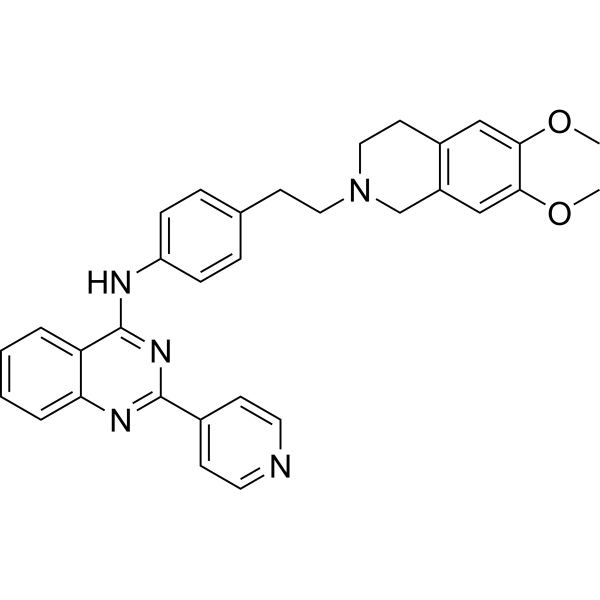
Physicochemical Properties
| Molecular Formula | C32H31N5O2 |
| Molecular Weight | 517.620846986771 |
| Exact Mass | 517.247 |
| CAS # | 2050747-49-2 |
| PubChem CID | 132822387 |
| Appearance | Light yellow to brown solid powder |
| LogP | 5.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 39 |
| Complexity | 742 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C)C1C(=CC2=C(C=1)CN(CCC1C=CC(=CC=1)NC1C3C=CC=CC=3N=C(C3C=CN=CC=3)N=1)CC2)OC |
| InChi Key | TXKOCTPGTLRGNL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H31N5O2/c1-38-29-19-24-14-18-37(21-25(24)20-30(29)39-2)17-13-22-7-9-26(10-8-22)34-32-27-5-3-4-6-28(27)35-31(36-32)23-11-15-33-16-12-23/h3-12,15-16,19-20H,13-14,17-18,21H2,1-2H3,(H,34,35,36) |
| Chemical Name | N-[4-[2-(6,7-dimethoxy-3,4-dihydro-1H-isoquinolin-2-yl)ethyl]phenyl]-2-pyridin-4-ylquinazolin-4-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The target of the compound 12k (a derivative of N-(4-(2-(6,7-Dimethoxy-3,4-dihydroisoquinolin-2(1H)yl)ethyl)phenyl)quinazolin-4-amine) is P-glycoprotein (P-gp, also known as ATP Binding Cassette Transporter, Subfamily B, Member 1, ABCB1). It exhibits inhibitory activity against P-gp, with an EC₅₀ value of 57.9 ± 3.5 nM in reversing doxorubicin (DOX) resistance in K562/A02 cells. Additionally, it inhibits P-gp ATPase activity without affecting CYP3A4 activity or P-gp expression[1] |
| ln Vitro |
P-gp inhibitor 1 (12k) reverses doxorubicin (DOX) resistance in K562/A02 cells for 80 minutes with high efficacy (EC50=57.9±3.5 nM), minimal cytotoxicity, and extended duration of activity (1 μM)[1]. P-gp inhibitor 1 can also block Pgp-mediated Rh123 efflux, raise the accumulation of DOX, inhibit P-gp ATPase in K562/A02 MDR cells Activity (0.1, 1, 5 μM, 1 hour), and boost the potency of other MDR-related cytotoxic medicines with different structures [1]. 1. Reversal of DOX resistance in K562/A02 cells: The compound 12k showed high potency in reversing DOX resistance in K562/A02 cells, with an EC₅₀ of 57.9 ± 3.5 nM, and exhibited low cytotoxicity to K562/A02 cells at the concentration effective for reversing drug resistance[1] 2. Enhancement of efficacy of other MDR-related cytotoxic agents: 12k could increase the potency of various MDR-related cytotoxic agents with different chemical structures in K562/A02 cells, improving their inhibitory effects on the growth of drug-resistant cells[1] 3. Increase in intracellular DOX accumulation: When K562/A02 cells were treated with 12k, the intracellular accumulation of DOX was significantly elevated, which was attributed to the inhibition of P-gp-mediated drug efflux[1] 4. Blockade of P-gp-mediated Rh123 efflux: 12k could block the efflux of rhodamine 123 (Rh123), a substrate of P-gp, from K562/A02 cells. After treatment with 12k, the intracellular retention of Rh123 was increased, indicating the inhibition of P-gp's efflux function[1] 5. Inhibition of P-gp ATPase activity: 12k suppressed the ATPase activity of P-gp in K562/A02 cells, which is closely related to the inhibition of P-gp's drug transport function (as P-gp relies on ATP hydrolysis to drive drug efflux)[1] 6. No effect on CYP3A4 activity and P-gp expression: Treatment of relevant cells with 12k did not alter the activity of the cytochrome P450 enzyme CYP3A4, nor did it affect the expression level of P-gp (neither protein nor mRNA level changes were observed)[1] |
| ln Vivo |
1. Pharmacokinetic properties: The compound 12k exhibited favorable in vivo pharmacokinetic properties, including a good half-life (t₁/₂) and oral bioavailability, which supports its potential for oral administration in clinical applications[1] 2. Effect on DOX metabolism: 12k had no impact on the metabolism of DOX in vivo. This characteristic is crucial because it avoids the side effects that would otherwise be caused by increased plasma concentrations of cytotoxic agents (such as DOX) due to altered metabolism[1] |
| Enzyme Assay |
1. P-gp ATPase activity assay: First, P-gp-containing samples were prepared from K562/A02 cells (e.g., by isolating cell membranes enriched with P-gp). Then, the samples were incubated with different concentrations of 12k and a constant amount of ATP (the substrate for P-gp ATPase). During the incubation period, P-gp catalyzes the hydrolysis of ATP to ADP and inorganic phosphate (Pi). After the reaction, the amount of Pi generated (an indicator of ATPase activity) was detected using a specific detection method (e.g., a colorimetric or fluorometric assay). The effect of 12k on P-gp ATPase activity was evaluated by comparing the Pi production in the presence and absence of 12k; a decrease in Pi production indicated that 12k inhibited P-gp ATPase activity[1] 2. CYP3A4 activity assay: An enzyme reaction system containing recombinant CYP3A4 enzyme, a specific CYP3A4 substrate (e.g., a fluorescent or luminescent substrate), and cofactors required for CYP3A4 activity was established. Different concentrations of 12k were added to the system, and the mixture was incubated at an appropriate temperature for a certain period. After the incubation, the amount of the metabolite generated from the substrate (which reflects CYP3A4 activity) was detected using a suitable analytical method (e.g., high-performance liquid chromatography or fluorescence detection). By comparing the metabolite production in the presence and absence of 12k, it was determined that 12k had no effect on CYP3A4 activity[1] |
| Cell Assay |
Western Blot Analysis[1] Cell Types: K562/A02 Cell Tested Concentrations: 0.1, 0.5 or 2.0 μM Incubation Duration: 72 hrs (hours) Experimental Results: MDR reversal of 12k is not caused by diminished protein expression but is most likely due to direct inhibition of P-gp Outflow[1]. 1. K562/A02 cell drug resistance reversal assay (MTT method): K562/A02 cells were seeded into 96-well plates at an appropriate density and cultured for a certain period to allow cell adhesion. Then, different concentrations of 12k were added to the wells, followed by the addition of a fixed concentration of DOX (a cytotoxic agent to which K562/A02 cells are resistant). After co-incubation for a specific time (e.g., 48 hours), MTT reagent was added to each well and incubated continuously. The formazan crystals formed by viable cells were dissolved with a solubilization solution, and the absorbance at a specific wavelength was measured using a microplate reader. The cell viability was calculated based on the absorbance values, and the EC₅₀ value of 12k for reversing DOX resistance was determined by analyzing the dose-response relationship[1] 2. Intracellular DOX accumulation assay: K562/A02 cells were harvested in the logarithmic growth phase and resuspended in a suitable buffer to adjust the cell concentration. Different concentrations of 12k were added to the cell suspension and pre-incubated for a certain time. Then, DOX was added to the mixture to a final concentration and incubated for a specified period. After incubation, the cells were washed with ice-cold buffer to remove extracellular DOX and 12k. The intracellular fluorescence intensity of DOX was detected using a flow cytometer, and the relative accumulation of DOX in cells was calculated by comparing the fluorescence intensity in the presence and absence of 12k[1] 3. Rh123 efflux assay: Logarithmically growing K562/A02 cells were collected and resuspended in buffer. Rh123 was added to the cell suspension to a final concentration and incubated at 37°C for a certain time to allow Rh123 to enter the cells. The cells were then washed with ice-cold buffer to remove extracellular Rh123, and different concentrations of 12k were added to the cell suspension. After incubation at 37°C for a specified period, the cells were analyzed using a flow cytometer to detect the intracellular fluorescence intensity of Rh123. The efflux rate of Rh123 was evaluated by comparing the fluorescence intensity in the presence and absence of 12k, and the ability of 12k to block P-gp-mediated Rh123 efflux was determined[1] 4. Cytotoxicity assay for 12k: K562/A02 cells were seeded into 96-well plates at an appropriate density and cultured for a period. Different concentrations of 12k (without DOX) were added to the wells and incubated for 48 hours. MTT reagent was added, and the subsequent steps were the same as the MTT method described above. The cell viability was calculated, and the cytotoxicity of 12k alone to K562/A02 cells was evaluated[1] 5. P-gp expression detection (western blot and PCR): K562/A02 cells were treated with different concentrations of 12k for a certain time. For western blot analysis, the cells were lysed with a lysis buffer to extract total protein, and the protein concentration was determined. Equal amounts of protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a polyvinylidene fluoride (PVDF) membrane. The membrane was blocked with a blocking solution and then incubated with a primary antibody against P-gp and a secondary antibody labeled with a horseradish peroxidase (HRP) conjugate. The protein bands were visualized using an enhanced chemiluminescence (ECL) detection system, and the relative expression level of P-gp protein was quantified by densitometric analysis. For PCR analysis, total RNA was extracted from the treated cells using an RNA extraction kit, and complementary DNA (cDNA) was synthesized by reverse transcription. PCR amplification was performed using specific primers for P-gp and a housekeeping gene (as an internal reference). The amplification products were analyzed by agarose gel electrophoresis, and the relative expression level of P-gp mRNA was determined by comparing the band intensity with the internal reference[1] |
| Animal Protocol |
1. Pharmacokinetic study of 12k in rats: Male Sprague-Dawley rats were selected and acclimated to the experimental environment for a certain period before the experiment. 12k was formulated into a suitable oral dosage form (e.g., suspended in a solution containing a solubilizer) and an intravenous injection dosage form. For the oral administration group, rats were given 12k at a specific dose by gavage. For the intravenous injection group, rats were injected with 12k at a specific dose via the tail vein. Blood samples were collected from the orbital venous plexus at different time points after administration and placed in heparinized tubes. The plasma was separated by centrifugation, and the concentration of 12k in plasma was determined using a sensitive analytical method (e.g., liquid chromatography-tandem mass spectrometry, LC-MS/MS). Pharmacokinetic parameters such as half-life (t₁/₂), oral bioavailability (F), area under the concentration-time curve (AUC), peak plasma concentration (Cₘₐₓ), and time to peak concentration (Tₘₐₓ) were calculated using pharmacokinetic software[1] 2. In vivo study on the effect of 12k on DOX metabolism: Male Sprague-Dawley rats were randomly divided into groups, including a control group (treated with DOX alone) and an experimental group (treated with 12k plus DOX). 12k was administered to the experimental group at a specific dose and frequency (e.g., oral gavage once a day for several consecutive days), and DOX was administered at a specific dose via intravenous injection or another route on a specific day. Blood samples were collected at different time points after DOX administration, and plasma was separated. The concentration of DOX and its metabolites in plasma was determined using LC-MS/MS or other suitable methods. The pharmacokinetic parameters of DOX in the two groups were compared to evaluate whether 12k had an effect on DOX metabolism[1] |
| ADME/Pharmacokinetics |
1. Half-life (t₁/₂): 12k had a good half-life in rats after oral or intravenous administration[1] 2. Oral bioavailability (F): 12k exhibited favorable oral bioavailability in rats[1] 3. Effect on drug metabolism: 12k did not affect the metabolism of DOX in vivo[1] |
| Toxicity/Toxicokinetics |
1. In vitro cytotoxicity: 12k showed low cytotoxicity to K562/A02 cells at the concentration effective for reversing drug resistance[1] 2. Drug-drug interaction: 12k did not affect the activity of CYP3A4, indicating a low potential for drug-drug interactions involving CYP3A4 substrates[1] |
| References |
[1]. Design, Synthesis, and Pharmacological Characterization of N-(4-(2 (6,7-Dimethoxy-3,4-dihydroisoquinolin-2(1H)yl)ethyl)phenyl)quinazolin-4-amine Derivatives: Novel Inhibitors Reversing P-Glycoprotein-Mediated Multidrug Resistance. J Med Chem. 2017 Apr 27;60(8):3289-3302. |
| Additional Infomation |
1. Background: P-glycoprotein (P-gp)-mediated multidrug resistance (MDR) is a major barrier to successful cancer chemotherapy. 12k is a novel P-gp inhibitor with a quinazoline scaffold, which was identified as the most promising candidate for in-depth study among the synthesized derivatives[1] 2. Mechanism of action: 12k reverses P-gp-mediated MDR mainly by inhibiting the efflux function of P-gp (blocking the efflux of substrates such as DOX and Rh123) and suppressing P-gp ATPase activity, without altering P-gp expression or affecting CYP3A4 activity[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~16.67 mg/mL (~32.21 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9319 mL | 9.6596 mL | 19.3192 mL | |
| 5 mM | 0.3864 mL | 1.9319 mL | 3.8638 mL | |
| 10 mM | 0.1932 mL | 0.9660 mL | 1.9319 mL |