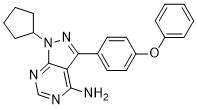
PCI 29732 is a novel, potent, selective and irreversible Btk (Bruton's tyrosine kinase) inhibitor with IC50 of 8.2 nM in a FRET based biochemical enzymology assay. PCI-29732 functions by preventing CD20+ B cells' expression of genes mediated by the B cell antigen receptor (BCR).
Physicochemical Properties
| Molecular Formula | C22H21N5O |
| Molecular Weight | 371.4350 |
| Exact Mass | 371.175 |
| Elemental Analysis | C, 71.14; H, 5.70; N, 18.85; O, 4.31 |
| CAS # | 330786-25-9 |
| Related CAS # | 2934.99.9001 |
| PubChem CID | 22347110 |
| Appearance | White to off-white solid powder |
| LogP | 5.564 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 28 |
| Complexity | 499 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C1C([H])=C([H])C([H])=C([H])C=1[H])C1C([H])=C([H])C(=C([H])C=1[H])C1C2=C(N([H])[H])N=C([H])N=C2N(C2([H])C([H])([H])C([H])([H])C([H])([H])C2([H])[H])N=1 |
| InChi Key | GMJUPMONHWAZCP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H21N5O/c23-21-19-20(26-27(16-6-4-5-7-16)22(19)25-14-24-21)15-10-12-18(13-11-15)28-17-8-2-1-3-9-17/h1-3,8-14,16H,4-7H2,(H2,23,24,25) |
| Chemical Name | 1-cyclopentyl-3-(4-phenoxyphenyl)pyrazolo[3,4-d]pyrimidin-4-amine |
| Synonyms | PCI-29732; PCI29732; PCI 29732 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
BTK (IC50 = 0.5 nM); BLK (IC50 = 0.5 nM); Bmx (IC50 = 0.8 nM); EGFR (IC50 = 5.6 nM); YES (IC50 = 6.5 nM) Bruton tyrosine kinase (Btk) (reversible inhibitor, IC50 = 0.3 nM)[3] |
| ln Vitro |
PCI-32765 blocks BCR signaling in peripheral human B cells at doses that have no effect on signaling from T cell receptors. The phosphorylation of Btk (IC50, 11 nM), Btk's physiological substrate PLCγ (IC50, 29 nM), and ERK (IC50, 13 nM), a further downstream kinase, is all inhibited in DOHH2 cells by PCI-32765. The antigen receptor signaling in B cells is inhibited by PCI-32765 more than 1,000 times more selectively than in T cells, and only B-cell inhibition is maintained after a brief course of therapy[1]. PCI-29732, a reversible Btk inhibitor, was used as a control to compare with the irreversible inhibitor PCI-32765 in human peripheral B-cell activation assays. In purified human CD20+ B cells stimulated via the B-cell receptor (BCR), continuous exposure to PCI-29732 (1 μM) for the duration of the assay (6 hours) blocked the transcriptional up-regulation of a panel of B-cell activation genes (including CD69, CD83, EGR1, EGR2, EGR3, FOS) induced by anti-IgM stimulation. A 1-hour pulse exposure to PCI-29732 (1 μM) followed by washout before BCR stimulation did NOT result in inhibition of gene up-regulation, confirming its reversible mechanism of action. This was in contrast to the irreversible inhibitor PCI-32765, where pulse exposure was sufficient for sustained inhibition.[3] The IC50 for enzymatic inhibition of Btk by PCI-29732 is reported as 0.3 nM.[3] |
| ln Vivo |
Oral PCI-32765 administration lowers the level of autoantibodies in circulation and totally suppresses the disease in mice with collagen-induced arthritis. In addition, PCI-32765 prevents the MRL-Fas(lpr) lupus model from producing autoantibodies or developing kidney disease. For mice given PCI-32765 orally, the mean terminal plasma half-life is between 1.7 and 3.1 hours [1]. In a nude mouse xenograft model established with ABCG2-overexpressing H460/MX20 cells, oral administration of PCI-29732 (20 mg/kg, every 3 days for 5 times) in combination with intraperitoneal topotecan (3 mg/kg, same schedule) resulted in a significant reduction in tumor volume and weight compared to control or single-agent groups. The combination therapy achieved a tumor growth inhibition rate (IR) of 52.53% without causing significant body weight loss or drug-related deaths [2] |
| Enzyme Assay |
The effect of PCI-29732 on the ATPase activity of ABCG2 was measured using a colorimetric ATPase assay. Crude membranes from insect cells expressing ABCG2 were incubated with various concentrations of PCI-29732 in ATPase assay buffer (containing KCl, sodium azide, EDTA, MgCl2, DTT) with or without sodium orthovanadate. The reaction was initiated by adding Mg-ATP, incubated, and terminated with SDS. The amount of released inorganic phosphate was quantified colorimetrically after adding a detection reagent containing ammonium molybdate, zinc acetate, and ascorbic acid. The vanadate-sensitive ATPase activity was calculated. PCI-29732 stimulated ABCG2 ATPase activity at low concentrations and inhibited it at higher concentrations [2]. A photo-affinity labeling assay was performed to investigate the interaction between PCI-29732 and ABCG2. Crude membranes expressing ABCG2 were incubated with PCI-29732 and then with [¹²⁵I]-iodoarylazidoprazosin (IAAP). The mixture was cross-linked by UV illumination. Labeled ABCG2 was immunoprecipitated, separated by SDS-PAGE, and the radioactivity incorporated into the ABCG2 band was quantified using a phosphorimager. PCI-29732 competitively inhibited the photo-labeling of ABCG2 by [¹²⁵I]-IAAP in a concentration-dependent manner [2] |
| Cell Assay |
Purified human peripheral B cells (CD20+) were obtained by negative selection from buffy coat PBMCs. Cells were treated with vehicle or PCI-29732 (1 μM) for 1 hour. For the "washout" condition, the inhibitor-containing media was replaced with fresh media before stimulation. Cells were then stimulated with goat anti-human IgM F(ab')2 (10 μg/mL) for 6 hours to activate the BCR pathway. mRNA was prepared and gene expression levels of B-cell activation markers (CD69, CD83, EGR1, EGR2, EGR3, FOS) were measured by quantitative real-time RT-PCR (TaqMan). Expression levels were normalized to unstimulated cells.[3] |
| Animal Protocol |
Arthritic DBA/1 mice 3.125, 12.5, or 50 mg/kg per day oral An athymic nude mouse xenograft model was established by subcutaneously implanting H460/MX20 cells into the right flank. When tumors reached a measurable size (around day 7 post-inoculation), mice were randomly divided into four groups: (1) saline control (i.p., every 3 days, 5 times); (2) topotecan alone (3 mg/kg, i.p., every 3 days, 5 times); (3) PCI-29732 alone (20 mg/kg, p.o., every 3 days, 5 times); (4) combination of PCI-29732 (20 mg/kg, p.o.) and topotecan (3 mg/kg, i.p., administered 1 hour after PCI-29732, every 3 days, 5 times). Tumor dimensions and body weights were measured periodically. Tumors were excised and weighed at the endpoint (day 25) [2] |
| References |
[1]. Discovery of selective irreversible inhibitors for Bruton's tyrosine kinase. ChemMedChem. 2007 Jan;2(1):58-61. [2]. PCI29732, a Bruton's Tyrosine Kinase Inhibitor, Enhanced the Efficacy of Conventional Chemotherapeutic Agents in ABCG2-Overexpressing Cancer Cells. Cell Physiol Biochem. 2018;48(6):2302-2317. [3]. The Bruton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc Natl Acad Sci U S A. 2010;107(29):13075-13080. |
| Additional Infomation |
1-Cyclopentyl-3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine is an aromatic ether. PCI-29732 is a potent, reversible inhibitor of Bruton tyrosine kinase (Btk). It was used in this study primarily as a pharmacological control to demonstrate the distinct mechanism (reversible vs. irreversible binding) and functional consequences (requirement for continuous exposure) compared to the irreversible Btk inhibitor PCI-32765. Its structure is similar to PCI-32765 but lacks the covalent binding moiety.[3] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~74 mg/mL (~199.2 mM) Ethanol: ~4 mg/mL (~10.8 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1 mg/mL (2.69 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear EtOH stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1 mg/mL (2.69 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1 mg/mL (2.69 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear EtOH stock solution to 900 μL of corn oil and mix well. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6922 mL | 13.4611 mL | 26.9222 mL | |
| 5 mM | 0.5384 mL | 2.6922 mL | 5.3844 mL | |
| 10 mM | 0.2692 mL | 1.3461 mL | 2.6922 mL |