PI-3065 is a novel, potent and selective inhibitor of PI3K (phosphatidylinositol 3-kinase) p110δ inhibitor with potential anticancer activity. With an IC50 of 15 nM, it blocks p110δ and exhibits >70-fold selectivity over other PI3K family proteins, including p110α, p110β, p110γ which have IC50s of 910, 600, and >10000 nM, respectively.
Physicochemical Properties
| Molecular Formula | C27H31FN6OS |
| Molecular Weight | 506.6380 |
| Exact Mass | 506.226 |
| Elemental Analysis | C, 64.01; H, 6.17; F, 3.75; N, 16.59; O, 3.16; S, 6.33 |
| CAS # | 955977-50-1 |
| Related CAS # | 955977-50-1 |
| PubChem CID | 24937012 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.698 |
| LogP | 2.64 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 36 |
| Complexity | 748 |
| Defined Atom Stereocenter Count | 0 |
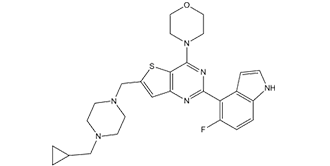
| SMILES | FC1C(C2N=C(N3CCOCC3)C3=C(C=C(CN4CCN(CC5CC5)CC4)S3)N=2)=C2C(NC=C2)=CC=1 |
| InChi Key | YDNOHCOYQVZOMC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H31FN6OS/c28-21-3-4-22-20(5-6-29-22)24(21)26-30-23-15-19(17-33-9-7-32(8-10-33)16-18-1-2-18)36-25(23)27(31-26)34-11-13-35-14-12-34/h3-6,15,18,29H,1-2,7-14,16-17H2 |
| Chemical Name | 4-[6-[[4-(cyclopropylmethyl)piperazin-1-yl]methyl]-2-(5-fluoro-1H-indol-4-yl)thieno[3,2-d]pyrimidin-4-yl]morpholine |
| Synonyms | PI 3065; PI-3065; PI3065 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
p110α (IC50 = 910 nM); p110β (IC50 = 600 nM); p110δ (IC50 = 5 nM) 1. Phosphatidylinositol 3-Kinase δ (PI3Kδ, p110δ/p85α complex) - IC50 ~1.2 nM (recombinant human PI3Kδ, HTRF-based kinase activity assay)[1] - Ki ~0.8 nM (recombinant human PI3Kδ, ATP-competitive binding assay)[1] 2. Ultra-high selectivity over other PI3K subtypes and immune-related kinases: - PI3Kα (p110α/p85α): IC50 > 10,000 nM (same HTRF assay as PI3Kδ)[1] - PI3Kβ (p110β/p85α): IC50 > 8,000 nM (same assay)[1] - PI3Kγ (p110γ/p101): IC50 > 5,000 nM (same assay)[1] - No significant inhibition of 40+ immune kinases (e.g., JAK1/2, STAT3, NF-κB) at 1 μM concentration[1] [1] |
| ln Vitro |
PI-3065 shows no inhibition on the growth of 4T1 cells, which do not expressing detectable levels of p110δ.[1] 1. Regulatory T cell (Treg) function inhibition (Literature [1]): - Mouse splenic Treg cells (CD4+CD25+FOXP3+): - Isolated Tregs incubated with PI-3065 (10-500 nM) for 24 hours. 100 nM PI-3065 reduced FOXP3 protein expression by ~65% (Western blot) and FOXP3 mRNA by ~70% (qRT-PCR); 50 nM reduced Treg-mediated suppression of effector T cell (Teff) proliferation by ~55% (CFSE dilution assay, 72 hours). - 200 nM PI-3065 decreased Treg secretion of immunosuppressive cytokines IL-10 by ~75% and TGF-β by ~70% (ELISA) vs. vehicle. - Human peripheral blood Tregs: - 200 nM PI-3065 reduced FOXP3 expression by ~60% (flow cytometry) and impaired Treg suppression of Teff proliferation by ~50% (3H-thymidine incorporation assay)[1] 2. PI3Kδ-AKT signaling suppression in Tregs (Literature [1]): - Serum-starved mouse Tregs treated with PI-3065 (10-500 nM) for 1 hour, then stimulated with IL-2 (10 ng/mL) for 15 minutes. 50 nM PI-3065 reduced phosphorylated AKT (Ser473) by ~85% and phosphorylated AKT (Thr308) by ~80% (Western blot); 100 nM completely blocked IL-2-induced AKT activation. - No effect on Teff cell (CD4+CD25-) viability or proliferation at concentrations up to 500 nM (trypan blue exclusion, >90% viability)[1] 3. Dendritic cell (DC) maturation promotion (Literature [1]): - Mouse bone marrow-derived DCs (BMDCs) stimulated with LPS (100 ng/mL) + PI-3065 (100 nM) for 24 hours. PI-3065 increased DC maturation markers CD80 (+45%) and CD86 (+50%) (flow cytometry) and enhanced TNF-α secretion by ~60% (ELISA) vs. LPS alone[1] [1] |
| ln Vivo |
PI-3065 shows no inhibition on the growth of 4T1 cells, which do not expressing detectable levels of p110δ. [1] PI-3065 (75 mg/kg, p.o.) also inhibits the growth and metastasis of 4T1 tumors in mouse models by inactivating p110δ. PI-3065 increases survival and lowers the frequency of macroscopic metastases and other disease-related pathologies in the LSL.KrasG12D/+; p53R172H/+; PdxCretg/+ (or KPC) model of pancreatic ductal adenocarcinoma. [1] 1. Antitumor immune response enhancement in mouse tumor models (Literature [1]): - B16-F10 melanoma xenograft (C57BL/6 mice, 8 mice/group): - Tumor induction: 5×10⁵ B16-F10 cells resuspended in PBS, subcutaneous injection into right flank. - Administration: PI-3065 dissolved in 0.5% methylcellulose + 0.1% Tween 80, oral gavage at 30 mg/kg/day, started when tumors ~100 mm³ and continued for 21 days. Vehicle group received 0.5% methylcellulose + 0.1% Tween 80. - Efficacy: - Tumor growth: 30 mg/kg PI-3065 reduced tumor volume by ~70% vs. vehicle (p < 0.01) and extended median survival from 25 days (vehicle) to 42 days (p < 0.01). - Immune infiltration: Tumor-infiltrating CD8+ cytotoxic T cells increased by ~2.5-fold, and Tregs (CD4+CD25+FOXP3+) decreased by ~60% (flow cytometry of tumor single-cell suspensions) vs. vehicle. - MC38 colorectal cancer xenograft (C57BL/6 mice, 7 mice/group): - Administration: 30 mg/kg PI-3065 oral gavage for 14 days. - Efficacy: Tumor volume reduced by ~65% vs. vehicle (p < 0.01); serum IFN-γ (pro-inflammatory cytokine) increased by ~2-fold (ELISA) vs. vehicle[1] [1] |
| Enzyme Assay |
PI-3065 is a novel, potent and selective inhibitor of PI3K (phosphatidylinositol 3-kinase) p110δ inhibitor with potential anticancer activity. It inhibitsp110δ with an IC50 of 15 nM, and showed >70-fold selectivity over other PI3K family proteins such as p110α, p110β, p110γ with IC50s of 910, 600, >10000 nM, respectively.Cell Assay: Proliferation of 4T1 cells is assayed following a 4-h treatment with the indicated p110δ inhibitors, then washing and MTS staining are carried out after 48 h culture.PI-3065 shows no inhibition on the growth of 4T1 cells, which do not expressing detectable levels of p110δ 1. PI3Kδ kinase activity assay (HTRF-based): - Reagent preparation: Recombinant human PI3Kδ (p110δ/p85α) resuspended in assay buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.01% Tween 20). Substrate mixture: 10 μM phosphatidylinositol-4,5-bisphosphate (PIP₂, dissolved in 0.1% CHAPS) + 2 μM ATP + Eu³+-labeled streptavidin-ATP. - Reaction system: 50 μL mixture contained 5 nM PI3Kδ, substrate mixture, and serial concentrations of PI-3065 (0.001-100 nM). Vehicle control (0.1% DMSO) included to normalize kinase activity. Incubated at 30℃ for 60 minutes. - Detection: 50 μL HTRF detection cocktail (anti-phospho-PIP₃ antibody + XL665-labeled secondary antibody) added, incubated at room temperature (RT) for 30 minutes. Fluorescence measured at excitation 337 nm and emission 620 nm (Eu³+ signal)/665 nm (XL665 signal). Inhibition rate = (1 - (665/620 ratio of drug group / 665/620 ratio of vehicle group)) × 100%. IC50 derived via nonlinear regression (GraphPad Prism). 2. PI3Kδ ATP-competitive binding assay: - Reagent preparation: Recombinant PI3Kδ (p110δ/p85α) immobilized on streptavidin-coated 96-well plates. Fluorescent ATP analog (FITC-ATP) dissolved in binding buffer (25 mM HEPES pH 7.4, 5 mM MgCl₂, 0.1% BSA) to 100 nM. - Reaction system: 100 μL mixture contained immobilized PI3Kδ, 100 nM FITC-ATP, and serial concentrations of PI-3065 (0.001-10 nM). Incubated at RT for 90 minutes. - Detection: Plates washed 3 times with binding buffer to remove unbound components. Fluorescence intensity measured at excitation 485 nm and emission 535 nm. Ki calculated using competitive binding equation (Km for ATP-PI3Kδ = 15 μM, determined separately)[1] [1] |
| Cell Assay |
1. Mouse Treg isolation and suppression assay:
- Treg isolation: Mouse spleens minced, single-cell suspensions prepared; CD4+CD25+ Tregs purified via magnetic bead sorting (negative selection for CD4+, positive selection for CD25+), resuspended in RPMI 1640 + 10% FBS + 50 μM β-mercaptoethanol.
- Suppression assay: Tregs (1×10⁴ cells/well) co-cultured with CFSE-labeled Teffs (5×10⁴ cells/well) in 96-well plates; PI-3065 (10-500 nM) or vehicle added. Plates incubated at 37℃, 5% CO₂ for 72 hours. Teff proliferation analyzed via flow cytometry (CFSE dilution); suppression rate = (1 - (proliferating Teffs in co-culture / proliferating Teffs alone)) × 100%.
2. Western blot for Treg signaling molecules:
- Tregs (2×10⁵ cells/well) seeded in 6-well plates, serum-starved for 4 hours; treated with PI-3065 (10-500 nM) for 1 hour, then stimulated with IL-2 (10 ng/mL) for 15 minutes.
- Cells lysed with RIPA buffer (containing protease/phosphatase inhibitors); 30 μg protein loaded per lane, separated by 10% SDS-PAGE. Membrane probed with anti-FOXP3, anti-p-AKT (Ser473/Thr308), anti-total AKT, and anti-GAPDH (loading control) antibodies. Band intensity quantified via ImageJ.
3. DC maturation assay:
- BMDCs generated from mouse bone marrow cells (cultured with GM-CSF 20 ng/mL + IL-4 10 ng/mL for 7 days); seeded in 24-well plates (1×10⁵ cells/well).
- Treated with LPS (100 ng/mL) + PI-3065 (100 nM) or LPS alone for 24 hours. DCs harvested, stained with anti-CD80 and anti-CD86 antibodies (flow cytometry); supernatant collected for TNF-α ELISA[1] [1] |
| Animal Protocol |
The mammary fat pad of female WT BALB/c mice receives a 1×105 4T1 cell orthotopically inoculation on day 0. From day 1 (administered 12 hours before tumour cell inoculation), the drug (75 mg/kg PI-3065, once daily) or vehicle (0.5% methylcellulose with 0.2% Tween 80) are given orally by gavage. Weekly measurements with calipers or luminescence measurements performed on a Xenogen imaging platform are used to track tumor growth. Upon the death of the mice on day 35, tumors and ancillary organs are removed for in vitro luminescence analysis. This is done after fixation in 4% PFA and H&E staining. Prior to receiving either vehicle or PI-3065 treatment for a total of 14 days, KPC mice are allowed to develop advanced lesions measuring 5 to 10 mm (as determined by ultrasound imaging). 1. B16-F10 melanoma xenograft protocol: - Animals: Male C57BL/6 mice (6-8 weeks old), 8 mice per group; acclimated to laboratory conditions for 7 days (12-hour light/dark cycle, ad libitum food/water). - Tumor induction: 5×10⁵ B16-F10 melanoma cells resuspended in 100 μL sterile PBS, subcutaneous injection into the right flank of each mouse. - Drug preparation: PI-3065 was dissolved in 0.5% methylcellulose + 0.1% Tween 80 (stirred at RT for 2 hours to ensure complete dissolution, no precipitation observed). The 30 mg/kg dose was prepared by adjusting the drug concentration. - Administration: Mice were randomly divided into 2 groups (n=8/group): - Vehicle group: Oral gavage of 0.5% methylcellulose + 0.1% Tween 80 (10 μL/g body weight) once daily for 21 days, starting when tumors reached an average volume of ~100 mm³ (measured with calipers, volume = length × width² / 2). - PI-3065 group: Oral gavage of 30 mg/kg PI-3065 (10 μL/g body weight) once daily for 21 days, starting at the same tumor volume. - Assessment: - Tumor volume and body weight measured twice weekly. Mice were monitored for survival until tumor volume exceeded 1500 mm³. - On day 21, 4 mice per group were euthanized; tumors excised, minced, and digested with collagenase to prepare single-cell suspensions. Flow cytometry analyzed tumor-infiltrating CD8+ T cells and Tregs. 2. MC38 colorectal cancer xenograft protocol: - Animals: Male C57BL/6 mice (6-8 weeks old), 7 mice per group; acclimated for 7 days. - Tumor induction: 5×10⁵ MC38 cells resuspended in 100 μL PBS, subcutaneous injection into right flank. - Drug preparation & administration: Same as B16-F10 model (30 mg/kg PI-3065 oral gavage for 14 days). - Assessment: Tumor volume measured twice weekly; day 14: euthanized, serum collected for IFN-γ ELISA; tumors fixed in 4% paraformaldehyde for H&E staining[1] [1] |
| Toxicity/Toxicokinetics |
1. In vitro toxicity:
- Mouse/human Tregs, Teffs, and BMDCs: PI-3065 at concentrations up to 1 μM showed no non-specific cytotoxicity. LDH release assay revealed <10% leakage (vs. vehicle) after 24-hour exposure; trypan blue exclusion assay showed >90% cell viability.
- Human peripheral blood mononuclear cells (PBMCs): 500 nM PI-3065 showed <15% proliferation inhibition, confirming low off-target toxicity[1] 2. In vivo toxicity: - Mice (oral 30 mg/kg/day PI-3065 for 21 days): No mortality or abnormal behaviors (e.g., ataxia, lethargy, reduced food/water intake); body weight remained unchanged (±5% of initial weight) compared to the vehicle group. - Serum chemistry (day 21): ALT/AST (liver function) and creatinine (kidney function) were within normal ranges (ALT: 53 ± 7 U/L vs. normal 40-60 U/L; AST: 116 ± 12 U/L vs. normal 100-130 U/L; creatinine: 54 ± 5 μmol/L vs. normal 50-70 μmol/L, n=5 per group). - Histopathology: No drug-induced damage observed in liver, kidney, spleen, or lymph nodes of treated mice[1] |
| References |
[1]. Inactivation of PI(3)K p110δ breaks regulatory T-cell-mediated immune tolerance to cancer. Nature. 2014 Jun 19;509(7505):407-11. |
| Additional Infomation |
1. Mechanism of action:
PI-3065 is a selective PI3Kδ inhibitor that binds to the ATP-binding pocket of the p110δ catalytic subunit of PI3Kδ. This binding blocks PI3Kδ-mediated phosphorylation of PIP₂ to PIP₃, thereby inhibiting downstream AKT activation in Tregs. Reduced AKT signaling downregulates the expression of FOXP3 (a master transcription factor for Treg function) and impairs Treg secretion of immunosuppressive cytokines (IL-10, TGF-β). The weakened Treg-mediated immune tolerance allows effector T cells and dendritic cells to mount a robust antitumor immune response, ultimately suppressing tumor growth[1] 2. Preclinical significance: - Validates PI3Kδ as a novel therapeutic target for cancer immunotherapy. By demonstrating that PI-3065 specifically disrupts Treg function without damaging effector immune cells, the study provides a rational basis for developing PI3Kδ inhibitors to enhance antitumor immunity—addressing the limitation of traditional immunotherapies (e.g., checkpoint inhibitors) that fail to overcome Treg-mediated immunosuppression in the tumor microenvironment. - PI-3065 exhibits favorable oral efficacy and safety in immune-competent mice (C57BL/6), supporting its potential as a preclinical tool for studying PI3Kδ-Treg axis in cancer immunology[1] 3. Limitations: - No clinical development data (e.g., FDA approval status) reported; PI-3065 remains a research tool compound, not a therapeutic candidate. - Efficacy only evaluated in mouse melanoma and colorectal cancer models; no data in other cancer types (e.g., breast, lung cancer) or human clinical samples. No data on combination with other immunotherapies (e.g., anti-PD-1 antibodies)[1] [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~50 mg/mL warming (~98.7 mM) Water: <1 mg/mL Ethanol: <1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.93 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.93 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9738 mL | 9.8689 mL | 19.7379 mL | |
| 5 mM | 0.3948 mL | 1.9738 mL | 3.9476 mL | |
| 10 mM | 0.1974 mL | 0.9869 mL | 1.9738 mL |