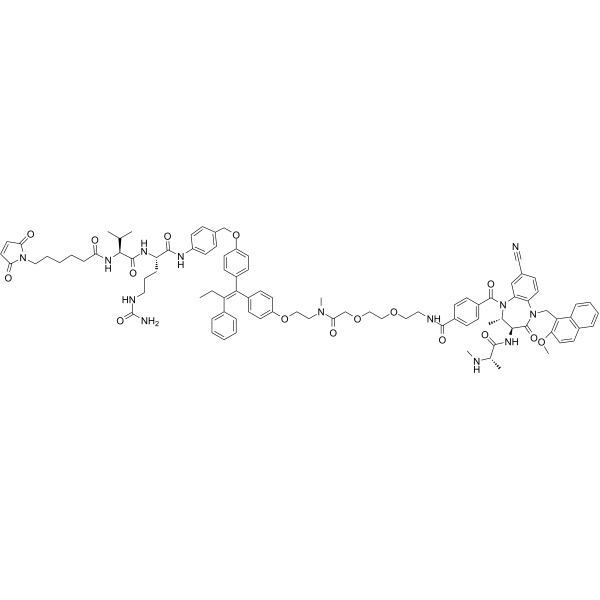Physicochemical Properties
| Molecular Formula | C94H107N13O16 |
| Molecular Weight | 1674.93290352821 |
| Exact Mass | 1674.799 |
| CAS # | 2158322-33-7 |
| Related CAS # | PROTAC ERα Degrader-1;2417369-94-7 |
| PubChem CID | 132144829 |
| Appearance | White to off-white solid powder |
| LogP | 10.2 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 43 |
| Heavy Atom Count | 123 |
| Complexity | 3550 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | C(C1C=CC(C(=O)NCCOCCOCC(=O)N(C)CCOC2C=CC(C(C3C=CC(OCC4C=CC(NC(=O)[C@H](CCCNC(=O)N)NC(=O)[C@H](C(C)C)NC(=O)CCCCCN5C(C=CC5=O)=O)=CC=4)=CC=3)=C(C3C=CC=CC=3)CC)=CC=2)=CC=1)(N1[C@H]([C@@H](C(=O)N(CC2=C(C=CC3C=CC=CC2=3)OC)C2=CC=C(C#N)C=C12)NC(=O)[C@H](C)NC)C)=O |
| InChi Key | AAGUYPGVVXJDLD-GGHCKNFKSA-N |
| InChi Code | InChI=1S/C94H107N13O16/c1-9-74(65-19-12-10-13-20-65)85(68-34-41-73(42-35-68)123-58-63-25-37-71(38-26-63)100-90(114)77(23-18-47-99-94(96)118)101-91(115)86(60(2)3)102-81(108)24-14-11-17-49-105-82(109)45-46-83(105)110)67-32-39-72(40-33-67)122-52-50-104(7)84(111)59-121-54-53-120-51-48-98-89(113)69-28-30-70(31-29-69)92(116)107-62(5)87(103-88(112)61(4)97-6)93(117)106(78-43-27-64(56-95)55-79(78)107)57-76-75-22-16-15-21-66(75)36-44-80(76)119-8/h10,12-13,15-16,19-22,25-46,55,60-62,77,86-87,97H,9,11,14,17-18,23-24,47-54,57-59H2,1-8H3,(H,98,113)(H,100,114)(H,101,115)(H,102,108)(H,103,112)(H3,96,99,118)/b85-74+/t61-,62-,77-,86-,87-/m0/s1 |
| Chemical Name | N-[2-[2-[2-[2-[4-[(Z)-1-[4-[[4-[[(2S)-5-(carbamoylamino)-2-[[(2S)-2-[6-(2,5-dioxopyrrol-1-yl)hexanoylamino]-3-methylbutanoyl]amino]pentanoyl]amino]phenyl]methoxy]phenyl]-2-phenylbut-1-enyl]phenoxy]ethyl-methylamino]-2-oxoethoxy]ethoxy]ethyl]-4-[(3S,4S)-7-cyano-1-[(2-methoxynaphthalen-1-yl)methyl]-4-methyl-3-[[(2S)-2-(methylamino)propanoyl]amino]-2-oxo-3,4-dihydro-1,5-benzodiazepine-5-carbonyl]benzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | HER2 antibodies including PAC Anti-HER2 (Endox-XIAP) significantly reduced the levels of estrogen receptor alpha (ERα) in HER2-expressing cells, with an IC50 of 132 ng/mL. PROTAC-antibody conjugate (PAC) molecules are made up of an antibody attached to a PROTAC through a linker (L1); the PROTAC is made up of a protein binding group, a linker ("L2"), and a ubiquitin E3 ligase binding group ("E3LB"). group ("PB"). The elements that comprise the PAC are explained in the sections that follow. These components are offered in order to create a PAC with optimal therapeutic index and effective efficacy. 1. Antibodies (Ab): While PACs containing antibodies against non-antigens reside on the cell surface, the antibody portion of PAC can target cells expressing the antigen, thereby delivering antigen-specific PACs to target cells intracellularly, usually by endocytosis. Pinocytosis of the PAC may still occur, but the identification of the PROTAC moiety may lead to less targeted intracellular transport of the PROTAC moiety. 2. Linker (L1): To create a PAC, one or more PROTAC moieties (D) can be linked to an antibody (Ab) by a "linker" (L1), which is a bifunctional or multifunctional moiety. L1 with reactive functional groups for covalent attachment to PROTAC and antibodies can be used in some embodiments to prepare PAC. 3. PROTAC(D) [1]. |
| References |
[1]. Protac antibody conjugates and methods of use. WO2017201449A1. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~70 mg/mL (~41.79 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (1.49 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 1.75 mg/mL (1.04 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 17.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.5970 mL | 2.9852 mL | 5.9704 mL | |
| 5 mM | 0.1194 mL | 0.5970 mL | 1.1941 mL | |
| 10 mM | 0.0597 mL | 0.2985 mL | 0.5970 mL |