PF-06250112 is a novel, potent, highly selective, orally bioavailable BTK inhibitor with an IC50 of 0.5 nM, shows inhibitory effect toward BMX nonreceptor tyrosine kinase and TEC with IC50s of 0.9 nM and 1.2 nM, respectively. PF-06250112 inhibits both BCR-mediated signaling and proliferation, as well as FcR-mediated activation. To assess the therapeutic impact of BTK inhibition, aged NZBxW_F1 mice were treated with PF-06250112 and demonstrate that PF-06250112 significantly limits the spontaneous accumulation of splenic germinal center B cells and plasma cells. Correspondingly, anti-dsDNA and autoantibody levels were reduced in a dose-dependent manner. Moreover, administration of PF-06250112 prevented the development of proteinuria and improved glomerular pathology scores in all treatment groups. Strikingly, this therapeutic effect could occur with only a modest reduction observed in anti-dsDNA titers, implying a critical role for BTK signaling in disease pathogenesis beyond inhibition of autoantibody production. PF-06250112 prevents proteinuria in an FcR-dependent, Ab-mediated model of glomerulonephritis. Importantly, these results highlight that BTK inhibition potently limits the development of glomerulonephritis by impacting both cell- and effector molecule-mediated pathways. These data provide support for evaluating the efficacy of BTK inhibition in systemic lupus erythematosus patients.
Physicochemical Properties
| Molecular Weight | 438.430010795593 |
| Exact Mass | 438.161 |
| CAS # | 1609465-89-5 |
| Related CAS # | (Rac)-PF-06250112;1609465-88-4 |
| PubChem CID | 90118673 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 32 |
| Complexity | 712 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | C1C[C@@H](CN(C1)C#N)N2C(=C(C(=N2)C3=CC=C(C=C3)OC4=C(C=C(C=C4)F)F)C(=O)N)N |
| InChi Key | SQFDBQCBXUWICP-HNNXBMFYSA-N |
| InChi Code | InChI=1S/C22H20F2N6O2/c23-14-5-8-18(17(24)10-14)32-16-6-3-13(4-7-16)20-19(22(27)31)21(26)30(28-20)15-2-1-9-29(11-15)12-25/h3-8,10,15H,1-2,9,11,26H2,(H2,27,31)/t15-/m0/s1 |
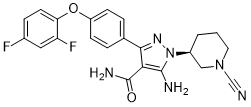
| Chemical Name | 1‐(1‐Cyanopiperidine‐3α‐yl)‐3‐[4‐(2,4‐difluorophenoxy)phenyl]‐5‐amino‐1H‐pyrazole‐4‐carboxamide |
| Synonyms | PF-06250112; PF 06250112; PF06250112. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | PF-06250112 prevented the development of proteinuria in both a spontaneous model of SLE and an effector phase–restricted model mediated by administration of anti-GBM Abs. [1] |
| ln Vivo |
In NZBxW_F1 mice, treatment with PF-06250112 significantly reduced spontaneous germinal center and plasma cell formation. [1] Treatment with PF-06250112 prevented the development of glomerulonephritis in the Ab-driven, anti-GBM–induced nephritis model.[1] |
| Cell Assay | Isolated B cells were resuspended at 50 × 106/ml in PBS, and 5 × 106 cells were treated with different concentrations of PF-06250112 or DMSO as a vehicle control for 1 h at 37°C. Cells were then stimulated with 15 μg/ml anti-human IgM for 5 min at 37°C, lysed in cell lysis buffer containing protease and phosphatase inhibitors . [1] |
| Animal Protocol | NZBxW_F1 mice were treated daily with PF-06250112 or vehicle by oral gavage. For anti-glomerlular basement membrane-induced nephritis studies, mice were treated by oral gavage with PF-06250112 or vehicle 2 h before injection of mouse anti-rabbit IgG mAb and treatment was continued daily thereafter.[1] |
| References |
[1]. Selective inhibition of BTK prevents murine lupus and antibody-mediated glomerulonephritis. J Immunol. 2013 Nov 1;191(9):4540-50. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2809 mL | 11.4043 mL | 22.8087 mL | |
| 5 mM | 0.4562 mL | 2.2809 mL | 4.5617 mL | |
| 10 mM | 0.2281 mL | 1.1404 mL | 2.2809 mL |