⁶⁰⁰ᴱ biology, including its role in melanoma, thyroid cancer, and resistance to RAF inhibition [1,3,4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: 2% DMSO +50% PEG 300 +5% Tween 80 +ddH2O: 5mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4165 mL | 12.0823 mL | 24.1645 mL | |
| 5 mM | 0.4833 mL | 2.4165 mL | 4.8329 mL | |
| 10 mM | 0.2416 mL | 1.2082 mL | 2.4165 mL |