Physicochemical Properties
| Molecular Formula | C22H23F3N4O3 |
| Molecular Weight | 448.44 |
| Exact Mass | 448.17 |
| Elemental Analysis | C, 58.92; H, 5.17; F, 12.71; N, 12.49; O, 10.70 |
| CAS # | 2159123-14-3 |
| PubChem CID | 132200405 |
| Appearance | White to off-white solid powder |
| LogP | 1.7 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 32 |
| Complexity | 664 |
| Defined Atom Stereocenter Count | 4 |
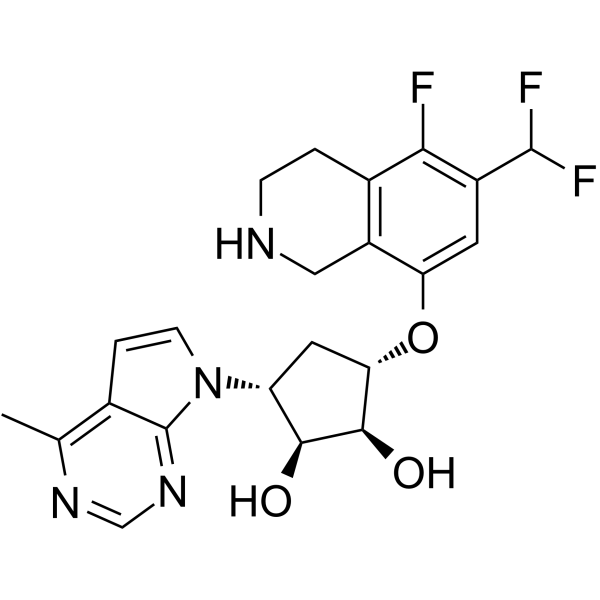
| SMILES | CC1=C2C=CN(C2=NC=N1)[C@@H]3C[C@@H]([C@H]([C@H]3O)O)OC4=C5CNCCC5=C(C(=C4)C(F)F)F |
| InChi Key | HBGHQRGHFNTSDP-DGJRPJRTSA-N |
| InChi Code | InChI=1S/C22H23F3N4O3/c1-10-11-3-5-29(22(11)28-9-27-10)15-7-17(20(31)19(15)30)32-16-6-13(21(24)25)18(23)12-2-4-26-8-14(12)16/h3,5-6,9,15,17,19-21,26,30-31H,2,4,7-8H2,1H3/t15-,17?,19+,20-/m1/s1 |
| Chemical Name | (1S,2S,5R)-3-((6-(difluoromethyl)-5-fluoro-1,2,3,4-tetrahydroisoquinolin-8-yl)oxy)-5-(4-methyl-7H-pyrrolo[2,3-d]pyrimidin-7-yl)cyclopentane-1,2-diol |
| Synonyms | PF-06939999; PRMT5-IN-3;PF06939999; PF 06939999; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PRMT5 |
| ln Vitro |
PF-06939999 reduces cellular biomarkers and proliferation of NSCLC cell lines.[1] PF-06939999 inhibition in NSCLC impacts alternative splicing pathways.[1] Splicing dysregulation is associated with NSCLC sensitivity to PF-06939999 inhibition.[1] |
| ln Vivo |
PF-06939999 demonstrates tumor growth inhibition in splicing mutant NSCLC.[1] To further validate the antitumor effects of PF-06939999 treatment in vivo, tumor growth inhibition studies (TGI) were conducted in two mouse xenograft models of NSCLC. PF-06939999 showed significant tumor suppression as an orally administered single agent in two splicing factor mutant NSCLC xenograft models. PF-06939999 demonstrated dose dependent TGI in A427 (RBM10I348N) tumors at day 44, of 52.1% at 3 mg/kg, 74.9% at 10 mg/kg, and 100.6% at 30 mg/kg once daily doses (Fig. 5A). Modulation of SDMA in A427 tumors was evaluated via ELISA at the end of the study (Fig. 5B). PF-06939999 was well tolerated with minimal body weight loss (Fig. 5C). In the NCI-H441 (U2AF1S34F) model, PF-06939999 demonstrated dose-dependent TGI at day 36 of 55.5% at 5 mg/kg twice a day, 14.9% at 3 mg/kg once daily, 46.6% at 10 mg/kg once daily, and 87.2% at 30 mg/kg once daily doses (Fig. 5D). SDMA levels assessed in the NCI-H441 tumors were evaluated via ELISA at the end of the study (Fig. 5E). PF-06939999 was well tolerated with minimal body weight loss (Fig. 5F). Pharmacokinetics of PF-06939999.[1] PF-06939999 showed moderate plasma clearance and steady-state volume of distribution (∼40 mL/min/kg and 3.8 L/kg, respectively) in male Wistar-Han rats following a single intravenous administration at the dose of 2 mg/kg (Supplementary Fig. S1B). Oral bioavailability was moderate (∼40%) in rats following a single oral administration at the dose of 10 mg/kg. Elimination half-lives for the intravenous and oral administration were 1.5 and 3.2 hours, respectively, suggesting flip-flop kinetics, although it might be due to the limited time points after 7 hours postdose. These results demonstrate that PF-06939999 is orally available in animals. |
| Enzyme Assay |
Co-crystal structures[1] Crystallization of full-length human PRMT5/MEP50 complexed with cofactor site inhibitors was performed at 13°C by hanging-drop vapor-diffusion methods. A solution of 5:1 molar ratio (2.5 μL) of inhibitor compound to PRMT5/MEP50 complex (13 mg/mL) was mixed with 2.5 μL of reservoir solution containing 13% to 15% (w/v) PEG3350, 0.1M MES, pH 6.5 to 7.5, 0.25 mol/L NaCl, and 20% (v/v) ethylene glycol. Microseeding from initial crystals produced crystals suitable for data collection. Crystals for data collection were flash-frozen in liquid N2 using 25% (v/v) ethylene glycol in the mother liquor as a cryoprotectant and shipped for to the Advanced Photon Source IMCA-CAT beamline 17-ID at Argonne National Labs for diffraction data collection. Diffraction data were processed with autoPROC from Global Phasing and structure solution and refinement were done with BUSTER using the published structure of human PRMT5/MEP50 (PDB ID 4GQB) as the initial model. Model building was done with COOT. Crystallographic statistics are included in Supplementary Table S1. |
| Cell Assay |
Western blotting[1] Cells were lysed in RIPA buffer + protease/phosphatase inhibitor, sonicated, and cleared by centrifugation. Histones were purified using the Histone Purification Kit (Active Motif). Protein lysates were loaded at 20 μg and histones at 500 ng. Antibody information is in the supplemental data. Blots were imaged on a LI-COR Odyssey CLx imager and bands quantified using the LI-COR Image Studio software. Proliferation assays[1] NSCLC cells were seeded in 96-well plates in recommended culture media and incubated overnight at 37°C, 5% CO2. The following day, fresh media with compound (diluted in DMSO) was added and cells incubated at 37°C, 5% CO2 for 7 days with media/compound refreshed at day 3 to 4. Cells were lysed in Cell Titer Glo reagent and read on a plate reader with luminescence filter. Alternatively, CyQuant reagent was added to the plates at day 7, the plate incubated at 37°C for 1 hour and read on plate reader with fluorescent filter. Cell-cycle analysis[1] Cells were seeded at appropriate densities to allow for growth of 80% to 90% confluency by day 5. Cells were treated in dose response with PF-06939999 with DMSO as a control for either 4 or 5 days, stained with propidium iodide and analyzed by flow cytometry to observe changes in the phases of the cell cycle. RNA sequencing[1] A427, NCI-H441, and NCI-H1975 cells were seeded in triplicate in recommended growth media overnight at 37°C, 5% CO2. The following day, plates were treated with either 0.1% DMSO or 30 nmol/L PF-06939999 for 72 hours at 37°C, 5% CO2. After RNA isolation, the library was prepared using the TruSeq Stranded mRNA Library kit (Illumina) and sequenced at 100 MM read depth with the Illumina HiSeq X 10 platform. Sequencing reads (150 bp paired end) were mapped to hg19 genome using STAR and quantified using RSEM. The DESeq2 program was used for differential expression analysis and alternative splicing analysis was done using rMATS version 4.02. A minimum number of reads mapping to a splice junction was imposed to filter low-coverage splice junctions from the analysis: the minimal number of junction reads for at least one sample group for rMAT testing is 10. Gene pathway analysis utilized a hypergeometric test with FDR correction against the MSigDB database. |
| Animal Protocol |
In vivo human lung cancer xenograft models[1] A427 xenograft experiments were conducted at Pfizer and performed using 6- to 8-week-old female NSG mice purchased from The Jackson Laboratory (strain name NOD.Cg-PrkdcscidIL2rgtm1Wjl/SzJ). In brief, 5 × 106 A427 cells (0.2 mL in 50% Matrigel; and 50% serum-free RPMI1640) were inoculated subcutaneously into the right flank of each mouse. NCI-H441 xenograft experiments were conducted at Crown Bio using 7- to 8-week-old male Nu/Nu mice purchased from Charles River Laboratories (strain name Crl:NU-Foxn1nu). NCI-H441 cells (5 × 106; 0.1 mL in 50% Matrigel, 50% PBS) were injected subcutaneously into the flank of each mouse. Once tumors were palpable, tumor length and width were measured by calipers 2 to 3 times weekly. Animals were randomly assigned to experimental groups and treatment was initiated at day 0, when tumor volume reached 150 mm3 on average. Ten animals were enrolled in each treatment arm. Tumor volume was calculated by the standard formula L × W2 × 0.5. For all lung xenograft experiments, PF-06939999 was administered daily by oral gavage at doses of 3, 10, and 30 mg/kg [vehicle: 0.5% methylcellulose (w/v) solution with 0.1% polysorbate 80 (w/v) in water]. In addition, the NCI-H441 in vivo experiment included an additional treatment arm of PF-06939999 dosed orally at 5 mg/kg twice daily. |
| References | [1]. SAM-Competitive PRMT5 Inhibitor PF-06939999 Demonstrates Antitumor Activity in Splicing Dysregulated NSCLC with Decreased Liability of Drug Resistance. Mol Cancer Ther . 2022 Jan;21(1):3-15. doi: 10.1158/1535-7163.MCT-21-0620. Epub 2021 Nov 4. |
| Additional Infomation | Protein Arginine Methyltransferase 5 Inhibitor PF-06939999 is an orally available inhibitor of protein arginine N-methyltransferase 5 (histone-arginine N-methyltransferase PRMT5; PRMT5) with potential antiproliferative and antineoplastic activities. Although the mechanism of action has not yet been fully elucidated, orally administered PRMT5 inhibitor PF-06939999 inhibits the methyltransferase activity of PRMT5, thereby decreasing the levels of monomethylated and dimethylated arginine residues in histones H2A, H3, and H4, and modulating the expression of genes involved in several cellular processes including cell proliferation. This may increase the expression of antiproliferative genes and/or decrease the expression of genes that promote cell proliferation, and may decrease the growth of rapidly proliferating cells, including cancer cells. PRTM5, an arginine methyltransferase that can catalyze the formation of both omega-N monomethylarginine (MMA) and symmetrical dimethylarginine (sDMA) on histones and a variety of other protein substrates, is overexpressed in several neoplasms. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2300 mL | 11.1498 mL | 22.2995 mL | |
| 5 mM | 0.4460 mL | 2.2300 mL | 4.4599 mL | |
| 10 mM | 0.2230 mL | 1.1150 mL | 2.2300 mL |