Physicochemical Properties
| Molecular Formula | C20H19F3IN5O2 |
| Molecular Weight | 545.296846628189 |
| Exact Mass | 545.054 |
| Elemental Analysis | C, 44.05; H, 3.51; F, 10.45; I, 23.27; N, 12.84; O, 5.87 |
| CAS # | 548756-68-9 |
| PubChem CID | 5287529 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 4.33 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 31 |
| Complexity | 565 |
| Defined Atom Stereocenter Count | 0 |
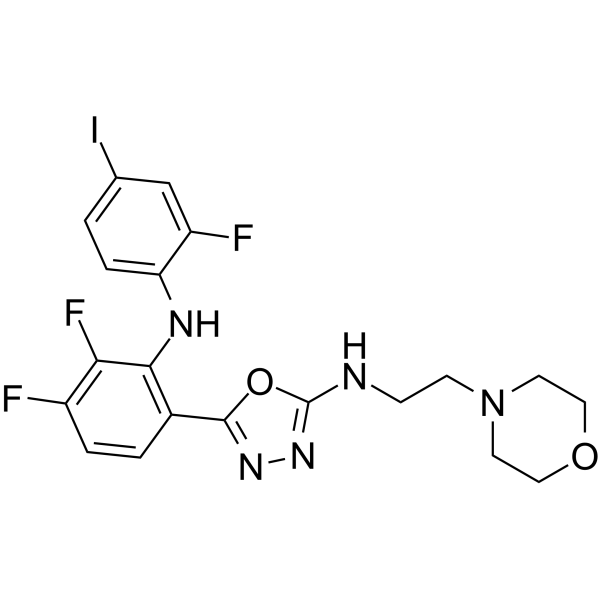
| SMILES | IC1=CC(F)=C(NC2=C(F)C(F)=CC=C2C2OC(NCCN3CCOCC3)=NN=2)C=C1 |
| InChi Key | LZZYEMSEMRUPIM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H19F3IN5O2/c21-14-3-2-13(18(17(14)23)26-16-4-1-12(24)11-15(16)22)19-27-28-20(31-19)25-5-6-29-7-9-30-10-8-29/h1-4,11,26H,5-10H2,(H,25,28) |
| Chemical Name | 5-[3,4-difluoro-2-(2-fluoro-4-iodoanilino)phenyl]-N-(2-morpholin-4-ylethyl)-1,3,4-oxadiazol-2-amine |
| Synonyms | 548756-68-9; PD 334581; PD-334581; 5-[3,4-difluoro-2-(2-fluoro-4-iodoanilino)phenyl]-N-(2-morpholin-4-ylethyl)-1,3,4-oxadiazol-2-amine; 5-{3,4-DIFLUORO-2-[(2-FLUORO-4-IODOPHENYL)AMINO]PHENYL}-N-(2-MORPHOLIN-4-YLETHYL)-1,3,4-OXADIAZOL-2-AMINE; LZZYEMSEMRUPIM-UHFFFAOYSA-N; {5-[3,4-Difluoro-2-(2-fluoro-4-iodo-phenylamino)-phenyl]-[1,3,4]oxadiazol-2-yl)-(2-morpholin-4-yl-ethyl)-amine; {5-[3,4-Difluoro-2-(2-fluoro-4-iodo-phenylamino)-phenyl]-[1,3,4]oxadiazol-2-yl}-(2-morpholin-4-yl-ethyl)-amine; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | MEK1 |
| ln Vitro |
Structure of MEK2 is highly homologous to MEK1[1] To determine whether the nonscompetitive binding pocket and concurrent binding of MgATP and inhibitor observed in MEK1 exist in the highly homologous MEK2, the crystal structure of MEK2 was solved as a ternary complex with MgATP and an analog of PD184352 to a resolution of 3.2 Å. After structural determination by molecular replacement using the MEK1 structure as a search model, the overall structure of MEK2 was found to be highly homologous to MEK1 with a r.m.s. deviation of ∼0.8 Å for the Cα trace. However, the MEK2 structure is more disordered when compared with MEK1, with regions in the N and C termini, the activation loop and the proline-rich loop that were not observed in the electron density. Simultaneous binding of MgATP and PD334581 was observed for MEK2. In addition, the kinase activation loop and helix C also adopt a similar conformation to that observed in MEK1, suggesting that the same mechanism of inhibition is involved in both MEK1 and MEK2, even though the two structures were determined with different PD184352-like compounds. |
| References |
[1]. Structures of human MAP kinase kinase 1 (MEK1) and MEK2 describe novel noncompetitive kinase inhibition. Nat Struct Mol Biol. 2004 Dec;11(12):1192-7. |
| Additional Infomation | MEK1 and MEK2 are closely related, dual-specificity tyrosine/threonine protein kinases found in the Ras/Raf/MEK/ERK mitogen-activated protein kinase (MAPK) signaling pathway. Approximately 30% of all human cancers have a constitutively activated MAPK pathway, and constitutive activation of MEK1 results in cellular transformation. Here we present the X-ray structures of human MEK1 and MEK2, each determined as a ternary complex with MgATP and an inhibitor to a resolution of 2.4 A and 3.2 A, respectively. The structures reveal that MEK1 and MEK2 each have a unique inhibitor-binding pocket adjacent to the MgATP-binding site. The presence of the potent inhibitor induces several conformational changes in the unphosphorylated MEK1 and MEK2 enzymes that lock them into a closed but catalytically inactive species. Thus, the structures reported here reveal a novel, noncompetitive mechanism for protein kinase inhibition.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8339 mL | 9.1693 mL | 18.3385 mL | |
| 5 mM | 0.3668 mL | 1.8339 mL | 3.6677 mL | |
| 10 mM | 0.1834 mL | 0.9169 mL | 1.8339 mL |