Description: PD 144418 is a novel, high affinity, highly potent and specific sigma 1 (σ1) receptor ligand with Ki values of 0.08 nM and 1377 nM for σ1 and σ2 respectively. PD 144418 oxalate devoids of any significant affinity for other receptors, ion channels and enzymes. PD 144418 oxalate shows highly potential antipsychotic activity.
Physicochemical Properties
| Molecular Formula | C18H22N2O.C2H2O4 |
| Molecular Weight | 372.41496 |
| Exact Mass | 372.169 |
| CAS # | 154130-99-1 |
| Related CAS # | PD 144418 oxalate;1794760-28-3 |
| PubChem CID | 9817231 |
| Appearance | White to light yellow solid-liquid Mixture |
| LogP | 3.242 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 21 |
| Complexity | 360 |
| Defined Atom Stereocenter Count | 0 |
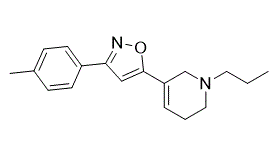
| SMILES | CC1C=CC(C2C=C(C3CN(CCC)CCC=3)ON=2)=CC=1 |
| InChi Key | FOQRKFCLRMMKAT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H22N2O/c1-3-10-20-11-4-5-16(13-20)18-12-17(19-21-18)15-8-6-14(2)7-9-15/h5-9,12H,3-4,10-11,13H2,1-2H3 |
| Chemical Name | 3-(4-methylphenyl)-5-(1-propyl-3,6-dihydro-2H-pyridin-5-yl)-1,2-oxazole |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | While haloperidol potentiates the reduction in 5-equilibrium tryptophan induced by mesolimbic brain regions, it has no effect on 5-HT and dopamine (DA) synthesis per se. Externally, PD 144418 supplementation increased N-methyl-D-aspartate (NMDA)-induced cyclic GMP (cGMP) in cerebellar slices without affecting basal levels, suggesting that the σ1 site may regulate glutamine-induced PD 144418. |
| ln Vivo | Single anti-mescaline-induced scratching is treated with PD 144418 (10 mg/kg; i.p.; constant CD-1 mice) at a dose that does not change spontaneous locomotor activity; PD 144418 has an ED50 value of 7.0 mg/kg IP [ |
| Animal Protocol |
Animal/Disease Models: Male CD-1 mice induced with mescaline[1] Doses: 10 mg/kg Route of Administration: intraperitoneal (ip) injection Experimental Results: Antagonizes mescaline-induced induction at a dose that does not alter spontaneous locomotor activity Scratching. |
| References |
[1]. The pharmacology of the novel and selective sigma ligand, PD 144418. Neuropharmacology. 1997 Jan;36(1):51-62. [2]. Relationship between cerebral sigma-1 receptor occupancy and attenuation of cocaine's motor stimulatory effects in mice by PD144418. J Pharmacol Exp Ther. 2014 Oct;351(1):153-63. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~354.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.85 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (8.85 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6852 mL | 13.4261 mL | 26.8521 mL | |
| 5 mM | 0.5370 mL | 2.6852 mL | 5.3704 mL | |
| 10 mM | 0.2685 mL | 1.3426 mL | 2.6852 mL |