Physicochemical Properties
| Molecular Formula | C36H49F3N8O9 |
| Molecular Weight | 794.817679166794 |
| Exact Mass | 794.357 |
| CAS # | 1228078-65-6 |
| PubChem CID | 71311736 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 9 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 56 |
| Complexity | 1210 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | FC(C(=O)O)(F)F.O=C([C@H](CC1C=CC(=CC=1)O)NC([C@H](C)N)=O)N1CCC[C@H]1C(NCC(N[C@H](C(N[C@H](C(N)=O)CC1C=CC=CC=1)=O)CCCCN)=O)=O |
| InChi Key | BGPJLFVICWHITH-HKJXYENISA-N |
| InChi Code | InChI=1S/C34H48N8O7.C2HF3O2/c1-21(36)31(46)41-27(19-23-12-14-24(43)15-13-23)34(49)42-17-7-11-28(42)33(48)38-20-29(44)39-25(10-5-6-16-35)32(47)40-26(30(37)45)18-22-8-3-2-4-9-22;3-2(4,5)1(6)7/h2-4,8-9,12-15,21,25-28,43H,5-7,10-11,16-20,35-36H2,1H3,(H2,37,45)(H,38,48)(H,39,44)(H,40,47)(H,41,46);(H,6,7)/t21-,25-,26-,27-,28-;/m0./s1 |
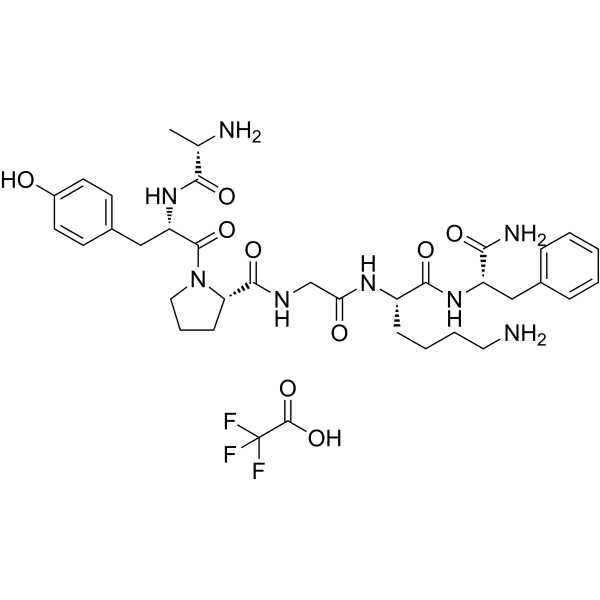
| Chemical Name | (2S)-N-[2-[[(2S)-6-amino-1-[[(2S)-1-amino-1-oxo-3-phenylpropan-2-yl]amino]-1-oxohexan-2-yl]amino]-2-oxoethyl]-1-[(2S)-2-[[(2S)-2-aminopropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]pyrrolidine-2-carboxamide;2,2,2-trifluoroacetic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Proteinase-activated receptor-4 (PAR-4) agonist [1] |
| ln Vivo |
At distension levels of 0.04 to 0.1 mL, SCID mice exhibited considerably stronger abdominal responses to colorectal distension (CRD) compared to BALB/cBy controls. The EMG response intensities rose by 384% to 132%, respectively (P<0.01; P<0.01; P<0.001). This hypersensitive reaction is effectively reversible (P<0.01, P<0.05, P<0.05) upon activation of PAR-4 [1]. In mice, intracolonic (IC) infusion of PAR-4 Agonist Peptide (AYPGFK-NH₂, 100 µg in 0.15 mL saline) prior to colorectal distension (CRD) produced a significant antinociceptive (analgesic) effect, reducing abdominal electromyographic (EMG) response compared to vehicle at distension volumes from 0.04 to 0.08 mL. This antinociceptive effect was absent when the same dose was administered intraperitoneally (IP), indicating a local site of action. The analgesic effect of IC PAR-4 Agonist Peptide was abolished by co-administration of the tight junction blocker 2,4,6-triaminopyrimidine (TAP), suggesting the peptide needs to penetrate the colonic epithelium to reach target receptors. In severe combined immunodeficiency (SCID) mice, which exhibited baseline hypersensitivity to CRD, IC PAR-4 Agonist Peptide effectively reversed this hypersensitivity, indicating its action is independent of functional B or T lymphocytes. In a water avoidance stress (WAS) model of visceral hypersensitivity, IC PAR-4 Agonist Peptide suppressed the stress-induced increase in sensitivity to CRD, restoring responses to baseline levels. In a low-grade 2,4,6-trinitrobenzenesulfonic acid (TNBS) colitis model of inflammatory hyperalgesia, IC PAR-4 Agonist Peptide also reversed the enhanced sensitivity to CRD. Furthermore, IC PAR-4 Agonist Peptide significantly reduced (by 46%) the CRD-induced overexpression of Fos protein (a marker of neuronal activation) in the lumbosacral (L5-S1) spinal cord. [1] |
| Animal Protocol |
Adult male C57BL/6J, BALB/cBy, or SCID mice (8-9 weeks old) were used. Under subcutaneous anesthesia (xylazine + ketamine), nickel-chrome electrodes were implanted into the abdominal external oblique muscle for EMG recording. After 4-7 days recovery, visceral sensitivity was assessed by measuring the abdominal EMG response to graded colorectal distension (CRD). The distension was performed by inflating a rectal balloon with saline in 0.02 mL increments from 0 to 0.10 mL. For drug testing, PAR-4 Agonist Peptide (AYPGFK-NH₂) was dissolved in 0.9% NaCl (saline). In most experiments, a dose of 100 µg in 0.15 mL saline was administered via intracolonic (IC) infusion. Visceral sensitivity measurements began 1 hour after the end of the IC infusion. In one experiment, the same dose (100 µg) was administered via intraperitoneal (IP) bolus injection, and measurements began 15 minutes later. In the tight junction blockade experiment, mice received an IC infusion of 2,4,6-triaminopyrimidine (TAP, total 30 µmol/mouse) 1 hour before and concurrently with the IC infusion of PAR-4 Agonist Peptide. In disease models, the peptide was administered IC 1 hour before CRD in mice that had previously undergone water avoidance stress (WAS, 1 hour daily for 4 days) or 72 hours after induction of low-grade colitis by IC instillation of TNBS (20 mg/kg in 30% ethanol). [1] |
| References |
[1]. Proteinase-activated receptor-4 evoked colorectal analgesia in mice: an endogenously activatedfeed-back loop in visceral inflammatory pain. Neurogastroenterol Motil. 2012 Jan;24(1):76-85, e13. |
| Additional Infomation |
PAR-4 Agonist Peptide (AYPGFK-NH₂) is a synthetic peptide activator of Proteinase-activated receptor-4 (PAR-4) based on its tethered ligand sequence. It is described as having no effect on PAR-1 or PAR-2, and its effects are blocked by a PAR-4 antagonist (P4pal-10). The study suggests that its analgesic effect in visceral pain models depends on local activation of receptors located on or accessible from the colonic lumen, likely on mucosal nerve terminals, rather than a systemic effect. The results also indicate that endogenous PAR-4 activation, potentially by proteases like cathepsin G which is elevated in TNBS colitis, may serve as a feedback mechanism to attenuate inflammatory colonic hyperalgesia. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 250 mg/mL (~314.54 mM) H2O : ~100 mg/mL (~125.81 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (2.62 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (2.62 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (2.62 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 50 mg/mL (62.91 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2581 mL | 6.2907 mL | 12.5815 mL | |
| 5 mM | 0.2516 mL | 1.2581 mL | 2.5163 mL | |
| 10 mM | 0.1258 mL | 0.6291 mL | 1.2581 mL |